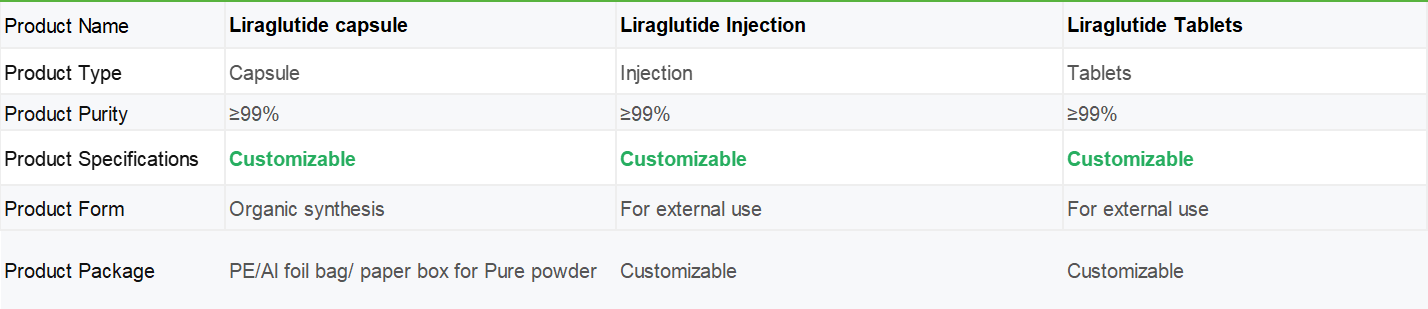
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of liraglutide capsule in China. Welcome to wholesale bulk high quality liraglutide capsule for sale here from our factory. Good service and reasonable price are available.
Liraglutide capsule is an artificially synthesized acylated analogue of human glucagon like peptide-1 (GLP-1). The changes in molecular structure mainly include the following two points: first, the 34th lysine of natural GLP-1 molecule is replaced by arginine, and second, a fatty acid side chain is added to the 26th lysine. This molecular change not only preserves and prolongs the binding time between acylation products and proteins, but also significantly overcomes the disadvantage of GLP degradation.
Our product




Additional information of chemical compound:
Our Product



Liraglutide+. COA


Pharmacological action
Liraglutide capsule is an acylated human glucagon like peptide-1 (GLP-1) receptor agonist, with 97% of its amino acid sequence homologous to endogenous human GLP-1 (7-37). GLP-1 (7-37) accounts for less than 20% of all endogenous GLP-1 in the blood. Similar to GLP-1 (7-37), liraglutde can activate GLP-1 receptors, which are membrane-bound cell surface receptors that couple with adenylate cyclase via stimulatory G protein Gs in pancreatic beta cells.
When glucose concentration increases, liraglutde can increase intracellular cyclic adenosine monophosphate (cAMP), leading to insulin release. When the blood glucose concentration decreases and tends to be normal, insulin secretion decreases. Liraglutde can also reduce glucagon secretion in a glucose dependent manner. The mechanism of decreased blood sugar levels also involves delayed gastric emptying.
GLP-1 is a physiological regulator of appetite and calorie intake, and GLP-1 receptors are present in multiple regions of the brain involved in appetite regulation. In animal experiments, peripheral administration of liraglutde resulted in the presence of liraglutde in specific brain regions that regulate appetite, including the hypothalamus. Although liraglutde activates neurons in brain regions known to regulate appetite, no specific brain region has been found to mediate the effect of liraglutde on appetite in rats.
Pharmacodynamic effects
The effect of liraglutde lasts for 24 hours and can improve blood glucose control by reducing fasting and postprandial blood glucose in patients with type 2 diabetes.
In patients with type 2 diabetes, a single administration of rilalutide can observe that the insulin secretion rate increases in a glucose concentration dependent manner (Figure 4).
Fig. 4 Average insulin secretion rate (ISR) and glucose concentration during stepwise glucose infusion in patients with type 2 diabetes (N=10) after receiving 7.5 ug/kg (about 0.7 mg) of lilalutide or placebo for a single time.

Drug interactions
In vitro studies have confirmed that the likelihood of pharmacokinetic interactions between liraglutide capsule and other active substances related to cytochrome P450 and plasma protein binding is extremely low. Mild delay in gastric emptying caused by liraglutde may affect the absorption of other drugs taken orally at the same time. The interaction study did not show any clinically relevant delays in drug absorption. Therefore, there is no need to adjust the dosage.

1. Warfarin and other coumarin derivatives
The possibility of clinically significant interactions between liraglutde and active ingredients with poor solubility or narrow therapeutic indices (such as warfarin) cannot be ruled out. Patients receiving warfarin or other coumarin derivatives treatment are recommended to undergo more frequent INR (International Normalized Ratio) monitoring after starting treatment with this product.
2. Paracetamol
Liraglutde does not alter the overall exposure after a single dose of 1000mg of paracetamol. The peak concentration (Cmax) of paracetamol decreased by 31%, while the median time to peak (Tmax) was delayed by 15 minutes. When used in combination with paracetamol, no dosage adjustment is required.
3. Atorvastatin
Liraglutide did not produce clinically significant changes in overall exposure after a single dose of 40mg atorvastatin. Therefore, there is no need to adjust the dosage when atorvastatin is used in combination with this product. Under the action of liraglutde, the peak concentration (Cmax) of atorvastatin decreased by 38%, while the median time to peak (Tmax) was extended from 1 hour to 3 hours.
4. Griseomycin
Liraglutde does not alter the overall exposure after a single administration of 500mg of gibberellin. The peak concentration (Cmax) of oxytetracycline increased by 37%, while the median time to peak (Tmax) remained unchanged. The combination of oxytetracycline and other low solubility and high permeability drugs with this product does not require dosage adjustment.


5. Digoxin
A single dose of 1mg digoxin combined with Liraglutde can reduce the area under the curve (AUC) of digoxin by 16% and Cmax by 31%. Tmax has been extended from 1 hour to 1.5 hours. Based on the above results, there is no need to adjust the dosage of digoxin.
6. Lisinopril
A single dose of 20mg of lisinopril combined with liraglutde can reduce the area under the curve (AUC) of lisinopril by 15% and 16%, and the peak concentration (Cmax) by 27%, respectively. The median Tmax for reaching the peak concentration of lisinopril was extended from 6 hours to 8 hours. Based on the above results, there is no need to adjust the dosage of lisinopril.
9. Compatibility taboos
The substances added to this product may cause degradation of liraglutde. Without conducting compatibility contraindication studies, this product should not be mixed with other drugs.
7. Oral contraceptive pills
After a single administration of an oral contraceptive pill, Liraglutde reduced the peak concentrations (Cmax) of ethinylestradiol and levonorgestrel by 12% and 13%, respectively. Liraglutde extended the peak time (Tmax) of both components by 1.5 hours. There were no clinically significant effects on overall exposure to ethinylestradiol or levonorgestrel. Therefore, the combination of liraglutde is not expected to affect the contraceptive effectiveness of oral contraceptives.
8. Dexmedetomidine insulin
In patients with type 2 diabetes, 0.5U/kg insulin detemir (single dose) and 1.8mg linaglutide (steady state) were injected subcutaneously, and no pharmacokinetic interaction between linaglutide and insulin detemir was observed.

Brief History of Listing
Lilalutide, developed by Danmino and Nordisk, was approved to be listed in the European Union and the United States in July 2009 and January 2010, respectively, and in China in 2011. It was approved by the State Food and Drug Administration for the treatment of adult type 2 diabetes.
On December 4, 2020, the US FDA approved liraglutde for the treatment of obesity in adolescents aged 12 to 17 years with a body weight of 60 kg or more or a BMI ≥ 30 kg · m-2. It can be used as an adjuvant drug for low calorie diets and increased exercise.
On March 30, 2023, Hangzhou Zhongmei Huadong Pharmaceutical Co., Ltd., a wholly-owned subsidiary of East China Pharmaceutical, received the Drug Registration Certificate approved and issued by the State Drug Administration (NMPA). The application for the marketing license of Lilalutide injection applied for adult type 2 diabetes patients to control blood sugar was approved. (Drug approval number: National Medical Products Administration Approval No. S20233109, National Medical Products Administration Approval No. S20233110), product name: Lirupine ®, Specification: 3ml: 18mg (pre filled injection pen); 3ml: 18mg (pen core).
On July 12, 2023, Hangzhou Zhongmei Huadong Pharmaceutical Co., Ltd. received the "Drug Registration Certificate" approved by the National Medical Products Administration (NMPA), and the application for marketing authorization of Liraglutde Injection for obesity or overweight indications was approved. (Drug approval number: National Medical Products Administration Approval No. S20233109, National Medical Products Administration Approval No. S20233110), product name: Lirupine ®, Specification: 3ml: 18mg (pre filled injection pen); 3ml: 18mg (pen core)
Challenges and Prospects of Capsule Formulation Development
Development Challenges
Formulation technology: The development of Liraglutde formulations requires solving the problem of stable release and absorption of drugs in the gastrointestinal tract. Special formulation techniques such as enteric coating and osmotic pump technology are required to ensure the stability and bioavailability of the drug in the gastrointestinal tract.
Clinical trials: New clinical trials are required to validate the efficacy, safety, and tolerability of capsule formulations. Clinical trials should include different doses, different dosing regimens, and long-term safety studies.
Market competition: Currently, the market for Liraglutde Injection is relatively mature, and capsule formulations need to face competition from injections. We need to win market share through differentiation strategies such as medication convenience, patient compliance, etc.
Development prospects
Medication convenience: Capsule formulations are more convenient to administer compared to injections, which can improve patient compliance. For patients who have a fear of or inconvenience with injections, capsule formulations may be a better choice.
Market demand: With the increasing number of patients with type 2 diabetes and obesity, the demand for new hypoglycemic and weight reducing drugs is also growing. If the formulation of Liraglutde is successfully developed, it will meet the market demand for new hypoglycemic and weight loss drugs.
Policy support: The government's increasing support for the biopharmaceutical industry has provided a favorable policy environment for the development of Liraglutide capsule formulations. Capsule formulations may reduce patients' financial burden and improve drug accessibility through inclusion in medical insurance catalogs and other means.
Hot Tags: liraglutide capsule, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale




