The corporate brand of cultural level permeates our Indicator Reagent, Pure Tetracaine, melamine powder, which has higher connotation than the product level. We always believe that talents are the first element, the first resource and the first driving force of enterprise development. We care for our employees from the life, emotional and growth aspects.
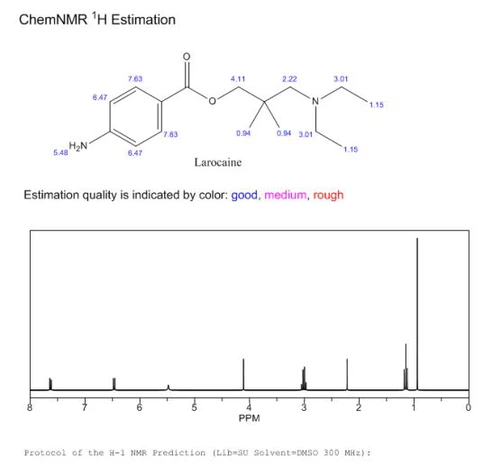
Larocaine powder CAS 94-15-5 is mainly used in laboratory Research and Development process and chemical and pharmaceutical production process. It is a local anesthetic with the chemical name N - [2- (methylamino) ethyl] -4-pyridylformamide or 2- (methylamino) ethyl-N - (4-pyridyl) formamide. It is a white to almost white powder. Its color may vary depending on its source and manufacturing method, and is essentially an analog of 2-methyl-4-pyridinecarboxylic acid (2-MP). Compared to other local anesthetics, it is a weak anesthetic that can be used to alleviate mild pain or itching. The melting point is 142-144 ° C. The melting point can be used as a factor for separately identifying purity and quality. It is a relatively stable compound. It is a low-dose local anesthetic widely used in some medical and research fields. However, Larocaine should be stored in a dry and cool place, avoiding direct light exposure. In addition, Larocaine reacts with acids, so mixing with fiber optic caustic soda should be avoided.

The laboratory has accurately determined his chemical information, and the results are as follow:
|
CF |
C16H26N2O2 |
|
EM |
278 |
|
MW |
278 |
|
m/z |
278 (100.0%), 279 (17.3%), 280 (1.4%) |
|
EA |
C, 69.03; H, 9.41; N, 10.06; O, 11.49 |
|
|
|
|
Structure: The chemical structure of Larocaine consists of three parts: an aryl ring, an acrylic chain, and an imidazole group. The aryl ring is composed of a benzene ring and an amino group, the propylene chain contains two methoxy groups, and the imidazole group contains one nitrogen atom.
Solubility: Larocaine is soluble in organic solvents such as benzene and ethanol, and almost insoluble in water.
Chlorination reaction: Larocaine and chlorine will undergo substitution reaction, in which only one methoxy group will be replaced by chlorine. The product of this reaction is called monochromo Larocaine.
Biosynthesis: Larocaine is synthesized by the reaction of aniline, acrolein, and imidazole in the presence of HCl. The reaction product alkylates propylene and imidazole according to the Auger mechanism, ultimately producing Larocaine.
Other properties: Larocaine powder can also be used as a buffer in electrolytes and as a pain medication. In addition, it can also be used as an intermediate for organic materials.

Remark: BLOOM TECH(Since 2008), ACHIEVE CHEM-TECH is the subsidiary of us.
Addition information of chemical compound: Melting point 53.0 to 57.0 °C, Boiling point 421.21 ° C (rough estimate), Density 1.035, Refractive index 1.5290 (estimated), Acidity coefficient (PKA) 10.08 ± 0.25 (predicted), Steam pressure 1.01E-06mmHg at 25°C, Polarization: 32.9 10-24cm3, Surface tension: 40.1 dyne/cm, Enthalpy of evaporation: 65.48 kJ/mol.
The chemical synthesis of Larocaine powder mainly involves esterification reactions using phenylacetic acid and pyridine as raw materials, followed by stereoselective reduction and alkylation of the obtained esterification products to obtain Larocaine. The specific steps are as follows:
Reaction 1: Esterification reaction of phenylacetic acid and pyridine:
Firstly, phenylacetic acid and pyridine are mixed, and a catalyst dimethyl sulfoxide is added. The esterification reaction is carried out in an inert atmosphere using acetic acid or other solvents. After the reaction is complete, the product N-phenylacetylpyridine is obtained.
Reaction 2: Stereoselective reduction reaction:
Add N-phenylacetylpyridine to the mixture of alumina and acetonitrile, and then add a reducing agent for stereoselective reduction reaction to obtain the product N-(S)-phenylacetyl-4-phenylpiperid-2-one.
Reaction 3: Alkylation reaction:
Add N-(S)-phenylacetyl-4-phenylpiperidine-2-one with excess propanol to a mixture of catalyst chromium trioxide and tryptophan acetate for alkylation reaction to obtain the target product N-propyl-4-phenylpiperidine-2-one (Larocaine).

Larcaine powder is an organic synthesis intermediate and pharmaceutical intermediate, which is mainly used in laboratory research and development process and chemical pharmaceutical production process. It is a synthetic cocaine analogue. In the 1930s, it was used as a local anesthetic in dentistry and ophthalmology, and as a dopamine uptake inhibitor. Therefore, it can not flow in the market, only for experimental purposes.
Dimethicaine, as a local anesthetic, is not primarily used as a precursor for polymers. However, in the broad fields of chemistry and materials science, compounds can be transformed into polymers or polymer precursors with specific properties through different chemical reaction pathways.
Firstly, it should be clarified that polymer precursors typically refer to low molecular weight compounds that can be converted into high molecular weight polymers through polymerization reactions such as addition polymerization, condensation polymerization, etc. These precursors typically have specific functional groups or reactive sites that can react chemically with other molecules to form long chain or network like polymer compounds.
For this substance, functional groups such as amino and ester groups in its molecular structure provide potential chemical reactivity. However, due to its main use in the medical field as a local anesthetic, its research and application as a polymer precursor are relatively limited. Possible functions can be explored from the following aspects:
Synthesis of functional polymers

Drug controlled release system
Local anesthesia can be used to prepare drug controlled release systems by introducing it into a polymer matrix. For example, it can be grafted onto biodegradable polymer chains to form drug carriers with sustained release capabilities. This carrier can slowly release lidocaine in the body, thereby achieving long-term local anesthesia effects. Although this application does not directly involve the function of dicaaine as a polymer precursor, it demonstrates its potential application in the synthesis of functional polymers.
Antibacterial polymer
Has certain antibacterial properties (although this is not prominent in mainstream literature, drugs with similar structures may have similar properties). Therefore, polymer materials with antibacterial properties can be prepared by introducing themselves or their derivatives into the polymer chain. This material has broad application prospects in fields such as healthcare, hygiene, and packaging.
Chemical reactivity

Amidation reaction
The amino groups in this substance can undergo amidation reactions with acylating reagents such as acyl chlorides to generate corresponding amide derivatives. This reaction provides the possibility for it to serve as a polymer precursor. By controlling the reaction conditions and the ratio of reactants, polymer compounds with different content and distribution of amide groups can be prepared. These compounds may have unique physical and chemical properties, such as good solubility, mechanical properties, and thermal stability.
Alkylation reaction
Its amino or ester groups can also participate in alkylation reactions, generating compounds with different alkyl chain lengths and structures. These compounds can be further used as polymer precursors to prepare polymer materials with specific properties through polymerization reactions. For example, compounds with long alkyl chains may have lower glass transition temperatures and better flexibility; Compounds with short alkyl chains may have higher rigidity and strength.


White to yellow crystal, easy to change color, 2-8 ℃ storage for 10-15 days will become yellow. Larocaine is a local anesthetic. Its successor product is a local anesthetic preparation. Dimethocaine is often used as a local anesthetic in dentistry and ophthalmology. Therefore, it should be widely used in beauty and makeup. It is often used in hydrochloride form for ease of use. Inhalation: If inhaled, move victim to fresh air. Skin contact: Take off contaminated clothing and rinse skin thoroughly with soapy water and clean water. Seek medical advice if you feel unwell. Eye contact: separate eyelid and rinse with running water or normal saline. Seek immediate medical attention. Eat: gargle, do not vomit. Seek immediate medical attention.
Dimethicaine, as a local anesthetic, its chemical stability and reactivity are crucial for its application in medical and industrial fields.
1. Chemical stability
(1). General stability
It usually exhibits good chemical stability at room temperature and pressure. This means that under normal storage and usage conditions, it is not easy for the two to spontaneously decompose or deteriorate.
(2). Storage conditions
To maintain stability, it is recommended to store it in a sealed, dry environment at room temperature. Avoid direct sunlight and high temperatures to prevent drug degradation or deterioration.
(3). Influencing factors
Although it is generally stable, its stability may be affected under extreme conditions such as high temperature, strong acid, strong alkali, or oxidizing agents. These conditions may lead to decomposition or the production of harmful substances.
2. Reactivity
(1). Functional group reactivity
Its molecule contains functional groups such as amino and ester groups, which endow dicaaine with certain reactivity.
Amino (NH2) is a basic functional group that can react with acids to form corresponding ammonium salts.
Ester group (COO) is a functional group that can undergo hydrolysis reactions and can be broken to generate corresponding acids and alcohols under the catalysis of acids or bases.
(2). Typical reactions
Acylation reaction: to transform into the corresponding amide derivative through the action of acyl chloride. This reaction is of great significance in organic synthesis and can be used to prepare compounds with specific biological activities.
Alkylation reaction: The amino or ester groups in can also participate in the alkylation reaction, generating secondary or tertiary amine compounds. This reaction also has wide applications in organic synthesis and drug development.
(3). Potential risks
Due to its certain reactivity, special attention should be paid to avoiding contact with incompatible substances during processing and storage. For example, it should be avoided to store it in combination with highly reactive substances such as oxidants, acids, and bases to prevent accidental reactions from causing danger
Safety Profile: Balancing Efficacy and Toxicity
While Larocaine is generally safe when used as directed, its margin of safety is narrower than that of lidocaine, necessitating careful dosing and monitoring.
Adverse Effects:
Local reactions: Erythema, itching, or edema at the injection site (5–10% of cases).
Systemic toxicity: Rare but possible with accidental intravenous injection or overdose. Symptoms include:
Central nervous system (CNS): Tremors, seizures, coma (due to NaV channel blockade in the brain).
Cardiovascular: Hypotension, bradycardia, arrhythmias (from reduced myocardial excitability).
Contraindications:
Hypersensitivity to amide anesthetics (e.g., lidocaine, bupivacaine).
Severe hepatic impairment (reduced metabolism increases toxicity risk).
Third-degree heart block (risk of asystole).
Drug Interactions:
CYP3A4 inhibitors (e.g., ketoconazole, erythromycin): May increase plasma levels of Larocaine by inhibiting its metabolism, raising toxicity risk.
Antiarrhythmics (e.g., amiodarone): Additive effects on cardiac conduction, increasing arrhythmia risk.
Larocaine powder occupies a unique position in the world of local anesthetics, offering a blend of efficacy and versatility that appeals to medical professionals and, unfortunately, illicit actors. While its clinical uses in dentistry, surgery, and veterinary medicine are valuable, its role as a cocaine adulterant underscores the dangers of unregulated substances. As research continues, Larocaine may find new legitimate applications, but its legacy will always be tinged by the shadows of misuse and controversy. For now, caution, education, and strict regulation remain essential to ensuring its safe and responsible use.
We actively promote green manufacturing, develop environment-friendly BMK Powder CAS 94-15-5 with Best Price, and build a beautiful China together. We hope that based on the accumulated experience, we will do our best to achieve the goal of building a well-off society in an all-round way. The experienced engineers in our R&D department have the ability to develop and design products very efficiently to meet the various needs of different customers.