Our N tert Butoxycarbonyl 4 piperidone, 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID, iptg powder has been well-received by our customers due to its high quality and competitive prices. We continue to improve our industrial policies, have our own innovative brand and high independent innovation, independent intellectual property research capabilities, constantly enrich the quality of our products and constantly improve our skills and core competitiveness. We insist on improving customer satisfaction and loyalty, to obtain long-term customers, and on this basis continue to build our service brand, so that our long-term healthy development.
Procaine hydrochloride CAS 51-05-8 can be used as a raw material, chemical material in the laboratory. it is a relatively stable salt, usually a white crystalline powder or colorless transparent crystal. Its color is usually influenced by chemical purity and production processes. The solubility in water is quite high. It can also be dissolved or dispersed in some organic solvents, such as ethanol, ether, chloroform, and benzene. The melting point of is 154-158 ℃. This melting point can be used as one of the standards for detecting its quality. It is a relatively stable anesthetic. It can be stably stored at room temperature. It can also resist oxidation in the air and is not affected by light.

|
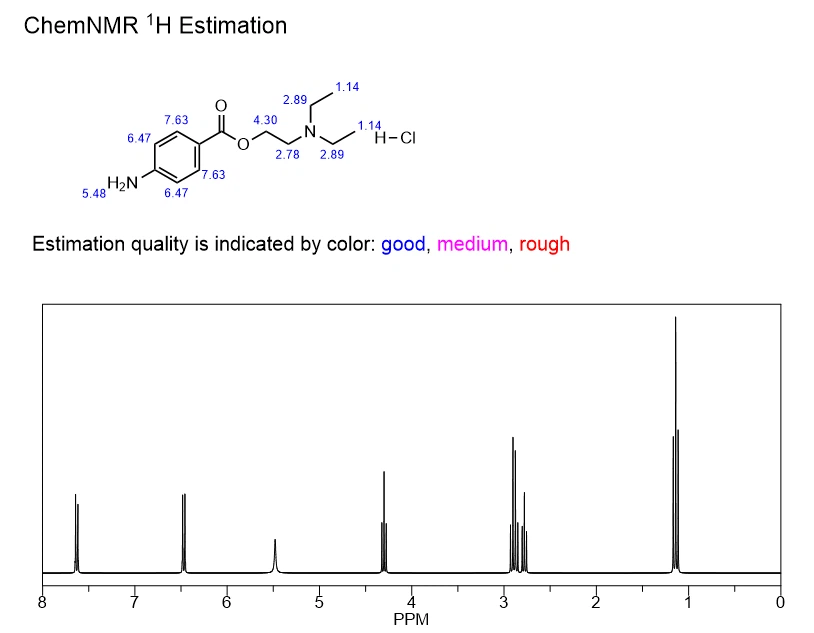
Chemical Formula |
C13H21ClN2O2 |
|
Exact Mass |
272.13 |
|
Molecular Weight |
272.77 |
|
m/z |
272.13 (100.0%), 274.13 (32.0%), 273.13 (14.1%), 275.13 (4.5%) |
|
Elemental Analysis |
C, 57.24; H, 7.76; Cl, 13.00; N, 10.27; O, 11.73 |
|
|
|
|
|

It is a local anesthetic and is also used as an over-the-counter medication to relieve mild pain, such as toothache. It is widely used in the medical industry because it is a safe and effective local anesthetic. Here we will discuss several methods for synthesizing it.
1. Reactions between Para aminobenzoic acid (PABA) and diethylamine ether
The earliest preparation method of product was to react PABA and diethylamine to form procaine under alkaline conditions. This method was discovered by Ernest Fourneau and Pierre Refrain in 1905.
Firstly, PABA is acidified with concentrated sulfuric acid to form the corresponding amide, and then it reacts with ethylenediamine under the action of sodium hydroxide to form a procaine base. Finally, the procaine base is neutralized with hydrochloric acid solution to obtain product.
2. Reaction between Procainamide and PABA
Procainamide is an advanced psychotropic drug obtained by sulfonation reaction of procaine. Therefore, procaine reacts with p-Toluenesulfonic acid and H2SO4 to produce procaine mide methylated at position 14, and reacts with PABA to produce HCL salt.
|
|
|
|

