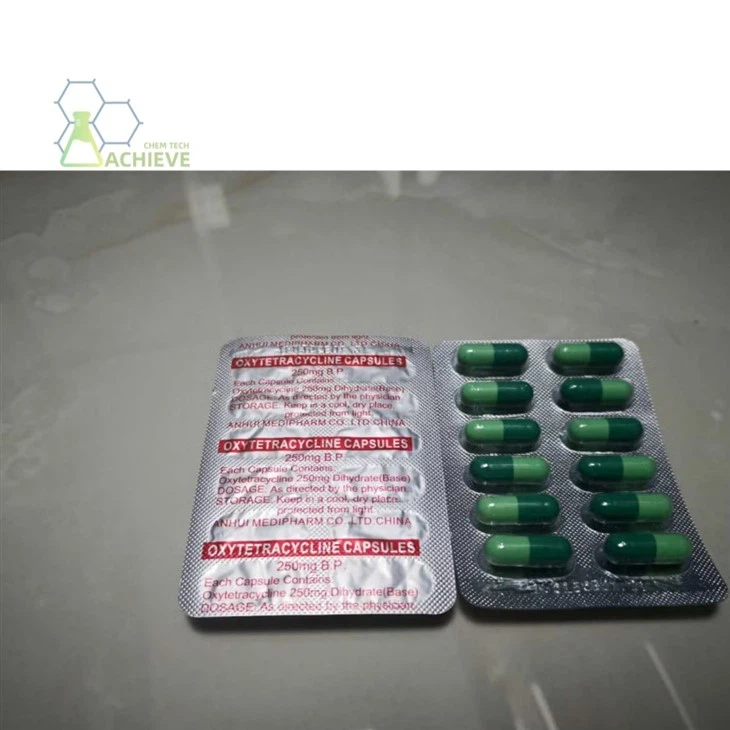
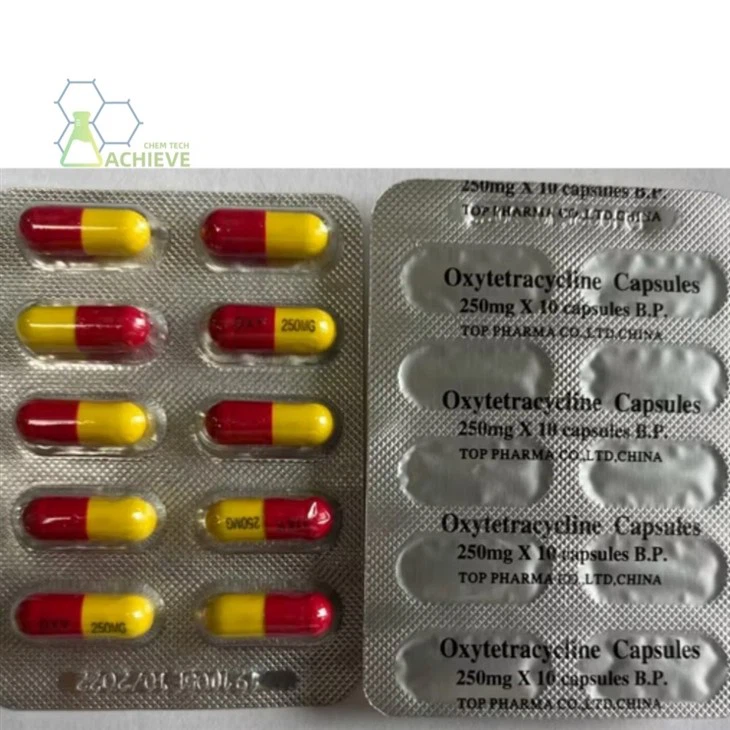
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of oxytetracycline capsule 250mg in China. Welcome to wholesale bulk high quality oxytetracycline capsule 250mg for sale here from our factory. Good service and reasonable price are available.
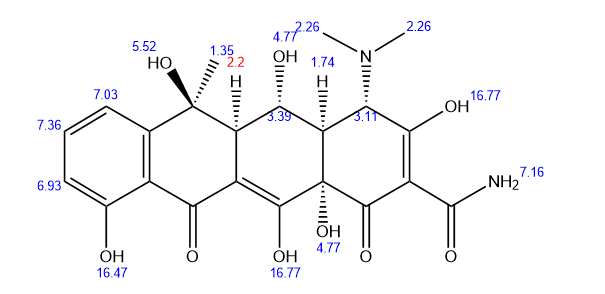
The core component of oxytetracycline capsule 250mg is oxytetracyclie, with the chemical name 6-methyl-4- (dimethylamino) -3,5,6,10,12,12a-hexahydroxy-1,11-dioxo-1,4,4a, 5,5a, 6,11,112a-octahydro-2-tetrabenzamide. Its hydrochloride form is a yellow crystalline powder that is easily soluble in water. Clinical preparations often use such derivatives to improve solubility. The formulation is in the form of capsules, with a typical specification of 0.125g (125000 units) or 0.25g (250000 units) per capsule, and packaging specifications ranging from 24 capsules/box to 100 capsules/box.
Our product



Additional information of chemical compound:
| Product Name | Oxytetracycline Powder | Oxytetracycline Tablets | Oxytetracycline Capsule | Oxytetracycline Cream |
| Product Type | Powder | Tablet | Capsule | Cream |
| Product Purity | ≥99% | ≥99% | ≥99% | ≥99% |
| Product Package | Customizable | Customizable | Customizable | Customizable |
Our Product



Toltrazuril +. COA
 |
||
Certificate of Analysis |
||
|
Compound name |
Oxytetracycline | |
|
CAS No. |
79-57-2 | |
|
Grade |
Pharmaceutical grade | |
|
Quantity |
Customized | |
|
Packaging standard |
Customized | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
|
Lot No. |
20250109001 |
|
|
MFG |
Jan 12th 2025 |
|
|
EXP |
Jan 8th 2029 |
|
|
Structure |
|
|
| TEST STANDARD | GB/T24768-2009 Industry. Stnndard | |
|
Item |
Enterprise standard |
Analysis result |
|
Appearance |
White or almost white powder |
Conformed |
|
Water content |
≤4.5% |
0.30% |
| Loss on drying |
≤1.0% |
0.15% |
|
Heavy Metals |
Pb≤0.5ppm |
N.D. |
|
As≤0.5ppm |
N.D. | |
|
Hg≤0.5ppm |
N.D. | |
|
Cd≤0.5ppm |
N.D. | |
|
Purity (HPLC) |
≥99.0% |
99.5% |
|
Single impurity |
<0.8% |
0.48% |
|
Residue on ignition |
<0.20% |
0.064% |
|
Total microbial count |
≤750cfu/g |
80 |
|
E. Coli |
≤2MPN/g |
N.D. |
|
Salmonella |
N.D. | N.D. |
|
Ethanol (by GC) |
≤5000ppm |
400ppm |
|
Storage |
Store in a sealed, dark and dry place at-20 degrees |
|
|
|
||

The mechanism of action of oxytetracycline capsule 250mg can be systematically analyzed from four aspects: molecular targeting, antibacterial spectrum coverage, resistance mechanism response, and clinical application correlation
The core component of oxytetracyclie capsules, oxytetracyclie, belongs to the tetracycline class of antibiotics, and its target is the A site of the bacterial ribosome 30S subunit. The specific mechanism is as follows:
Binding site competition: Oxytetracyclie specifically binds to the A site of the 30S subunit, blocking the docking of aminoacyl tRNA and mRNA ribosome complexes, thereby inhibiting peptide chain elongation.
Interruption of protein synthesis: Due to the occupation of the A site, bacteri are unable to complete peptide bond formation, resulting in the termination of protein synthesis. This process is highly selective for bacteri, as mammalian ribosomes have an 80S structure (containing 40S and 60S subunits) and do not cross react with bacterial 30S subunits.
Concentration dependent effect: At low concentrations, it exhibits antibacterial activity (blocking protein synthesis), while at high concentrations, it exhibits bactericidal activity against sensitive bacteri (such as rickettsia and mycoplasma) (by disrupting cell membrane integrity).

