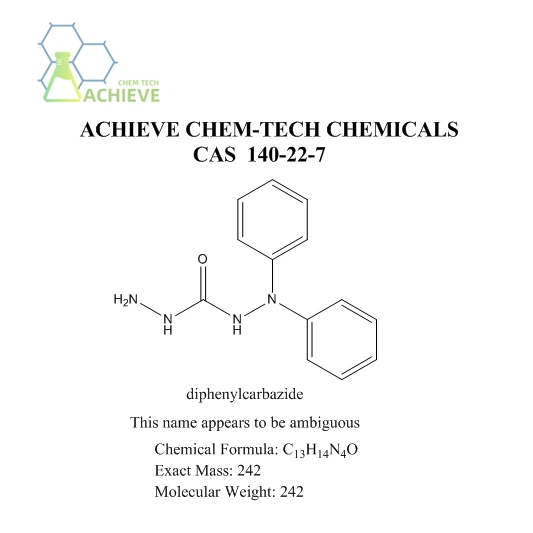
Diphenylcarbazide reagent, white crystalline powder, molecular formula C13H14N4O, CAS 140-22-7, Can be used as an indicator for titrating iron. It is usually used in chemical laboratories and belongs to hazardous chemicals. White crystalline powder. Gradually turning red in the air. Slightly soluble in water, soluble in ethanol and acetone, insoluble in ether. Reacts with Cr2O to turn red. It also undergoes color reactions with metal ions such as Ag+, Ni 2+, Pb 2+, Cu 2+, Fe 3+, Hg+, and Hg 2+. Used as an indicator for titrating iron. Colorimetric determination of chromium, mercury, and lead, detection of cadmium, mercury, magnesium, aldehydes, and soil alkaloids. Determine the chloride reduction indicator for dichromate. Mercury content method for determining adsorption indicators of chloride and cyanide. (Used as a chromogenic reagent for spectrophotometric determination of chromium and for detecting Cu2+, Fe3+, Hg2+, etc. It is also used as an indicator for oxidation-reduction, adsorption, and complexometric titration. Since 2008, we have been focusing on organic synthesis technology, and our ability to develop high-quality products relies entirely on our skilled R&D team.).

|
Chemical Formula |
C13H14N4O |
|
Exact Mass |
242.12 |
|
Molecular Weight |
242.28 |
|
m/z |
242.12 (100.0%), 243.12 (14.1%), 243.11 (1.5%) |
|
Elemental Analysis |
C, 64.45; H, 5.82; N, 23.13; O, 6.60 |
|
Morphological |
Powder or Flakes |
|
Color |
White to cream |
|
Melting point |
170 – 175 °C ( lit. ) |
|
Boiling point |
385.1 °C ( rough estimate ) |
|
Density |
1.31 g / cm3 |
|
Storage conditions |
2-8 ° C |
|
Acidity coefficient ( pKa ) |
9.98 ± 0.43 ( Predicted ) |
|
|
|

Synthesis of diphenylaminourea: it is obtained by reacting phenylhydrazine with urea. Phenylhydrazine and urea were added to xylene, refluxed for 32 hours, placed overnight, and the crude product was filtered out. Then dissolve it in a mixed acid solvent of ethanol and a little acetic acid, cool it quickly and crystallize it. After filtration, soak it with ethanol again, and then dry it.

