We keep building new advantages, creating quality h3po4 phosphoric acid, Nitroso R salt, tetravisc with our strength and commitment, and allowing superior technology to stand firm in the global market. Our independent innovation capability continues to improve, and our technological competitiveness continues to increase. How about to start your good organization with our corporation? We are all set, properly trained and fulfilled with pride.
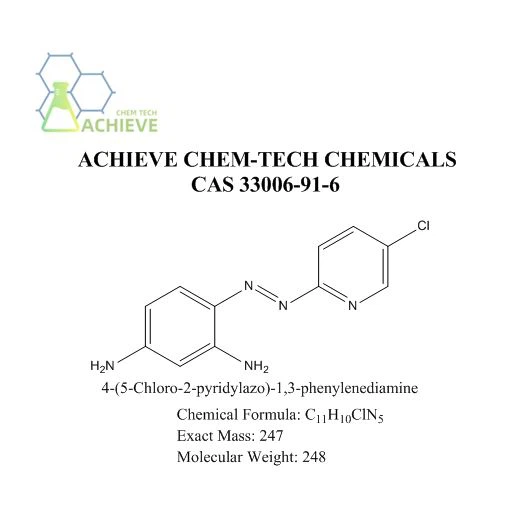
4-(5-Chloro-2-pyridylazo)-1,3-phenylenediamine we provide is 98% pure substance, Dithizone Indicator Reagent, we achieve it with our technology.
Spectrophotometric Determination of Trace Strontium with 4- (5-chloro-2-pyridylazo) - 1,3-diaminobenzene.The color reaction of strontium (Ⅱ) with 4- (5-chloro-2-pyridylazo) - 1,3-diaminobenzene was studied in the presence of sodium dodecyl sulfonate (SDS) micelle solution The test shows that in acetic acid sodium acetate buffer solution with pH 6.0, strontium (Ⅱ) forms a 1:1 yellow complex with the reagent. The maximum absorption peak of the complex is at 460 nm wavelength, and the apparent molar absorption coefficient ε Is 1.29 × In 105.25 ml solution, strontium (II) content is 0 ~ 12 μ The method has been applied to the determination of Trace Strontium in aluminum alloys and chemical reagents with satisfactory results.

|
|
|
|
Chemical Formula |
C11H10ClN5 |
|
Exact Mass |
247.06 |
|
Molecular Weight |
247.79 |
|
m/z |
247.06 (100.0%), 249.06 (32.0%), 248.07 (11.9%), 250.06 (3.8%), 248.06 (1.8%) |
|
Elemental Analysis |
C, 53.34; H, 4.07; Cl, 14.31; N, 28.28 |