Pure API(Active pharmaceutical ingredient) for science researching only, Standard substance for analysis, Pharmacokinetic study, receptor resistance test etc. Procaine hydrochloride, white crystalline or crystalline powder is local anesthetic with strong effect but less toxicity and short time-effective. Clinical practice mainly uses procaine hcl, procaine hcl powder for infiltration anesthesia and conduction anesthesia. Because of its poor penetration, it is generally not used for surface anesthesia.
Chemical property:
(a) Stability:
It can be stably stored at room temperature and pressure. However, it will change color, decompose, and lose its characteristics in sunlight and air.
(b) Solubility:
It is easily soluble in water and has extremely low volatility. It can also be soluble in some organic solvents such as methanol and ethanol. When the solubility is 25 ℃, there is 8.8g of product in every 100ml of water.
(c) PH value:
The aqueous solution ofit is weakly acidic, with a pH value of approximately 3.0-5.0.
(d) Oxidation-reduction properties:
It appears blue in the presence of cations such as Fe3+and Cu2+. It can also be oxidized by reagents such as HI, HNO3, FeCl3 trachoma reagent, Molisch reagent, etc., and heated under alkaline conditions to form a purple substance.
(e) Dilution:
The dilution of product solution gradually increases, but it cannot decompose.
(f) Alkalinity:
It is weakly acidic and can decompose into hydrogen ions and stable hydrochloric acid molecules with equal moles.
(g) Instability:
It has strict requirements for stability. It must be stored in a sealed, moisture-proof, and light proof environment, and avoid contact with alkaline solvents, metal ions, and oxidants.

inspect
Acidity:
Take 0.40g of this product, dissolve it in 10mL of water, and add 1 drop of methyl red indicator solution. If it turns red, add 0.20mL of sodium hydroxide titrant (0.02mol/L), and the strain will turn orange.
Clarity of solution:
Take 2.0g of this product and dissolve it in 10mL of water. The solution should be clear.
Aminobenzoic acid:
determined by high-performance liquid chromatography (General Rule 0512).
Test solution:
Take this product, weigh it accurately, dissolve it in water, and dilute it quantitatively to prepare a solution containing 0.2mg per 1mL.
Reference solution:
Take an appropriate amount of para aminobenzoic acid reference substance, accurately weigh it, dissolve it in water and dilute it quantitatively to prepare a solution containing approximately 1 µ g per 1mL.
System suitability solution:
Take 1mL of the test solution and 9mL of the reference solution, mix well.
Measurement method:
Precisely measure the test solution and control solution, inject them into the liquid chromatograph separately, and record the chromatogram.
Dry weight loss:
Take this product and dry it at 105 ° C to a constant weight, with a weight loss of no more than 0.5% (General Rule 0831).
Burning residue:
Take 1.0g of this product and check according to the law (General Rule 0841). The residual residue should not exceed 0.1%.
Limit:
If there are chromatographic peaks in the chromatogram of the test solution that have the same retention time as the peak of para aminobenzoic acid, the peak area should not exceed 0.5%, calculated according to the external standard method.
24H Online Service
Convallis posuere morbi urna molestie elementum pulvinar odio etiam.
Global Shipping
Urna condimentum mattis pellentesque nibh. Integer vitae justo eget magna
Chromatographic conditions:
Octadecylsilane bonded silica gel was used as the filler, and a 0.05mol/L potassium dihydrogen phosphate solution containing 0.1% sodium heptanesulfonate (adjusted to pH 3.0 with phosphoric acid) - methanol (68:32) was used as the mobile phase. The detection wavelength was 279nm, and the injection volume was 10 µ L.
System suitability requirements:
In the chromatogram of the system suitability solution, the theoretical plate number calculated based on the peak of para aminobenzoic acid should not be less than 2000, and the separation degree between the procaine peak and the para aminobenzoic acid peak should be greater than 2.0.
Iron salt:
Take the residue left under the ignition residue, add 2mL of hydrochloric acid, evaporate it on a water bath, add 4mL of dilute hydrochloric acid, dissolve it at a low temperature, add 30mL of water and 50mg of ammonium persulfate, and check according to the general rule 0807. Compared with the control solution made of 1.0mL of standard iron solution, it should not be deeper (0.001%).

