Our company has developed a series of N N Dimethylformamide pure, Indicator Reagent, Diisopropylammonium with independent intellectual property rights with international advanced technology and has realized industrialization. In the context of globalization, in the face of fierce market competition, it is necessary to improve the quality of employees. We will pursue opportunities, increase investment in science and technology, and further strengthen trade and cooperation with well-known companies at home and abroad. Over the years, our company has trained a number of professional and technical personnel, enriched various production sites and expanded the technical strength of the company.
GS-441524 remdesivir is a small molecule, known as a competitive nucleoside triphosphate inhibitor, showing strong antiviral activity against many RNA viruses. It can inhibit the replication of several different types of RNA viruses. GS-441524 is a small molecule, called a competitive nucleoside triphosphate inhibitor, which shows strong antiviral activity against many RNA viruses. It can inhibit the replication of several different types of RNA viruses. It is a small molecule nucleoside analogue that undergoes intracellular phosphorylation under the action of cell kinases in the body, generating an active triphosphate metabolite (NTP). NTP can compete with natural nucleotides in the body, inhibit RNA polymerase, interfere with RNA replication of feline infectious peritonitis virus, and thus block the synthesis of feline infectious peritonitis virus. The active form of GS441524, GS441524 triphosphate metabolite (NTP), is an important substance in intracellular signaling, and its structural analogues compete with natural triphosphate nucleosides to participate in the RNA transcription process. GS441524 is phosphorylated into nucleoside monophosphate through cellular kinase and then transformed into an active triphosphate metabolite (NTP). This active form can act as a competitor of natural triphosphate nucleosides in viral RNA synthesis, competing with natural nucleosides to participate in RNA transcription.
Client Feedback
 |
 |
 |
About Shipment

Successfully delivery all over the world:

|
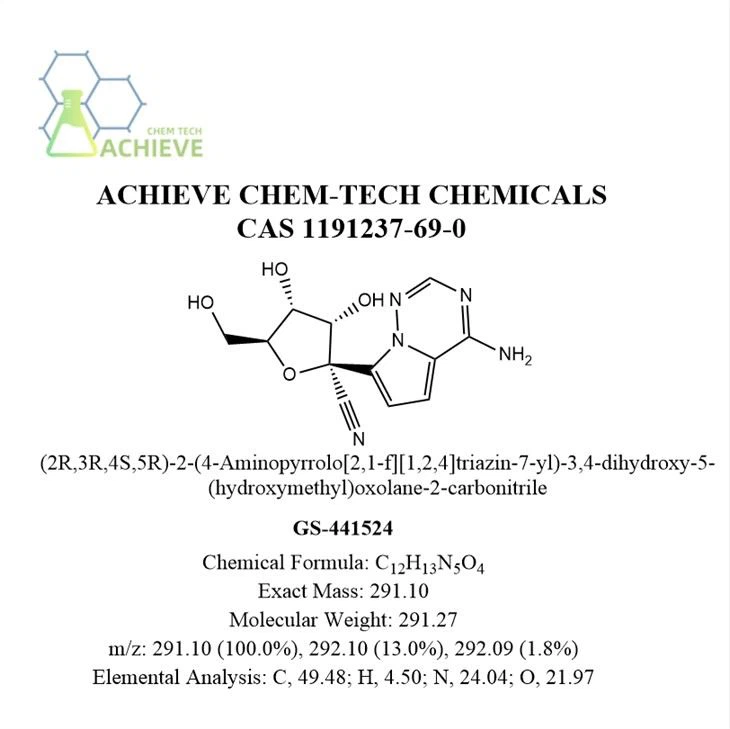
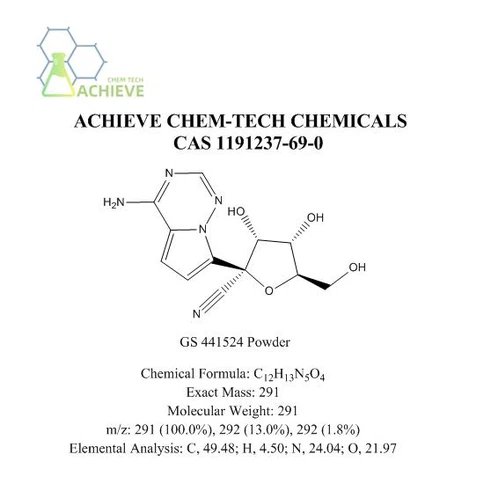
Chemical Formula |
C12H13N5O4 |
|
Exact Mass |
291.10 |
|
Molecular Weight |
291.27 |
|
m/z |
291.10 (100.0%), 292.10 (13.0%), 292.09 (1.8%) |
|
Elemental Analysis |
C, 49.48; H, 4.50; N, 24.04; O, 21.97 |
|
|
|
|
Please refer to our enterprise standard or COA. If you want to get more details, welcome to contact our sales.
Additional information of chemical compound: Density 1.84 + / - 0.1 g/cm3 (Predicted), Acidity coefficient (pKa)12.13±0.70(Predicted)