4-(5-Chloro-2-pyridylazo)-1,3-phenylenediamine shows the phase separation conditions of cobalt-4- ( 5-chloro-2-pyridylazo ) -1,3-diaminobenzene ( 5-Cl-PADAB ) complex with TritonX-100 at cloud point were studied. The complex was enriched by TritonX-100 when the micelle solution was heated to 92 ± 1 °C for 40 min in pH 4.0 ~ 6.0 medium. The absorbance of the enrichment solution was measured at 575 nm. Beer ' s law was obeyed for cobalt content in the range of 0 – 4 μg / 5 mL. The interference ions could be eliminated by adding H _ 2SO _ 4 into TritonX-100 phase separation solution. The proposed method is sensitive, simple and has been used for the direct determination of trace cobalt in human hair and tap water without separation.
Analytical Chemistry
5-Cl-PADAB is a highly selective and sensitive colorimetric reagent for the determination of specific metals, particularly cobalt and cadmium. When this reagent reacts with cobalt or cadmium ions, it forms stable and colored metal-ligand complexes. The color intensity of these complexes is directly proportional to the concentration of the metal ions in the solution.
Mechanism of Reaction
- The reaction between 5-Cl-PADAB and cobalt or cadmium ions involves the coordination of the metal ions to the nitrogen atoms in the pyridylazo and phenylenediamine moieties of the reagent. This coordination leads to the formation of a colored complex, which has a distinct absorption spectrum in the visible region of the electromagnetic spectrum.
Analytical Procedure
- To perform a quantitative analysis using 5-Cl-PADAB, a known volume of the sample containing the metal ions of interest is mixed with a known concentration of the reagent. The mixture is then allowed to react for a specific period, typically a few minutes, to ensure complete complex formation. The color intensity of the resulting solution is then measured spectrophotometrically at a wavelength corresponding to the maximum absorption of the metal-ligand complex.
- By comparing the measured absorption with a calibration curve obtained using standard solutions of known metal ion concentrations, the concentration of the metal ions in the sample can be determined accurately.
Food Analysis
4-(5-Chloro-2-pyridylazo)-1,3-phenylenediamine is widely used in food analysis, mainly for detecting and analyzing the content of cobalt and cadmium in various foods and agricultural products. These two heavy metals, if present in excessive amounts, will pose potential risks to human health. Therefore, accurate detection of their content is crucial to ensure that food and agricultural products meet national and industrial safety standards, and to safeguard the health of consumers.
Industrial Quality Control
In the field of industrial quality control, this reagent also plays an important role. It is used to monitor the concentration of cobalt and cadmium in industrial process streams and final products. The content of these two elements directly affects the performance and quality of industrial products; at the same time, effective monitoring of their concentration in process streams helps to optimize production processes, achieve strict process control, and ensure the stability and qualification rate of final products.
|
|
|
Research and Development
Due to its specific chemical properties, 4-(5-chloro-2-pyridylazo)-1,3-phenylenediamine is often used in scientific research to study the interactions between ligands and metal ions, as well as in the development of new analytical methods for the determination of trace metals.
Interactions Between Ligands and Metal Ions
5-Cl-PADAB can act as a ligand, binding to metal ions through its azo (-N=N-) and pyridyl (-C5H4N-) groups. These interactions are crucial in understanding the coordination chemistry of metal ions, which involves the formation of metal-ligand bonds. By studying these interactions, researchers can gain insights into:
Stability Constants: Determining the strength of the metal-ligand bonds and the stability of the resulting complexes.
Structure and Geometry: Understanding the spatial arrangement of the ligand around the metal ion and the overall structure of the complex.
Spectroscopic Properties: Investigating the absorption and emission properties of the metal-ligand complexes, which can provide information about the electronic structure and bonding nature.
Development of New Analytical Methods
5-Cl-PADAB's sensitivity and selectivity towards certain metal ions make it an excellent candidate for the development of new analytical methods for trace metal determination. These methods can include:

Spectrophotometric Methods
Utilizing the color change upon complex formation to quantify metal ions in solution. The intensity of the color is proportional to the concentration of the metal ion, allowing for quantitative analysis.
Chromatographic Methods
Incorporating 5-Cl-PADAB as a derivatizing agent to enhance the detection of metal ions in chromatographic separations. This can improve the sensitivity and selectivity of the chromatographic methods.


Sensor Development
Employing 5-Cl-PADAB in the design of optical sensors for metal ions. These sensors can provide real-time monitoring of metal ion concentrations in various environments.
Environmental Monitoring
In environmental monitoring, this compound can be used to detect and quantify heavy metals in water, soil, and air samples. The presence of heavy metals in the environment can have adverse effects on ecosystems and human health, so their accurate determination is crucial for environmental protection.
Environmental Implications of Heavy Metals
Heavy metals like cobalt and cadmium can enter the environment through various anthropogenic and natural sources, including industrial emissions (such as waste gas and wastewater from smelting, electroplating and chemical industries), agricultural runoff (carrying heavy metals from pesticides, fertilizers and sewage irrigation), improper waste disposal (of heavy metal-containing products and industrial residues), and natural processes (like rock weathering and volcanic activity). Their persistent presence in water, soil, and air can have long-term adverse effects on ecosystems and human health, with specific impacts including the following aspects:
Water Contamination: Heavy metals such as cobalt and cadmium in surface water and groundwater are difficult to degrade naturally and can easily accumulate in aquatic organisms (including fish, shellfish and algae) through bioaccumulation. As these organisms enter the food chain, heavy metals will gradually enrich at higher trophic levels, ultimately posing serious health risks to human consumers who ingest them.
Soil Pollution: When heavy metals enter the soil, they can bind to soil particles and persist for a long time, resulting in soil degradation. Contaminated soil can lead to the absorption and accumulation of heavy metals by crops and other plants; these heavy metals cannot be metabolized by plants and will remain in edible parts, which can then be ingested by humans and animals, affecting their physical health.
Air Quality: Heavy metals released into the air (mostly in the form of particulate matter) from industrial emissions and other sources can float in the atmosphere for a long time. They can be directly inhaled by humans and animals, causing damage to the respiratory system, or deposited on the surface of soil and plants, further expanding environmental pollution and leading to potential long-term health risks.