Antibacterial spectrum coverage: the pathogenic basis of broad-spectrum inhibition

The antibacterial spectrum covers four major categories of pathogenic microorganisms, and their mechanisms of action are closely related to the characteristics of the pathogens:
Gram positive bacteri:
Sensitive strains: Staphylococcus aureus (non enzyme producing strain), Streptococcus pneumoniae, and Streptococcus pyogenes.
Weakness: Enterococcus is naturally resistant (due to the presence of TetM ribosome protective protein), while Staphylococcus has a resistance rate of 75% -85% (producing TetK efflux pumps).
Gram negative bacteri:
Sensitive strains: Escherichia coli, Pseudomonas aeruginosa, Shigella, Yersinia, Brucella.
Drug resistance mechanism: Deficiency of Gram negative bacterial outer membrane porin protein (such as downregulation of OmpF/OmpC) leads to drug permeation disorders. Combined with TetA efflux pump expression, the drug resistance rate is about 50% -60%.
Atypical pathogens:
Mycoplasma: Penetrating the cell membrane to inhibit ribosome function, MIC ≤ 0.5 μ g/mL for Mycoplasma pneumoniae.
Chlamydia: inhibits inclusion body formation and is highly sensitive to serotypes A-C of Chlamydia trachomatis.
Rickettsia: By inhibiting the synthesis of energy metabolism related proteins, the MIC of Rickettsia prussii is ≤ 0.25 μ g/mL.
Other pathogens:
Spiroplasma: inhibits the synthesis of movement related proteins, is ineffective against Treponema pallidum, but can treat leptospirosis.
Amoebic protozoa: Inhibits the synthesis of nourishing body proteins, and is more effective than carbapenem in treating acute amoebic dysentery.
Response to Drug Resistance Mechanisms: Cross Resistance and Combnation Drug Strategies

Drug resistance gene lineage:
Exopump genes: TetA (Gram negative bacteri) and TetK (Gram positive bacteri) encode membrane proteins that actively excrete drugs.
Ribosomal protection gene: TetM encodes proteins that bind to tetracycline and competitively displace drugs.
Enzyme modified gene: TetX encodes a monooxygenase that oxidizes and degrades the structure of oxytetracyclie (rare but highly threatening).
Cross resistance characteristics:
Oxytetracyclie has complete cross resistance with tetracycline and doxycycline (due to the same target of action).
There is no cross resistance with macrolides such as azithromycin (targeting the 50S subunit).
Combnation therapy plan:
Brucellosis: Combnation of oxytetracyclie (2g/day) and streptomycin (1g/day), with a treatment course of 6 weeks (covering the intracellular parasitic stage).
Pestis: Oxytetracyclie (2g/day) combined with gentamicin (160mg/day) for 10 days (to preven the progress of sepsis).
Drug resistant Mycoplasma pneumoniae pneumonia: Combnation of tetracycline (1.5g/day) and moxifloxacin (400mg/day) for 14 days.
Rickettsia disease:
Scrub typhus: By inhibiting the ribosome function of scrub typhus, the fever phase can be shortened for 3-5 days and the mortality rate can be reduced.
Q fever: Combined with doxycycline can preven chronic Q fever (the course of treatment needs to be extended to 3 years).
Atypical pneumonia:
Mycoplasma pneumonia: The cure rate of tetracycline combined with levofloxacin for macrolide resistant strains is 92%.
Chlamydia psittaci pneumonia: It is necessary to combine glucocorticoids (such as prednisone 20mg/day) to control the inflammatory storm.
Biological terrorism defense:
Pestis: As a first-line drug recommended by WHO, it can reduce the mortality of lung pestis (from 90% to 40%).
Anthrax: Effective against skin anthrax, but requires combined antitoxin treatment (as oxytetracyclie does not neutralize toxins).