The method of synthesizing GS441524 remdesivir in the laboratory requires a series of chemical reaction steps to construct the core structure of the compound. The following is a specific description of the laboratory synthesis method for GS441524:
Starting materials: The starting materials for synthesizing GS-441524 are usually basic sugar based donors, nitrogen based donors, and phosphorus based donors. These donors are commonly used starting materials in chemical reactions and can be easily obtained under laboratory conditions.
Step 1:
Protective groups: Before proceeding with the next reaction, certain functional groups need to be protected to avoid interference with subsequent reactions. These protective groups can be removed under specific conditions, thereby restoring the activity of these functional groups.
Step 2:
Phosphorylation reaction: The core reaction for synthesizing GS441524 is phosphorylation reaction. This reaction requires the use of a phosphate donor and the attachment of phosphate groups to nitrogen groups under the action of a catalyst. This reaction is reversible and requires the use of special conditions to promote its progress.
Step 3:
Remove protective groups: After completing the phosphorylation reaction, the previously added protective groups need to be removed. This process requires the use of specific protective reagents to avoid affecting other functional groups.
Step 4:
Modification and purification: After completing the main structural synthesis, some modification reactions are required to obtain the final GS441524 molecule. These modification reactions can alter certain properties of molecules, such as increasing solubility or removing impurities. Finally, purification steps are required to obtain high-purity GS441524.
Characterization and analysis: After synthesizing GS441524, it is necessary to characterize and analyze it to confirm its chemical structure and purity. This can be achieved through analytical methods such as nuclear magnetic resonance, mass spectrometry, and high-performance liquid chromatography.
|
|
|
Remark: BLOOM TECH(Since 2008), ACHIEVE CHEM-TECH is the subsidiary of us.

It can inhibit the replication of several different types of RNA viruses, such as zoonotic severe acute respiratory syndrome (SARS) coronavirus, Middle East respiratory syndrome virus, Ebola virus, Lassa fever virus, Junin virus and respiratory syncytial virus, while showing low cytotoxicity in a wide range of cell lines. GS441524 for cats is the most widespread usage.
GS441524 is a small molecule nucleoside analogue with antiviral activity, mainly used for the treatment of infectious peritonitis in cats. The following is a detailed analysis of the antiviral activity of GS441524:
Antiviral spectrum:
GS-441524 has broad-spectrum antiviral activity and can inhibit the replication of various RNA viruses, including feline infectious peritonitis virus, feline coronavirus, canine parvovirus, porcine reproductive and respiratory syndrome virus, etc. These viruses are the main pathogens of common diseases in pets and animals, and the antiviral activity of GS-441524 provides the possibility of treating these diseases.
Mechanism of action:
The mechanism of GS-441524's antiviral activity is mainly interference with the transcription process of viral RNA. GS-441524 can compete with natural nucleotides for RNA polymerase, thereby blocking the synthesis of viral RNA. In addition, GS-441524 can also induce cell apoptosis, thereby accelerating virus clearance.
Efficacy:
In clinical trials, the therapeutic effect of GS-441524 has been verified. For feline infectious peritonitis, GS-441524 can effectively alleviate symptoms and improve the survival rate of sick cats. At the same time, GS-441524 can also effectively prevent and treat other animal viral diseases, such as canine parvovirus.
4. Safety:
The safety of using GS-441524 in cats has been widely studied and clinically validated. According to existing data and literature reports, the vast majority of patients do not experience any abnormalities in blood indicators during use. However, there may be some possible side effects when using GS-441524, including pain at the injection site. It should be noted that GS-441524 may affect the development of permanent teeth in young cats without tooth replacement.

GS-441524, as a promising antiviral drug, its chemical reactivity constitutes the core of the drug's mechanism of action, and has had a profound impact on drug design, optimization, storage, and application. A deep understanding of chemical reactivity not only helps to reveal the secrets of its antiviral activity, but also provides important basis for quality control, stability improvement, and clinical application strategy formulation in drug development.
Phosphorylation reaction: activation and targeting: The primary chemical reaction inside cells is phosphorylation, which is catalyzed by specific cellular kinases to convert inactive substances into triphosphate metabolites (NTP) with antiviral activity. Phosphorylation not only enhances hydrophilicity, making it easier for it to penetrate cell membranes and accumulate within cells, but also increases its affinity for key enzymes or structures involved in viral replication, such as RNA polymerase and DNA polymerase, by increasing negative charges. This phosphorylation activation mechanism enables it to precisely target the key steps of virus replication, effectively inhibiting virus replication and transmission. It is worth noting that the efficiency and specificity of the phosphorylation process are influenced by various factors such as the type of cellular kinase, drug concentration, and cellular environment (such as pH and ion strength), which need to be fully considered in drug design and clinical applications.
Competition with natural nucleotides: The structural design enables it to simulate natural nucleotides and competitively bind with RNA polymerase binding sites. This mechanism significantly reduces the replication of viral RNA by interfering with its normal transcription process, thereby effectively inhibiting viral proliferation. It is worth noting that the binding of this substance to RNA polymerase may also cause conformational changes in the polymerase, further inhibiting its catalytic activity. This additional inhibitory effect enhances the antiviral effect of the drug. However, its affinity for RNA polymerase and its degree of influence on polymerase conformation are influenced by various factors, including drug concentration, polymerase type, cellular environment, etc. These factors need to be carefully evaluated in the process of drug screening and optimization.
Hydrolysis reaction: stability challenge: Under specific conditions (such as acidic environments), hydrolysis reactions are prone to occur, leading to the breakdown of drugs into their constituent amino acids or smaller molecular fragments. The hydrolysis reaction not only reduces the chemical purity, but may also affect its antiviral activity and bioavailability. Therefore, during storage, transportation, and use, it is necessary to strictly control environmental conditions (such as pH value, temperature, humidity) to reduce the occurrence of hydrolysis reactions. In addition, increasing the stability of drugs through chemical modifications such as esterification, amidation, etc. is also an effective strategy to solve hydrolysis problems.
Oxidation reaction: photosensitivity and air stability: Sensitive to light and oxygen in the air, it is prone to oxidative degradation, leading to changes in the chemical structure and loss of activity of drugs. Oxidation reactions not only reduce purity, but may also produce toxic degradation products, posing a threat to the safety and efficacy of drugs. Therefore, strict light avoidance, sealing, and antioxidant measures must be taken during production, packaging, transportation, and use to ensure the stability and safety of the drug.
Other influencing factors: In addition to the main chemical reactions mentioned above, chemical properties are also influenced by various factors such as drug concentration, solvent type, pH regulator, ionic strength, and temperature. The changes in these factors may lead to subtle changes in chemical properties, which in turn affect their antiviral activity and safety. Therefore, in the process of drug development and application, it is necessary to conduct in-depth research on these factors to optimize drug formulations and storage conditions, ensuring the effectiveness and safety of drugs.
In summary, the chemical reactivity of GS-441524 is a complex and multifaceted field, covering multiple aspects such as phosphorylation, competition with natural nucleotides, hydrolysis, and oxidation. These characteristics collectively determine the potential application and precautions in antiviral therapy. In order to fully exert its antiviral effect, it is necessary to conduct in-depth research on its chemical reactivity and influencing factors, optimize drug formulations and storage conditions, to ensure the safety and effectiveness of drugs. Meanwhile, future research should also explore strategies for combined use with other antiviral drugs to further improve antiviral efficacy and reduce the risk of drug resistance.