Diphenylcarbazide reagent is an organic compound with the chemical formula C13H12N2O. It is a white crystalline powder with a melting point of 180-184 ℃ and a relative density of 1.23. This compound has weak alkalinity and is soluble in hot water and alcohols, but insoluble in ethers. It is mainly used as a reagent for determining alkaloids, phenols, and amino acids, and is also used as a detoxifying agent and antihistamine.
One of the methods for preparing 1,5-Diphenylcarbazide is by reacting phenylhydrazine with urea. The following are the detailed steps for preparation:
1. Mix phenylhydrazine and urea in a 1:1 molar ratio, and add an appropriate amount of sodium hydroxide solution to completely dissolve it.
2. Heat the mixture to reflux temperature (about 80 ℃) and keep stirring at this temperature for several hours to allow the reaction to proceed fully.
3. After reaching the endpoint of the reaction, cool the reaction solution to room temperature and adjust the pH value to neutral with an appropriate amount of dilute acid solution.
4. Pour the solution into an appropriate amount of ethanol to form a precipitate. Collect the white solid obtained through filtration and washing.
5. Purify the collected solids, such as recrystallization or column chromatography, to obtain high-purity 1,5-Diphenylcarbazide.
The reaction between phenylhydrazine and urea can be expressed as the following chemical equation:
C6H6NHNH2 + H2N-CO-NH2 → C6H6N2H2O + NH3
In the reaction, phenylhydrazine and urea undergo dehydration condensation reaction to generate 1,5-Diphenylcarbazide and water. This reaction is a typical condensation reaction, in which phenylhydrazine and urea are used as reactants, and sodium hydroxide is used as a catalyst to conduct the reaction in a weakly alkaline environment. High purity 1,5-Diphenylcarbazide can be obtained by heating reflux and adjusting pH value.

Diphenylcarbazide reagent, also known as 1,5-Diphenylcarbazide in Chinese, is an organic compound with multiple uses.

1. Reagent for measuring alkaloids, phenols, and amino acids:
1,5-Diphenylcarbazide can be used to determine the content of alkaloids, phenols, and amino acids. This is because the compound has a specific color change under acid-base conditions and can be used to indicate the endpoint of the titration reaction. By reacting with these compounds, the color change of 1,5-Diphenylcarbazide can provide concentration information about the target compound.
2. Antidotes:
In some cases, 1,5-Diphenylcarbazide can be used for detoxification. Specifically, it can be used to alleviate toxic symptoms caused by excessive intake of alkaloids, phenols, or amino acids. By combining with its complex formed in the body, 1,5-Diphenylcarbazide can help expel these toxic substances from the body, thereby alleviating symptoms of poisoning.


3. Anti histamine drugs:
1,5-Diphenylcarbazide has anti histamine effects and can be used to treat allergic reactions. In allergic reactions, histamine is a chemical substance that can cause allergic symptoms. By binding with histamine, 1,5-Diphenylcarbazide can inhibit the action of histamine, thereby alleviating allergic symptoms.
4. Synthesis of other compounds:
1,5-Diphenylcarbazide can be used as an intermediate in the synthesis of other compounds. For example, it can be used to synthesize some drugs, pesticides, dyes, etc. Some of these compounds may have more specific biological activity or chemical reactivity properties.


5. A sensitive indicator commonly used in titration analysis.
In the process of titrating iron elements, it can serve as an indicator for the complexation reaction between iron ions and EDTA. Under the condition of pH=2-3, 1,5-Diphenylcarbazide reacts with iron ions to form a purple red complex. When all iron ions are complexed, the solution turns red, and the titration endpoint can be determined. The chemical equation for the complexation reaction is: Fe++1,5-DPC → Fe (1,5-DPC)+H+. Put the sample solution into a conical flask and add an appropriate amount of buffer to adjust the pH value to 4-6.
Then add an appropriate amount of EDTA standard solution and shake well. Add an appropriate amount of indicator solution dropwise and gently shake the conical flask to mix evenly. Stop adding indicator solution when the solution turns purple red. At this point, it is possible to determine whether the titration endpoint has been reached by observing the color change. If the color change is significant and stable, it indicates that the titration endpoint has been reached.

Diphenylcarbazide reagent, chemical formula C13H14N4O, molecular weight 242.28, CAS number 140-22-7. It is a white or beige crystalline powder with a density of approximately 1.292g/cm ³ and a boiling point range of 380.4 to 436.4 ℃. This substance is sensitive to light, so it should be sealed and stored away from light. In terms of chemical properties, diphenylcarbazide has multiple uses, especially in titration analysis. It serves as an indicator that can display the endpoint of a chemical reaction, thereby helping chemists to accurately perform quantitative analysis.