Role of 5-Cl-PADAB in Environmental Monitoring
5-Cl-PADAB is a reliable colorimetric reagent for the determination of cobalt and cadmium in environmental samples due to its high selectivity and sensitivity. The procedure involves:
Sample Collection: Collecting water, soil, or air samples from the environment.
Sample Preparation: Preparing the samples for analysis by dissolving or extracting the heavy metals if necessary.
Reagent Addition: Adding a known concentration of 5-Cl-PADAB to the sample.
Complex Formation: Allowing the reagent to react with the heavy metal ions to form colored complexes.
Spectrophotometric Measurement: Measuring the color intensity of the solution at a specific wavelength using a spectrophotometer.
Concentration Calculation: Comparing the measured absorption with a calibration curve to determine the concentration of cobalt and cadmium in the sample.

4-(5-Chloro-2-pyridylazo)-1,3-phenylenediamine, commonly abbreviated as Cl-PADA, is a versatile chemical compound belonging to the azo dye class. It is characterized by its unique molecular structure, which incorporates both a pyridyl ring substituted with a chlorine atom and a phenylenediamine moiety linked through an azo (-N=N-) bridge. This specific arrangement the compound distinct properties and applications in various fields.
Chemically, Cl-PADA exhibits a dark red to violet appearance, depending on its state and concentration. It is soluble in organic solvents such as ethanol and dimethylformamide, making it suitable for use in solution-based reactions and analytical procedures. The presence of the azo group is crucial for its color-forming ability, contributing to its utility in dyeing and staining applications.

In analytical chemistry, Cl-PADA finds application as a sensitive and selective chelating agent for metal ions, particularly those of nickel, cobalt, and copper. Its ability to form colored complexes with these metals allows for their quantitative determination through spectrophotometric methods. This analytical significance stems from the compound's high molar absorptivity and stability of the formed metal-ligand complexes.

Moreover, Cl-PADA's reactivity and spectral properties make it a valuable tool in the study of metal ion speciation, kinetic investigations, and environmental monitoring, where trace metal detection is paramount. Its use in these contexts underscores its importance in advancing our understanding and management of metal contaminants in natural and engineered systems.
In summary, 4-(5-Chloro-2-pyridylazo)-1,3-phenylenediamine is a remarkable azo dye with diverse applications rooted in its unique chemical structure and properties. Its capacity to form stable, colored complexes with specific metal ions has established it as a cornerstone in analytical chemistry, contributing to advancements in environmental science, material science, and beyond.
FAQ
What is another name for phenylenediamine?
+
-
Synonyms: 4-Phenylenediamine base; p-Diaminobenzene; Pelagol D; Renal PF; Futramine D; Fur Black 41866; C.I. Developer 12; Developer PF; PPD; Peltol D; BASF Ursol D; Tertral D; 4-Aminoaniline; 1,4-Diaminobenzene; Phenyhydrazine; 1,4-Benzenediamine; 1,4-Phenylenediamine; C.I.
What is 1 2-phenylenediamine used for?
+
-
1,2-phenylenediamine appears as colorless monoclinic crystals if pure; technical grade brownish-yellow crystals or a sandy brown solid. Used in manufacture of dyes, photography, organic synthesis.
What is meta phenylenediamine used for?
+
-
m-Phenylenediamine is used in the preparation of various polymers including aramid fibers, epoxy resins, wire enamel coatings and polyurea elastomers. Other uses for m-phenylenediamine include as an accelerator for adhesive resins, and as a component of dyes for leather and textiles.
What are the health risks associated with phenylenediamine?
+
-
Acute (short-term) exposure to high levels of p-phenylenediamine may cause severe dermatitis, eye irritation and tearing, asthma, gastritis, renal failure, vertigo, tremors, convulsions, and coma in humans. Eczematoid contact dermatitis may result from chronic (long-term) exposure in humans.
Innovative, durable and cost-efficient products have secured us a position as a leading global manufacturer of Chemical Reagent Bloom Tech Tetrabutylammonium Bromide CAS 1643-19-2. Only by attaching importance to international exchanges and cooperation and mastering the most cutting-edge information in the world can the company achieve rapid development better and faster. We actively pursue internal growth and create superior value for customers.