The discovery history of GS-441524 remdesivir can be traced back to 2012, when researchers at Gilead Sciences discovered a compound called GS-441524 while studying the replication mechanism of hepatitis C virus (HCV). This compound exhibits broad-spectrum antiviral activity against various microorganisms, pathogenic factors, or diseases, including hepatitis C, dengue fever, influenza A, SARS, norovirus, etc.

Gilead Science began to conduct in-depth research on GS-441524, and found that the compound had a very good inhibitory effect on novel coronavirus (SARS CoV) in vitro experiments. Subsequently, Gilead Science began to develop GS-441524 as a therapeutic drug against hepatitis C virus and novel coronavirus.
However, GS-441524 has not been approved as an official drug in any country worldwide. Nevertheless, Gilead Science donated a certain amount of GS-441524 to China for the treatment of novel coronavirus in an emergency.
Overall, the discovery history of GS-441524 can be attributed to the discovery and research process of a compound with broad-spectrum antiviral ability. Although the drug has not yet been approved, it has been used to treat novel coronavirus and other viral diseases under some special circumstances.
FAQ
Is remdesivir the same as GS-441524?
-
GS-441524 is the metabolite of the antiviral drug remdesivir (GS-5734) used to treat people with SARS-CoV-2 infections, and both drugs yield the same active metabolite in the host cell.
Does GS-441524 work?
+
-
The overall treatment success rate was 84.6%. This rate was higher when GS-441524 was combined with other antivirals and lower in cases of wet FIP or those with neurological complications. Combination therapy with other antivirals may improve outcomes in complicated FIP cases, although further studies are needed.
What is the new name for remdesivir?
+
-
VEKLURY is a prescription medicine used to treat COVID-19 in adults and children weighing at least 3 pounds who are hospitalized; or not hospitalized and have mild-to-moderate COVID-19, and are at high risk for progression to severe COVID-19, including hospitalization or death.
What does GS-441524 stand for?
+
-
GS-441524 is a nucleoside analogue antiviral drug which was developed by Gilead Sciences. It is the main plasma metabolite of the antiviral prodrug remdesivir, and has a half-life of around 24 hours in human patients.
What is the new treatment for cats with FIP?
+
-
A compounded oral remdesivir preparation (capsule form) has been used successfully to treat cats with FIP when GS-441524 was not available, for example in New Zealand (Renner et al., 2025). Molnupiravir (EIDD-2801) is another nucleoside analogue that inhibits viral replication and is metabolised into EIDD-1931 (NHC).
We recognize that our Remdesivir Metabolite 1191237-69-0 GS-441524 for Cat need to be improved in order to remain competitive in the marketplace. Through the development of management innovation, we strengthen the cultivation of the core values of the company and guide the changes in the thinking and behavior of employees. As we have customer centric approach, we have made frequent innovations in our products that further provide maximum satisfaction to our clients. We also offer customized products to meet the specific demands of our clients to achieve customer satisfaction.