As a representative drug of tetracycline antibiotics, the clinical application of oxytetracycline capsule 250mg should strictly follow the rules of drug interactions and contraindication management to avoid reduced efficacy or worsening adverse reactions.
Drug interaction mechanism and clinical management:
Oxytetracyclie exerts antibacterial effects by interfering with bacterial protein synthesis, but its chemical structure (containing multiple hydroxyl and carbonyl groups) is prone to physiological reactions with various drugs or food components, leading to abnormal absorption, distribution, metabolism, or excretion processes.
Acid making drugs and metal ion preparations: When used in combnation with acid making drugs such as sodium bicarbonate and magnesium aluminum carbonate, the pH in the stomach increases, causing a decrease in the solubility of oxytetracyclie and a reduction in absorption by 30% -50%. Drugs containing calcium (such as calcium tablets), magnesium (such as antacids), and iron (such as ferrous sulfate) can form insoluble chelates with tetracycline, reducing blood drug concentrations by 60% -70%.
Management strategy: The combnation of the above drugs should be taken 2-3 hours apart, and avoid taking them with milk or antacids.
Lipid lowering resin: Anion exchange resins such as colistin and colistin can adsorb oxytetracyclie and reduce its enterohepatic circulation.
Management strategy: It is necessary to take it every 4-6 hours or choose other antibiotics.
Renal toxicity drug synergy: When used in combnation with the general anesthetic methoxyflurane, oxytetracyclie can enhance its tubular toxicity, leading to a threefold increase in the risk of acute kidney failure; When used in combnation with strong diuretics such as furosemide, changes in renal hemodynamics may exacerbate renal injury.
Management strategy: Avoid combnation therapy and monitor renal function (creatinine, urea nitrogen) if necessary.
Combnation of hepatotoxic drugs: When used in combnation with anti-tumor chemotherapy drugs (such as methotrexate) and antiepileptic drugs (such as sodium valproate), oxytetracyclie can induce hepatic enzyme activity and exacerbate liver cell necrosis.
Management strategy: During the combnation period, liver function (ALT, AST) should be monitored, and the dosage should be adjusted if necessary.
Anticoagulant therapy interference: Oxytetracyclie can inhibit intestinal vitamin K synthesis, reduce prothrombin activity, and enhance the effect of anticoagulants such as warfarin and rivaroxaban by 20% -40%.
Management strategy: During the combnation period, INR values should be monitored and the dosage of anticoagulant drugs should be adjusted.
Hormonal drug failure: Oxytetracyclie can reduce the blood concentration of oral contraceptives (such as ethinylestradiol) and increase the risk of breakthrough bleeding.
Management strategy: It is recommended to use barrier contraception during the combnation period.
Adverse reactions and risk prevention and control
Digestive system: Nausea (incidence 15% -20%), vomiting (8% -12%), upper abdominal discomfort (10% -15%), abdominal distension (5% -8%), occasional pancreatitis (0.1% -0.3%), and esophageal ulcers (mostly seen in patients who immediately lie in bed after taking medication).
Hepatotoxicity: The incidence of fatty liver degeneration is 3% -5%, and the risk is increased in pregnant women and patients with renal dysfunction (OR value of 4.2-6.8).
Allergic reactions: papules (2% -3%), erythema (1% -2%), urticaria (0.5% -1%), occasional anaphylactic shock (0.01% -0.05%), and photosensitive dermatitis (increased incidence of erythema by 30% after sun exposure).
Double infecton: Long term medication can induce drug-resistant Staphylococcus aureus (incidence 5% -8%), Gram negative bacilli (3% -5%), and fungal infectons (2% -4%), and in severe cases, sepsis.
Hematological system: Decreased granulocytes (0.1% -0.3%), decreased platelets (0.05% -0.1%).
Central nervous system: benign intracranial hypertension (headache, vomiting, optic disc edema, incidence 0.01% -0.05%).
Pre medication screening: Patients with a history of tetracycline allergy are prohibited from using oxytetracycline capsule 250mg. When treating sexually transmitted diseases, Treponema pallidum infecton should be ruled out (dark field microscopy+serological examination).
Monitoring indicators: Blood routine and liver and kidney function (ALT/AST elevation rate of 5% -8%, blood urea nitrogen elevation rate of 3% -5%) are tested every 2 weeks for long-term drug users.
Medication education:
Drink 240ml of water while taking medication to avoid esophageal ulcers.
Avoid bed rest within 1 hour after taking medication to reduce the risk of gastroesophageal reflux.
Avoid direct sunlight and reduce the incidence of photosensitive dermatitis.
Hot Tags: oxytetracycline capsule 250mg, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale