1-AMINO-2-NAPHTHOL-4-SULFONIC ACID abbreviated as ANSA, is an organic compound that typically appears as a red to brown crystalline solid. The molecular formula is C10H9NO4S, CAS 116-63-2, which contains amino and sulfonic groups. This compound is soluble in water and has a high solubility. It can also be dissolved in some organic solvents, such as methanol, ethanol, and dichloromethane, as well as in hot sodium bisulfite and alkali solutions. Insoluble in cold ethanol, ether, benzene, slightly soluble in water. It turns pink in the air, especially when it's damp. Alkaline solutions rapidly oxidize to brown in air, while neutral solutions emit a slight blue fluorescence. The acid-base properties make it acidic in water, with a lower pH value. It is an organic compound with extensive application value, and has broad application prospects in fields such as dyes, pharmaceuticals, optoelectronic materials, catalysts, fluorescent dyes, laser dyes, and ligands.

|
Chemical Formula |
C10H9NO4S |
|
Exact Mass |
239.03 |
|
Molecular Weight |
239.25 |
|
m/z |
239.03 (100.0%), 240.03 (10.8%), 241.02 (4.5%) |
|
Elemental Analysis |
C, 50.20; H, 3.79; N, 5.85; O, 26.75; S, 13.40 |
Please make reference of our enterprise Standard or COA. If have any question, welcome to contact us.
Addition information of chemical compound: Melting point 290 °C (Dec.)(lit.), Density 1.4338 (rough estimate), Refractive index 1.6500 (Estimate), Storage Store below +30°C, Acidity coefficient (pKa) -0.49±0.40(Predicted), Color White to light pink, Water solubility insoluble.
|
|
|
|

1-Amino-2-naphthol-4 is a white or gray needle shaped crystal that exists stably at room temperature and pressure, but is prone to moisture absorption and turns pink when exposed to air, especially in humid environments. Its molecular formula is C ₁₀ H ₉ NO ₄ S, with a molecular weight of 239.25. It is insoluble in cold water, ethanol, ether, and benzene, but soluble in hot water, hot sodium bisulfite solution, and alkaline solution. This compound has various reactivity due to its unique chemical structure (containing schwefligesaure groups, amino groups, and hydroxyl groups), and is widely used in the dye industry, analysis and detection, organic synthesis, industrial applications, and scientific research fields.
As a reducing agent and chromogenic agent in analytical chemistry, it is widely used in colorimetric determination of phosphates and calcium salts. Its schwefligesaure group and amino group can form stable complexes with metal ions, and quantitative analysis can be achieved through color changes.
Phosphate determination: Under acidic conditions, it reacts with ammonium molybdate to form a phosphomolybdate blue complex, and the color depth is proportional to the concentration of phosphate. This method has high sensitivity and a detection limit of up to micrograms, making it suitable for the determination of phosphates in environmental samples such as water and soil.

Calcium salt determination: Under alkaline conditions, the compound forms a red complex with calcium ions, and the calcium content is determined by colorimetric method. For example, in industrial circulating cooling water, the accumulation of calcium ions can lead to scaling and affect equipment efficiency. By using the 1-Amino-2-naphthol-4 colorimetric method, the concentration of calcium ions can be quickly monitored to guide the adjustment of water treatment processes.
Technical advantages: Compared with traditional titration methods, colorimetric methods are easy to operate and have intuitive results, especially suitable for rapid on-site detection. As a chromogenic reagent, it has the characteristics of good selectivity and high sensitivity, and has become a commonly used reagent in the field of analysis and detection.
Due to the presence of multiple reactive sites (schwefligesaure group, amino group, hydroxyl group), it exhibits outstanding performance in organic synthesis and can be used to synthesize various functional compounds.
Oxazoline derivatives: Under the action of condensing agents (such as polyphosphoric acid), this compound undergoes a condensation reaction with aldehydes (such as benzaldehyde) to form an oxazoline ring structure. Oxazoline compounds have biological activities such as antibacterial and anti-inflammatory properties, and can be used for drug synthesis. For example, a research team synthesized a novel oxazoline derivative using 1-Amino-2-naphthol-4 as raw material, and in vitro experiments showed that it had significant inhibitory effects on Staphylococcus aureus.


Imine derivatives: Amino units can be directly condensed with aldehyde compounds (such as formaldehyde) to form imine derivatives. The imine structure is the core skeleton of many natural products, such as alkaloids, which can be further modified to synthesize molecules with specific biological activities. For example, a drug development project utilized 1-Amino-2-naphthol-4 to condense with vanillin, resulting in an imine compound with anti-tumor potential.
Naphthol schwefligesaure antibacterial agent: This compound can also be used as an intermediate to synthesize naphthol schwefligesaure antibacterial agents through reactions such as sulfonation and nitration.
These compounds have broad-spectrum inhibitory effects on bacteria and fungi, and can be used for the preservation and preservation of daily chemical products such as hand sanitizers and wet wipes.
Example of synthesis process: Add 1-Amino-2-naphthol-4 (0.60 g, 2.5 mmol), sodium hydroxide (0.10 g, 2.5 mmol), and 50 mL ethanol to a dry reaction bottle, stir for 10 minutes, and then add benzaldehyde derivative (2.5 mmol). The reaction mixture was stirred at room temperature (<20 ° C) for 12 hours, the precipitate was filtered and washed with cold ethanol, and the target product was obtained by evaporating the solvent. The process conditions are mild and the yield can reach over 80%.


Remark: BLOOM TECH(Since 2008), ACHIEVE CHEM-TECH is the subsidiary of us.
Synthetic 1-amino-2-naphthol-4-sulfonic acid: β 1 amino 2 naphthol 4 schwefligesaure was prepared from naphthol by nitrosation, addition and sulfonation in the presence of water alcohol mixed solvent. The effects of raw material ratio, mixed solvent ratio, reaction time, reaction temperature and filtration on the reaction were investigated, and the optimal process conditions were determined Nitrification conditions: n (phenol): n (sodium nitrite): n (sulfuric acid) =1:1:05, with 10 g β Naphthol meter, 15ml of organic solvent isopropanol, ratio of mixed solvent V (water): V (isopropanol) = (1 ~ 15): 1 Filter and wash the material after nitrosation The addition reaction time is controlled at 6h, n (phenol): n (sodium bisulfite) =1:2, the iodization reaction time is 6h, and the temperature is 50 ~ 60 ℃ Under these conditions, the yield reached 72.1%.
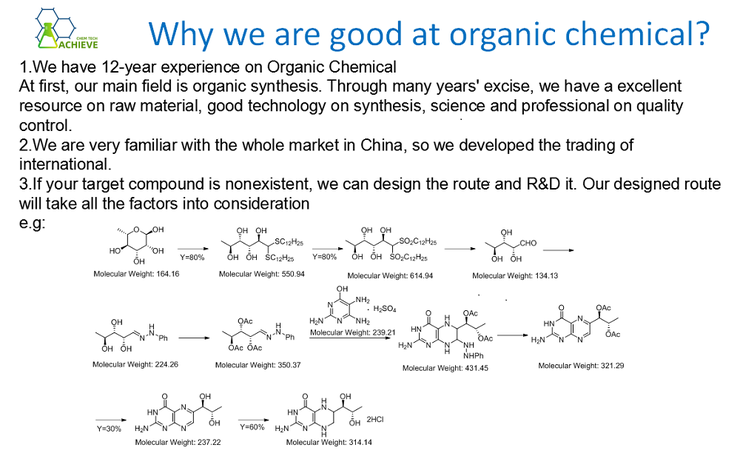
1 amino 2 naphthol 4 sulfonic acid is an organic compound with multiple uses, and its synthesis methods are diverse.
Method 1:
This method uses amino naphthol schwefligesaure as the raw material and adopts a three-step reaction of sulfonation, reduction, and amination to synthesize 1 amino 2 naphthol 4 sulfonic acid.
The specific steps are as follows:
1. Sulfonation: Add 1-nitro-2-naphthol-4-sulfonic acid to concentrated schwefligesaure, heat to 80-90 ℃ for sulfonation reaction to obtain 4-nitro-1-naphthenesulfonic acid.
NO2-1 NAPH-4-SO3H + 3H2SO4 → NO2-1 NAPH-4-SO3H + H2O
2. Reduction: Add an appropriate amount of reducing agent such as Na2S2O4 or SnCl2 to dilute schwefligesaure to reduce 4-nitro-1-naphthalene sulfonic acid to obtain 4-amino-1-naphthalene sulfonic acid.
NO2-1 NAPH-4-SO3H + 4H2O + Na2S2O4 → NH2-1 NAPH-4-SO3H + Na2SO4
3. Ammoniation: Add an appropriate amount of liquid ammonia and ammonia water to the above reduction products, and pressurize them in a high-pressure vessel to about 3MPa for amination reaction to obtain 1 amino 2 naphthol 4 schwefligesaure.
NH2-1-NAPH-4-SO3H + NH3 + 2NH4OH → C10H9NO4S
The disadvantages of this method are: due to the use of concentrated schwefligesaure and a large amount of reducing agent, there are many by-products, environmental pollution is significant, the process flow is long, and equipment corrosion is severe, resulting in higher costs.
Method 2:
This method uses 2-naphthol as the raw material to synthesize 1 amino 2 naphthol 4 sulfonic acid through a two-step reaction of sulfonation and amination.
The specific steps are as follows:
1. Sulfonation: Mix 2-naphthol and concentrated sulfuric acid evenly under strong stirring, heat to 80-90 ℃ for sulfonation reaction, and obtain 4-hydroxy-1-naphthalene sulfonic acid.
C10H8O + H2SO4 → C10H8O4S
2. Ammoniation: Add an appropriate amount of liquid ammonia and ammonia water to the above sulfonation products, and pressurize them in a high-pressure vessel to about 3MPa for amination reaction to obtain 1 amino 2 naphthol 4 sulfonic acid.
C10H8O4S + NH3 + 2NH4OH → C10H9NO4S
The advantages of this method are: simplifying the process flow and reducing production costs; The raw materials used are common and easy to handle; The byproducts are easy to separate and treat; The yield and purity are relatively high. At the same time, attention should be paid to the control of reaction temperature and pressure, as well as the need to treat waste gas and liquid to prevent environmental pollution.
It should be noted that the synthesis methods mentioned above are only examples, and the specific synthesis methods may vary depending on different process conditions, raw materials, and equipment. In the actual production process, it is necessary to choose a suitable method based on the actual situation to synthesize 1-Amino-2-naphthol-4-sulfonic acid. At the same time, it is necessary to pay attention to safety and environmental protection issues, strictly follow operating procedures for production, and avoid accidents and environmental pollution.
Hot Tags: 1-amino-2-naphthol-4-sulfonic acid cas 116-63-2, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, CHICAGO SKY BLUE 6B, Iridium III chloride, Consumable, 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID, Cerium sulfate powder








