Our goal is to satisfy our customers by offering golden service, good price and 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID, Raw Materials, Solvent. The company takes respect, communication, cooperation, and improvement as its management philosophy, improves its management process, updates technology and equipment, and continuously improves product quality and reduces product costs. In the ever-changing business environment, the speed of response to the market often determines the success or failure of enterprises.
37 Formaldehyde is an organic chemical, also known as formaldehyde, which is an organic compound with the chemical formula CH2O, CAS 50-00-0, The relative molecular weight is 30.03, the melting point is -92 ℃, the boiling point is -19.5 ℃, and the relative density is 0.815g/cm ³. A 35-40% formaldehyde aqueous solution is commonly known as formalin solution. It is a colorless and irritating gas that has a stimulating effect on the eyes, nose, etc. Easy to dissolve in water and ethanol. The concentration of aqueous solution can reach up to 55%, usually 35% -40%, and typically 37% formaldehyde, known as formaldehyde water or formalin. Has reducibility, especially stronger in alkaline solutions. Can burn, steam and air form explosive mixtures. It has a wide range of applications in industries such as petrochemicals, pharmaceuticals, textiles, biochemistry, as well as energy and transportation. It can be used as a disinfectant and preservative, and can also be used to prepare various products such as phenolic resins, urea formaldehyde resins, melamine resins, urotropin, and pentaerythritol. Formaldehyde has a strong irritant and tear jerking effect on mucous membranes, can cause protein coagulation, and can easily make the skin hard or even cause local tissue necrosis when touched

| Chemical Formula | CH2O |
| Exact Mass | 30 |
| Molecular Weight | 30 |
| m/z | 30 (100.0%), 31 (1.1%) |
| Elemental Analysi | C, 40.00; H, 6.71; O, 53.28 |
| Melting point | − 15 °C |
| boiling point | 97 °C ( 37 % solution ), − 19.5 °C ( pure ), |
| Density | 1.09 g / mL at 25 °C ( lit. ) |
| Vapor density | 1.03 ( vs air ) |
| Color APHA | ≤ 10, pH ( 25 °C ) : 7.0 – 7.5 |
| Solubility water | Acidity coefficient ( pKa ) 13.27 ( at 25 °C ) |
|
|
|
|

37 Formaldehyde has a wide range of applications in industries such as petrochemicals, pharmaceuticals, textiles, biochemistry, energy, and transportation. It can be used as a disinfectant and preservative, as well as in the preparation of various products such as phenolic resins, urea formaldehyde resins, melamine resins, urotropin, and pentaerythritol. Formaldehyde has a strong irritant and tear jerking effect on mucous membranes, can cause protein coagulation, and can easily make the skin hard or even cause local tissue necrosis when touched.

Synthetic resin
The maximum use of formaldehyde is in the production of urea formaldehyde resin, phenolic resin, and melamine formaldehyde resin, which are widely and extensively used in the wood processing industry, household and building decoration industry, furniture industry, etc. Secondly, they are used as additives in paper, textile, leather processing, concrete plasticizers, molding materials, casting materials, insulation materials, flame retardant coatings, and flocculants for sewage treatment. Amino resin is also used in the manufacture of molding materials, used in amino plastic products, electrical materials, building materials, and tableware substitutes. Phenolic resin is also used in the manufacture of automotive brake pads, equipment, telephones, and printing equipment. Special phenolic resin is also used in industries such as aerospace and electronics.
Synthetic polyols
Formaldehyde is an important raw material for the synthesis of polyols, widely used in the production of pentaerythritol (dipentaerythritol), trihydroxymethylpropane, trihydroxymethyl ethane, neopentyl glycol, dihydroxymethyl propionic acid, and 1,4-butanediol by alkyne aldehyde method.


