Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of edta disodium salt solution cas 139-33-3 in China. Welcome to wholesale bulk high quality edta disodium salt solution cas 139-33-3 for sale here from our factory. Good service and reasonable price are available.
EDTA disodium salt solution, mainly composed of disodium EDTA, also known as EDTA-2na, molecular formula C10H14N2Na2O8, CAS 139-33-3, is an odorless or slightly salty white or milky white crystalline or granular powder, odorless and tasteless. It is soluble in water but very difficult to dissolve in ethanol. It is an essential chelating agent, which can chelate metal ions in solution. It can prevent the discoloration, deterioration, turbidity caused by metals, and the oxidation loss of vitamin C and improve the oxidation resistance of oil (trace metals in fat such as iron and copper can promote oil oxidation). It is a good coordination agent in chemistry. It has six coordination atoms, and the formed complex is called a chelate. EDTA is often used in coordination titration, which is generally used to determine the content of metal ions. EDTA has essential applications in dye, food, medicine, and other industries. It shall be stored in a cool and dry warehouse, not in aluminum containers, and shall not be stored and transported together with strong acid, strong alkali, oxidant, and flammable chemicals.

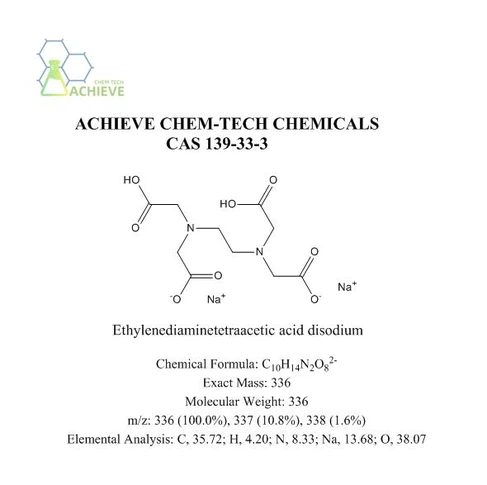
|
Chemical Formula |
C10H14N2O82- |
|
Exact Mass |
336 |
|
Molecular Weight |
336 |
|
m/z |
336 (100.0%), 337 (10.8%), 338 (1.6%) |
|
Elemental Analysis |
C, 35.72; H, 4.20; N, 8.33; Na, 13.68; O, 38.07 |
|
|
|
The characteristics of EDTA dispersion salt are as follows: 1 EDTA has a wide range of coordination properties and can form stable chelates with almost all metal ions. 2. the coordination ratio between EDTA and m-EDTA is mostly 1:1. 3. most chelates are charged so that they can be dissolved in water and react quickly. They are mainly used as complexing agents. 4. dissociation equilibrium: in an aqueous solution, two carboxyl h+ groups are transferred to amino N to form bipolar ions. H4y usually represents EDTA. Due to its negligible solubility in water and acid, the commonly used disodium salt is na2h2y · 2H2O, abbreviated as EDTA-2na. When the acidity of the solution is very high, the two carboxyl groups and two amino groups of data-2na can accept h+ again to form h6y2+, which is equivalent to a hexabasic acid and has a six order dissociation constant:
Ka1=100.9; Ka2=101.6 ; Ka3=102.1; Ka4=102.8; Ka5=106.2; Ka6=1010.3 .
Seven forms:
h6y2+, h5y+, h4y, h3y-, h2y2-, hy3-, Y4-
When ph<1, it mainly exists in the form of h6y2+;
When ph>11, it mainly exists as a y4- coordination ion.

We are the supplier of Edta Disodium Salt Solution.
Remark: BLOOM TECH(Since 2008), ACHIEVE CHEM-TECH is our subsidiary.
Based on the provided information, the chemical synthesis method of EDTA disodium mainly includes the following steps:
Reaction between sodium hydroxide and EDTA:
NaOH + C10H16N2O8 → Na-C10H16N2O8 + H2O
PH adjustment of reaction solution:
H2O + CO2 + Na-C10H16N2O8 → NaHCO3 + H-C10H16N2O8
NaHCO3 + NaOH → Na2CO3 + H2O
Na2CO3 + Ca(OH)2 → CaCO3↓ + 2NaOH
Generation of disodium EDTA:
Na-C10H16N2O8 + NaOH → C10H19N2NaO9+H2O