Procaine hydrochloride belongs to ester local anesthetics, which have local anesthetic, anti-inflammatory, anti rheumatic, vasodilator, anti arrhythmic and other effects. It is mainly used in clinical practice for infiltration anesthesia, block anesthesia, lumbar spine anesthesia, etc.
Belonging to ester local anesthetics, it has a fast onset and good anesthetic effect, and has a blocking effect on nerve tissues in various parts. Therefore, it is commonly used in clinical practice for local infiltration anesthesia, conduction anesthesia, spinal anesthesia, etc.
Belonging to ester local anesthetics, it has anti-inflammatory effects and can block inflammatory reactions. It is commonly used clinically to relieve symptoms such as toothache, headache, neuralgia, and joint pain.
It also has anti rheumatic effects, which can inhibit inflammatory factors in the patient's body, thereby alleviating symptoms such as joint pain and swelling caused by rheumatoid arthritis.
It can directly act on vascular smooth muscle, dilate blood vessels, increase blood flow, improve microcirculation, and is commonly used in clinical practice to treat dizziness and headache symptoms caused by ischemic cerebrovascular disease, peripheral vascular disease, etc.
It also has an anti arrhythmic effect, which can inhibit the central nervous system and alleviate symptoms such as palpitations, chest tightness, and dizziness caused by arrhythmia.
It should be noted that the above drugs should be used under the guidance of a doctor and should not be used blindly to avoid adverse reactions and damage to the body. In addition, it should be contraindicated for patients with local anesthetic allergies, liver and kidney dysfunction, etc.

Procaine hydrochloride, as a classic local anesthetic, has a complex and precise mechanism of action involving multiple physiological systems and molecular interactions.
As an ester local anesthetic, its local anesthetic effect is mainly achieved by blocking the conduction of nerve fibers. The specific mechanism is as follows:
(1) Neuronal membrane penetration: Procaine molecules rely on concentration gradients to diffuse through the neuronal membrane, which is the first step in their anesthetic effect.
(2) Sodium ion channel blockade: After entering nerve cells, procaine selectively binds to sodium ion channels on the cell membrane, preventing sodium ion influx. Sodium ions are key ions for the generation and propagation of action potentials in nerve cells, and their internal flow is obstructed, leading to an increase in the excitation threshold of nerve cells and a loss of excitability and conductivity.
(3) Information transmission blockade: As the generation and propagation of nerve impulses rely on the transmembrane flow of sodium ions, procaine effectively blocks the transmission of nerve impulses by blocking sodium ion channels, achieving local anesthesia effects.

Central nervous system impact

The impact on the central nervous system is closely related to the dosage of medication, specifically manifested as:
(1) Inhibition effect: At conventional doses, procaine mainly exerts inhibitory effects on the central nervous system. Patients may exhibit sedation, elevated pain threshold, and even coma or coma. This inhibitory effect helps alleviate patients' anxiety and pain, and improve the comfort of surgery or treatment.
(2) Excitatory effect: With the increase of medication dosage, the inhibitory effect of procaine on the central nervous system may gradually weaken, and even transform into an excitatory effect. Patients may experience states of excitement, mania, and in severe cases, may lead to adverse reactions such as seizures. Therefore, when using procaine, the dosage must be strictly controlled to avoid excessive use.
The impact on the cardiovascular system is also closely related to the dosage of medication, specifically manifested as:
(1) Changes in heart rate and blood pressure: may stimulate the sympathetic nervous system, causing an increase in heart rate and blood pressure. However, as the dosage of medication increases, it may inhibit peripheral vascular dilation, leading to a decrease in blood pressure. This complex dose-response relationship requires doctors to closely monitor patients' heart rate and blood pressure changes when using procaine.
(2) Antiarrhythmic effect: It also has an antiarrhythmic effect. It can reduce the occurrence of arrhythmia by blocking the transmission of nerve impulses and inhibiting the excitability of myocardial cells. However, this function needs to be used reasonably under the guidance of a doctor to avoid the occurrence of adverse reactions.