Synthetic fibers and dyeing and finishing auxiliaries
The earliest synthetic fiber, vinylon fiber, was produced using formaldehyde as raw material, mainly used for low-grade clothing, industrial packaging materials, and tire cord threads. There is still some production and application in China, North Korea, and Japan.
Urea formaldehyde addition products, hydroxymethylurea and dihydroxymethylurea, are excellent fiber processing agents used to treat fabrics, blends of fibers with synthetic fibers or wool, which can give them wrinkle resistance, crush resistance, flame resistance, shrinkage resistance, and non ironing properties. Therefore, they have a large application market in permanent forming finishing of fabrics.
The use of hydroxymethylmelamine derivatives and their etherification products for fabric finishing can result in high-quality surface coatings, which have better water washing resistance than hydroxymethylurea finishing agents. Tetrahydroxymethylphosphonium chloride (THPC) is an excellent fireproof agent for cotton fibers, as well as an effective antibacterial and antifungal agent, mainly used for linen fabric finishing. A white fabric finishing agent can be made by reacting formaldehyde, urea, and ethylamine.


Synthetic rubber and additives
Formaldehyde also has a wide range of applications in the preparation of rubber additives. The types of additives prepared with formaldehyde include: tert butylphenol formaldehyde thickening resin, para tert butylphenol formaldehyde resin, octylphenol formaldehyde thickening resin, phenolic reinforcement resin, antioxidants 3114, 1222, 702, and 2246, vulcanizing agent MOCA, vulcanizing agent VA-2, methylene bis (stearamide), 2,4,6-tris (dimethylaminomethyl) phenol, light stabilizer Irgastab 2002, etc.
Pesticide chemicals
Formaldehyde is the main raw material for the important pesticide chemical glyphosate. In China, the pesticide chemicals produced using formaldehyde (polyformaldehyde) mainly include the following varieties: glyphosate, glyphosate, chlorfenapyr, triazolone, thalonil, oat spirit, imidacloprid, methoxam, mequat, imidacloprid, tert butyl phosphate, vegetable phosphorus, isopropylphosphate, etc.
slow release fertilizer
Formaldehyde aqueous solution can also be directly used to treat crop seeds and roots, which can prevent black spot disease and strengthen roots and roots. During the flowering period of rice, an appropriate amount of formaldehyde solution can be sprayed in the field to prevent diseases and increase yield.

Daily chemicals
Formaldehyde is an important raw material used for the synthesis of certain daily chemicals, especially for the synthesis of certain fragrances and their intermediates, such as linalool, p-hydroxybenzaldehyde, p-methoxybenzyl alcohol (anise alcohol), p-methoxybenzaldehyde (anise aldehyde), vanillin (vanillin), lily aldehyde (lily aldehyde), cyclamen aldehyde, jasmonate, musk, amber acetate, dihydroxyacetone, etc.
Antiseptic solution
35% -40% formaldehyde aqueous solution, commonly known as formalin, has anti-corrosion and antibacterial properties and can be used to soak biological specimens, disinfect seeds, etc. However, due to protein denaturation, the specimens are prone to becoming brittle.
The main reason why formaldehyde has anti-corrosion and antibacterial properties is that formaldehyde can react with the amino groups on the proteins that make up living organisms.


Mmedical use
As a fixative, the key to the effective fixation effect of 37 Formaldehyde is the formation of cross-linked chains between protein end groups. The functional groups involved in formaldehyde fixation of proteins are mainly amino, imino, acylamino, peptide, guanidine, hydroxyl, hydrophobic, and aromatic rings. The reaction between formaldehyde and histones is diverse and complex, as it can bind to various functional groups and form bridging bonds between them in most cases. Formaldehyde has this cross-linking function, which is also its disadvantage. In tissues fixed with formaldehyde
immunohistochemistry is required, and enzyme digestion or hot antigen repair methods are often advocated to break the aldehyde bonds cross-linked between proteins and formaldehyde for subsequent staining. Formaldehyde can be prepared into simple or mixed fixatives. The simplest and easiest method to master is to take 10 ml of formaldehyde solution and add 90 ml of water, which is 10% formalin. Of course, the fixative used now has stricter requirements, and it is best to use buffered formalin fixative, which will be beneficial for future immunohistochemical staining needs.