Prepare reaction equipment and reagents: Prepare reactors, stirrers, thermometers, pH meters, and reagents such as sodium hydroxide and EDTA to ensure that the equipment is clean and the quality of the reagents is qualified.
Feeding: Add a certain amount of deionized water to the membrane chemical reactor, and then add liquid sodium hydroxide and EDTA in batches in a 2:4 mass ratio. After each batch is added, stir evenly with a mixer. Repeat this step until the pH of the reaction solution reaches 5.
Heating and insulation: Heat the reaction solution at 95 ℃ and maintain this temperature. Then alternate the feeding and insulation processes until the reaction is complete. Specifically, heat the reaction solution to boiling state at 95 ℃ and maintain this temperature. Then, according to the set time interval, add sodium hydroxide and EDTA alternately, and stir evenly with a stirrer after each addition. During the insulation process, control the pH value of the reaction solution to 5.
Cooling and crystallization: Cool the reaction solution to room temperature, and EDTA disodium will crystallize and precipitate.
Separation and purification: Separate the precipitated EDTA disodium crystals from the mother liquor through filtration, and wash with an appropriate amount of water or suitable solvent to remove impurities and improve purity.
Drying: Dry the obtained EDTA disodium crystals to remove residual moisture.
Quality inspection: Conduct quality inspection on the final EDTA disodium, including inspection of appearance, purity, content, etc., to ensure that the product quality is qualified.
The above steps are a simplified description of the synthesis method of EDTA disodium based on the provided information. In practical operation, it may be necessary to make some adjustments and optimizations in details, such as controlling the feeding speed, adjusting the pH value, etc., to ensure the acquisition of high-quality EDTA disodium products.

EDTA disodium salt solution (EDTA-2Na) is a white crystalline powder with six coordinating atoms that can form stable chelates with metal ions. Its chemical properties are stable, soluble in water and almost insoluble in ethanol. It is widely used in various fields such as medicine, industry, agriculture, food processing, and cosmetics.
1. Heavy metal detoxification and blood purification
EDTA-2Na is a commonly used heavy metal detoxifier in the medical field, especially suitable for the treatment of heavy metal poisoning such as lead and cadmium. It chelates metal ions to form soluble complexes, promoting the excretion of heavy metals through urine. During hemodialysis and blood purification, EDTA-2Na can remove heavy metal ions and other harmful substances from the blood, preventing metal ion deposition from causing damage to organs. For example, in patients with lead poisoning, EDTA-2Na treatment can significantly reduce blood lead levels and improve neurological symptoms.
2. Anticoagulation and blood tests
In blood tests, EDTA-2Na acts as an anticoagulant by chelating calcium ions in the blood, preventing prothrombin activation and thus preventing blood clotting. This characteristic makes it a commonly used anticoagulant for blood routine tests, blood type identification, and other experiments.


Compared with heparin, blood samples anticoagulated with EDTA-2Na are more stable and suitable for long-term storage and transportation.
3. Disease treatment and drug formulations
EDTA-2Na can also be used to treat diseases such as iron overload and copper overload. For example, in patients with hereditary hemochromatosis (iron overload), EDTA-2Na can reduce the damage of iron deposition to organs such as the liver and heart by chelating excess iron ions in the body. In addition, EDTA-2Na, as a drug stabilizer, can prevent metal ion catalyzed oxidation reactions in drugs and extend their shelf life. In the treatment of cardiovascular diseases, EDTA-2Na is helpful to improve blood circulation, but clinical trials in North America show that EDTA-2Na does not significantly improve the incidence of cardiovascular events in patients with diabetes and myocardial infarction history, and further research is needed on its clinical application value.
1. Dyes and coatings industry
In the production of dyes and coatings, EDTA-2Na serves as a stabilizer and complexing agent, which can prevent metal ions (such as iron and copper) from catalyzing dye decomposition and improve product color fastness and stability. For example, adding EDTA-2Na to acidic dyes can inhibit discoloration and precipitation of dyes during storage, and extend product shelf life.
2. Detergent and Soap Industry
EDTA-2Na is a common additive in detergents and liquid soaps, which chelates calcium and magnesium ions in water to prevent the impact of hard water on washing effectiveness. It can enhance the cleaning and emulsifying power of detergents, prevent sedimentation, and thus improve washing quality. For example, in hard water areas with high calcium and magnesium ions, adding EDTA-2Na detergent can significantly improve the cleaning effect.


3. Rubber and Plastic Industry
In the production of synthetic rubber such as styrene butadiene rubber, EDTA-2Na is used as a polymerization terminator and activator. By complexing with metal catalysts such as ferrous ions, the polymerization reaction rate is controlled to improve product quality. In addition, EDTA-2Na can also be used for metal ion shielding in plastic processing to prevent the influence of metal impurities on plastic properties.
4. Water treatment and environmental protection
EDTA disodium salt solution, as a water treatment agent, can prevent the adverse effects of metal ions such as calcium, magnesium, iron, and manganese in water. For example, adding EDTA-2Na to boiler water can prevent scale formation and improve thermal efficiency; In sewage treatment, EDTA-2Na can chelate heavy metal ions, reduce the toxicity of pollutants, and facilitate subsequent treatment.
1. Trace element fertilizers
EDTA-2Na, as a chelating agent, can form stable chelates with trace elements such as iron, copper, and zinc, improving the absorption efficiency of plants towards trace elements. In the treatment of plant nutrient deficiencies such as iron deficiency yellowing, EDTA-2Na chelated iron fertilizer can significantly improve plant leaf color and promote growth. For example, in alkaline soil, EDTA-2Na chelated iron fertilizer can solve the problem of iron ion precipitation and improve fertilizer utilization efficiency.
2. Pesticides and agricultural sprays
In pesticides and agricultural sprays, EDTA-2Na can be used as a stabilizer to prevent the decomposition of pesticides catalyzed by metal ions and extend the efficacy. For example, adding EDTA-2Na to copper containing fungicides can improve their stability and reduce their frequency of use.

Food processing field: from color protection to anti-corrosion

1. Food color protection and antioxidant properties
According to GB2760-2014, food grade EDTA-2Na can be used as a stabilizer, coagulant, antioxidant, and preservative, and is widely used in beverages (excluding packaged drinking water), jams, vegetable puree (sauce), compound seasonings, and other foods. It prevents discoloration, deterioration, turbidity, and oxidation loss of vitamin C caused by metal ions by chelating metal ions. For example, adding EDTA-2Na to pickled pickled Chinese cabbage and pickles can make the dishes even and bright, prolong the storage period significantly, and have better color fixation and fresh-keeping effect than VC and phosphate.
2. Anti corrosion synergy and stability effect
EDTA-2Na can work synergistically with preservatives such as sodium benzoate to enhance the preservative effect of food. EDTA-2Na can prevent corrosion of the inner wall of metal cans and inhibit microbial growth in canned foods. For example, adding EDTA-2Na to canned vegetables and mushrooms can maintain product color, prevent boiling water from becoming cloudy, and extend shelf life.
3. Oil antioxidant
Trace metals in oil, such as iron and copper, have the effect of promoting oil oxidation. EDTA-2Na can complex with trace metal ions, inhibit oxidation reactions, and extend the shelf life of oil. Adding EDTA-2Na to edible oil and fried foods can delay oil spoilage and maintain food flavor.

Cosmetics field: stabilizers and skin protection

1. Cosmetics stabilizer
EDTA disodium salt solution, as a stabilizer in cosmetics, can adjust the acidity and alkalinity of the product, prevent metal ions from catalyzing the decomposition of components, and extend the shelf life. For example, adding EDTA-2Na to lotion and face cream can prevent discoloration and deterioration of products.
2. Skin protection and potential risks
EDTA-2Na has a slight irritant effect on the skin, and long-term excessive use may cause symptoms such as redness and stinging of the skin. Pregnant or lactating women should avoid using cosmetics containing EDTA-2Na, as other populations usually do not have any impact on their bodies under normal usage levels. For example, adding EDTA-2Na to liquid detergents can improve washing power, but the dosage needs to be controlled to prevent skin irritation.
Hot Tags: edta disodium salt solution cas 139-33-3, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, naphtholbenzein indicator, Chemical Reagent, Dithizone Reagent, Phenol red powder, diphenylcarbazide reagent, Mercury Reagent