1. Indicator for titrating iron element
One of the most commonly used uses of diphenylcarbazide is as an indicator for titrating iron elements. In chemical analysis, iron is an important analytical object, and its content often needs to be determined by titration. Diphenylhydrazide can undergo specific chemical reactions with iron ions (especially Fe ³ ⁺) to produce a colored compound. The color change of this compound can serve as an endpoint indicator for titration reactions, helping chemists accurately determine the content of iron element.
3. Determination of redox indicators for dichromate
In redox titration, diphenylcarbazide also plays an important role. For example, it can serve as an oxidation-reduction indicator for determining dichromate. Dichromate is a chemical substance with strong oxidizing properties, often used as an oxidant in redox reactions. By adding an appropriate amount of reducing agent (such as ferrous ions) to a solution containing dichromate, a reduction reaction can occur. In this process, diphenylcarbazide can display the reaction endpoint, helping chemists accurately determine the amount of reducing agent and calculate the content of dichromate.


4. Mercury content method for determining adsorption indicators of chloride and cyanide
Mercurimetry is a commonly used titration analysis method that uses the chemical reaction between mercury ions and the substance being measured to determine its content. In the mercury quantification method, diphenylcarbazide can be used as an adsorption indicator. For example, when measuring chlorides and cyanides, an appropriate amount of mercury ions can be added to a solution containing the substance to be tested, causing it to undergo a chemical reaction. As the reaction progresses, the color in the solution will change. When diphenylcarbazide is adsorbed onto the generated precipitate, it exhibits a specific color change, indicating the endpoint of the reaction. This method has the advantages of high sensitivity and good accuracy, and has therefore been widely promoted in practical applications.
5. Chromogenic reagents for the determination of chromium by spectrophotometry
Spectrophotometry is a quantitative analysis method based on the absorption or emission characteristics of substances towards light. In spectrophotometry, diphenylcarbazide can be used as a chromogenic reagent for the determination of chromium. When chromium ions react with diphenylcarbazide, a colored compound is generated. The color depth of this compound is directly proportional to the concentration of chromium ions. Therefore, by measuring the absorbance or emission spectral intensity of the compound, the content of chromium ions can be calculated. This method has the advantages of easy operation, high sensitivity, and good accuracy, and has therefore been widely used in fields such as environmental monitoring and food analysis.


6. Color reagents for detecting Cu ² ⁺, Fe ³ ⁺, Hg ² ⁺, etc
In addition to chromium ions, diphenylcarbazide can also be used as a chromogenic reagent for detecting other metal ions. For example, it can be used to detect metal ions such as Cu ² ⁺, Fe ³ ⁺, Hg ² ⁺, etc. These metal ions play important roles in both nature and human activities, therefore accurate determination of their content is of great significance. By utilizing the color change generated by the reaction between diphenylcarbazide and these metal ions, colorimetric or spectrophotometric analysis can be performed to determine the content of metal ions. This method has the advantages of high sensitivity and good selectivity, and has therefore been widely promoted in practical applications.
7. Indicators for oxidation-reduction, adsorption, and complexometric titration
In addition to the above-mentioned uses, diphenylcarbazide can also be used as an indicator for oxidation-reduction, adsorption, and complexometric titration. In redox titration, it can indicate the amount of oxidant or reducing agent used; In adsorption titration, it can indicate the endpoint of the adsorption reaction; In titration, it can display the formation of the complex. These applications fully demonstrate the wide applicability of diphenylcarbazide in the field of chemical analysis.

Hot Tags: diphenylcarbazide reagent cas 140-22-7, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 4 5 Chloro 2 pyridylazo 1 3 phenylenediamine, Neutral red powder, p Dimethylaminobenzaldehyde, Laboratory Reagent, dithizone test, Sodium Diethyldithiocarbamate