From an histological perspective, formaldehyde is a good fixative with many advantages: less tissue shrinkage, less damage, and better preservation of intrinsic substances; Fixed and uniform, with strong penetration power; Can harden tissues, enhance tissue elasticity, and facilitate slicing; Can preserve fat and lipid substances; Low cost. Although formaldehyde has the above advantages, they are relative and no substance can be perfect. It also has many disadvantages: it contains a large amount of impurities, such as methanol, which can passivate enzymes and affect reactions; Contains trace amounts of formic acid, causing acidification of the fixative and affecting staining; Can produce formalin pigment, affecting observation; Cannot fix uric acid and carbohydrates; Easy to evaporate, pollute the environment, and can cause specimens to dry up; Can exist for a long time in a fixed organization. Someone has conducted an experiment where after fixing the tissue with formaldehyde and rinsing it in running water for 5 hours, there is still a considerable amount of formaldehyde bound to the protein, but it needs to be removed after a long period of rinsing with running water (24 days). It can be seen that formaldehyde present on tissues cannot be removed because clinical biopsies cannot have such a long time to wash tissues. Therefore, it should be pointed out that in various subsequent technical operations, special attention should be paid to the presence of formaldehyde, and methods must be found to remove it, otherwise it will affect various staining and even lead to failure.

In the early days, formaldehyde was mainly used as a disinfectant and preservative in the pharmaceutical industry. Formaldehyde has a wide range of applications in the preservation of animal tissues, as well as in the prevention of bacterial and fungal corrosion in wax products, insect glue products, fat products, starch products, sheep teeth products, fragrant flowers, oils, and colored fabrics.
Formaldehyde is widely used in the synthesis of many drugs and intermediates, such as glycine, sodium sarcosinate, tryptophan, metamaterial, calcium pantothenate, acrolein, furanone, haloperidol, methyl vinyl ketone, methyl thio sulfoxide, imidazole, 2-methylimidazole, 4-methylimidazole, sodium hydroxymethanesulfonate, salbutamol, bisoprolol, hippuric acid, salicylic acid, ketamine, etc.

Addition reaction
In organic solvents, formaldehyde can undergo catalytic addition reactions with monoolefins to produce dienes or corresponding alcohols. In acetic acid solution, formaldehyde reacts with toluene to form 1-phenyl-1,3-diacetic acid propylene glycol, and formaldehyde reacts with propylene to form 1,3-diacetic acid butanediol. In industry, formaldehyde was used to react with isobutene to produce isoprene, known as the Prins reaction.

In alkaline solution, formaldehyde reacts with hydrogen cyanide to form acetonitrile alcohol (hydroxyacetonitrile) HOCH2CN. In industry, this reaction is used to produce amino acid series products, commonly known as Mannich reaction [21]. To prepare the multivalent chelating agent NTA, N (CH2COOH) 3; Aminoacetonitrile, H2NCH2CN; Methyleneaminoacetonitrile, CH2=NCH2CN; Diethyl cyanamide, HN (CH2CN) 2, etc.

Under the action of catalysts such as acetylene copper, silver, and mercury, formaldehyde reacts with monoalkynes to form alkynes. In industry, the Reppe reaction involves the reaction of two molecules of formaldehyde with one molecule of acetylene to produce 1,4-butanediol, which is then hydrogenated to produce 1,4-butanediol. This reaction is an important method for producing 1,4-butanediol in the current industry.

Formaldehyde reacts with primary amines to form alkylaminomethanol, which is further heated or condensed under alkaline conditions to form tertiary amines.
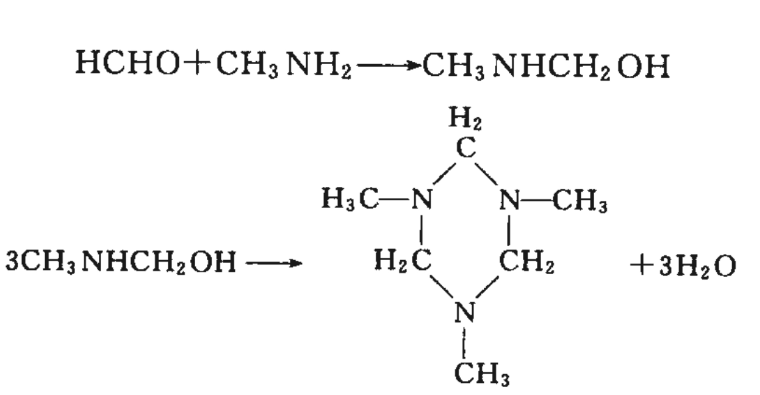
Condensation reaction
37 Formaldehyde itself can slowly undergo condensation reactions, producing lower hydroxy aldehydes, hydroxy ketones, and other hydroxy compounds, which can accelerate the reaction under alkaline conditions. Formaldehyde can undergo condensation reactions with various compounds, commonly known as Tollens reactions. Under alkaline conditions, hydroxymethyl derivatives (- CH2OH) are formed, while under acidic conditions or in the gas phase, methylene derivatives are formed through condensation reactions.

In the presence of alkali, formaldehyde and isobutyraldehyde shrink to form hydroxyaldehyde, which is then reduced to neopentyl glycol with excess formaldehyde under strong alkaline conditions. Formaldehyde is oxidized and reacts with NaOH to form sodium formate.

In the presence of alkali, formaldehyde condenses with n-butanal to form 2,2-dihydroxymethylbutanal, which is further reduced to trimethylolpropane with excess formaldehyde under alkaline conditions.
Aggregation reaction
Due to the presence of two hydrogen atoms on the carbon atom of the carbonyl group in formaldehyde molecules, this unique molecular structure makes formaldehyde very easy to polymerize. However, dry formaldehyde gas is quite stable and only slowly polymerizes at temperatures below 100 ℃. When the newly produced formaldehyde aqueous solution is left to stand, it will automatically generate low molecular weight polymers, forming a mixture of polyoxymethylene glycol, and some precipitation will occur. Formaldehyde aqueous solution will rapidly polymerize and release heat (63 kJ/mol or 15.05 kcal/mol) at room temperature in a closed container. Gaseous formaldehyde can self polymerize at room temperature, and formaldehyde aqueous solution can also self polymerize during the concentration process, generating polyformaldehyde - a white powdery linear structure polymer.

Pure formaldehyde gas can be produced by thermal decomposition of polyformaldehyde or low molecular weight polyoxymethylene monomers (such as trioxane, tetraoxane, etc.), and its formaldehyde purity can reach 90% -100% (volume fraction).
Carbonylation reaction
Under the action of cobalt or rhodium catalysts, formaldehyde can undergo carbonylation reaction with synthesis gas (H2/CO=1-3) at 110 ℃ and 13-15 MPa to produce ethanal, which can be further hydrogenated to produce ethylene glycol. Carbonylation reaction, also known as formaldehyde hydroformylation reaction.

Under the action of transition metal catalysts, liquid or solid acid catalysts, formaldehyde undergoes carbonylation reaction with carbon monoxide to produce glycolic acid, also known as hydroxyacetic acid.

Under the action of Co or Rh transition metal catalysts, formaldehyde undergoes carbonylation reaction with carbon monoxide in the presence of alcohols, producing malonic acid or malonic acid esters.

In the presence of acetamide, formaldehyde undergoes carbonylation reaction to produce acetyl glycine.

Under the action of carbonyl rhodium catalyst and halide promoter, formaldehyde can undergo homologous reaction with synthesis gas to produce acetaldehyde, which is further hydrogenated to produce ethanol.

Decomposition reaction
Formaldehyde has unexpected stability, and its decomposition rate is very slow without catalyst at temperatures below 300 ℃. The decomposition rate of formaldehyde at 400 ℃ is about 0.44% per minute (decomposition pressure of 101.3 kPa or 1 atm), and the main products of decomposition are CO and H2.