Neuromuscular blockade effect

Competitive inhibition of acetylcholine receptors on the motor endplate membrane results in ineffective transmission of nerve impulses to muscle cells, leading to neuromuscular blockade. This effect helps to reduce muscle tension and pain during surgery, and improve the smoothness of surgical procedures. However, it should be noted that the neuromuscular blocking effect of procaine may lead to adverse reactions such as muscle weakness or paralysis in patients, so it is necessary to closely monitor changes in muscle function during use.
adverse reaction
Procaine Hydrochloride, as a classic ester local anesthetic, has been widely used in infiltration anesthesia, block anesthesia, and epidural anesthesia since its clinical application in the early 20th century due to its rapid onset and moderate duration of action. However, adverse reactions that may occur during its use involve multiple organ systems such as the cardiovascular system, central nervous system, respiratory system, gastrointestinal tract, and allergic reactions.
Cardiovascular system adverse reactions
Clinical manifestations
Arrhythmia: Procaine hydrochloride can cause sinus tachycardia, atrial premature beats, ventricular premature beats, and even ventricular tachycardia. Clinical data shows that the incidence of arrhythmia in epidural anesthesia is about 2% -5%, which is more common when the administration speed is too fast or the dosage is too high.
Blood pressure fluctuations: Some patients may experience transient hypotension or hypertension. Hypotension is usually caused by vasodilation or myocardial inhibition, while hypertension may be associated with sympathetic nervous system excitation or pain stimulation.
Facial flushing: About 10% -15% of patients experience facial flushing after injection, which is related to the drug directly stimulating blood vessels or causing histamine release.
Mechanism of occurrence
Sodium channel blockade: Procaine hydrochloride inhibits sodium channels in myocardial cells, reducing the rate of action potential phase 0 rise and slowing down conduction, which may induce reentrant arrhythmia.
Calcium channel effects: High concentrations of drugs may inhibit L-type calcium channels, reduce calcium influx, lead to decreased myocardial contractility, and cause hypotension.
Histamine release: Ester local anesthetics can easily induce histamine release, causing vasodilation and increased capillary permeability, leading to facial flushing and hypotension.
Preventive measures
Control the administration speed: during infiltration anesthesia, the 0.25% -0.5% solution should not exceed 1.5g per hour; during block anesthesia, the 1% -2% solution should not exceed 1.0g per hour.
Monitoring vital signs: Continuously monitor electrocardiogram and blood pressure during surgery to promptly detect and treat arrhythmia.
Avoid entering blood vessels by mistake: Pull back before injection to ensure that the needle is not in the blood vessel and reduce systemic absorption.
Central nervous system adverse reactions
Clinical manifestations
Central excitation: Early symptoms include anxiety, tinnitus, dizziness, and in severe cases, muscle tremors, seizures, and even epileptic seizures may occur. The incidence rate is about 0.1% -0.5%, which is more common when the blood drug concentration exceeds 5 μ g/ml.
Central inhibition: A small number of patients may experience drowsiness and coma, which is related to the direct inhibition of the central nervous system by drugs.
Neurotoxicity: Long term or excessive use may lead to peripheral neuropathy, manifested as sensory abnormalities or decreased muscle strength.
Mechanism of occurrence
Blood brain barrier penetration: High concentration drugs can penetrate the blood brain barrier, inhibit inhibitory neurons in the central nervous system, and lead to increased excitability.
Gamma aminobutyric acid (GABA) receptor inhibition: Procaine hydrochloride may inhibit GABA receptor function, reduce chloride ion influx, and decrease neuronal inhibition.
Accumulation of metabolites: Metabolites such as para aminobenzoic acid (PABA) may have neurotoxicity, especially when accumulated in cases of liver dysfunction.
Preventive measures
Dose control: The maximum dose should not exceed 1.0g, calculated based on body weight, and the local injection should not exceed 20mg per kg.
Avoiding hunger: Patients with malnutrition or hunger are more prone to toxic reactions and should replenish energy in advance.
Prepare emergency equipment: benzodiazepines (such as diazepam) and sodium thiopental are available during the operation to control epileptic seizures.
We start from the needs of our customers and the public to determine the direction of enterprise development, develop first-class Reagent Grade Guaranteed 99% Purity CAS 51-05-8 Procaine Hydrochloride and provide first-class services. The company adheres to the road of professional development and has a team specializing in product research and development. The company will devote itself to following the laws of market development and establishing a modern corporate governance structure.