Redox reaction
Metals such as Pt, Cr, Cu, and metal oxides (such as Cr2O3, A12O3, etc.) can reduce formaldehyde to methanol, methyl formate, methane, or deeply oxidize formaldehyde to formic acid, CO2, and H2O.


37 Formaldehyde can be obtained by dehydrogenation or oxidation of methanol under the catalysis of silver, copper and other metals, and can also be separated from the oxidation products of hydrocarbons. It can be used as raw materials for phenolic resin, urea-formaldehyde resin, vinylon, urotropine, pentaerythritol, dyes, pesticides and disinfectants. Industrial formaldehyde solution generally contains 37 % formaldehyde and 15 % methanol as inhibitor, boiling point 101 °C.
On October 27, 2017, the World Health Organization International Agency for Cancer Research published a list of carcinogens, putting formaldehyde in a list of carcinogens. On July 23, 2019, formaldehyde was listed in the list of toxic and harmful water pollutants ( the first batch ). In 1923, after the large-scale production of methanol by German BASF company, the large-scale production of industrial formaldehyde has a good raw material foundation. Methanol air oxidation method has become the most commonly used method for the production of industrial formaldehyde. The detection methods of formaldehyde in living room, textiles and food in China and abroad mainly include spectrophotometry, electrochemical detection method, gas chromatography, liquid chromatography, sensor method, etc.
Research Brief History
Formaldehyde was first discovered by Russian chemist Buterelov.
German scientist Hofmann synthesized formaldehyde for the first time by oxidizing methanol with air in the presence of platinum catalyst.
From 1886 to 1889, industrial development work was carried out using copper catalysts.
The German companies Merklin and Losekam produced industrial formaldehyde.
The silver catalyst developed by Blank was introduced.
BASF, a German company, achieved large-scale production of methanol from synthetic gas.
Large-scale production and application of formaldehyde began to develop.
Adkins and Peterson first applied for a patent for iron molybdenum oxide catalysts.
Particle board, particleboard and other artificial boards were used in housing construction. In the mid-1960s, the adverse health effects of formaldehyde, especially its irritation to the eyes and upper respiratory tract, were first reported.
Standards for limiting and regulating formaldehyde emissions from wooden materials were first established in Germany and Denmark.
The global demand for formaldehyde had reached 25.4 million tons, and the demand continued to grow by over 5% annually.
On May 12, 2009, a new study released by the National Cancer Institute in the United States showed that chemical plant workers who were frequently exposed to formaldehyde had a much higher risk of dying from cancers such as leukemia and lymphoma compared to workers who had less exposure to formaldehyde.
Ihe National Cancer Institute in the United States discovered that formaldehyde can cause genetic mutations and chromosomal damage in mammalian cell nuclei. Formaldehyde has a combined effect with other polycyclic aromatic hydrocarbons, such as benzo [a] pyrene, which increases toxicity.
Formaldehyde is a paradoxical chemical: indispensable yet dangerous, ubiquitous yet controllable. Its role in adhesives, disinfectants, and industrial processes underscores its economic value, while its carcinogenicity demands rigorous safety protocols. As research advances, alternatives like MDI resins and bioremediation offer promising pathways to reduce reliance on formaldehyde. However, global coordination in regulation and public education remains critical to mitigating its health and environmental impacts.
The future of formaldehyde hinges on balancing innovation with responsibility. By embracing green chemistry and stringent oversight, society can harness its benefits while safeguarding human health and ecological integrity.
With the power of core competitiveness, an enterprise may stand out in the fierce market competition and enhance the value of High Quality 1100 Kg IBC Drum Formaldehyde 37% 40% and services in a certain period of time. We look not just for the economic benefits of the business, but more importantly, to achieve the success of our customers and reflect more social value. With our reputation for honesty, reliability and quality, we are committed to continuing to expand overseas markets by looking for global partners. We look forward to working with you in the near future.




