The excellent quality of our N N Dimethylformamide pure, 3 Dimethylaminophenol, Basic Chemicals has won us market recognition and recognition from the industry. We are ready to face the worries and make the improve to understand all the possibilities in international trade. We have strong capital operation strength and abundant supply of resources.
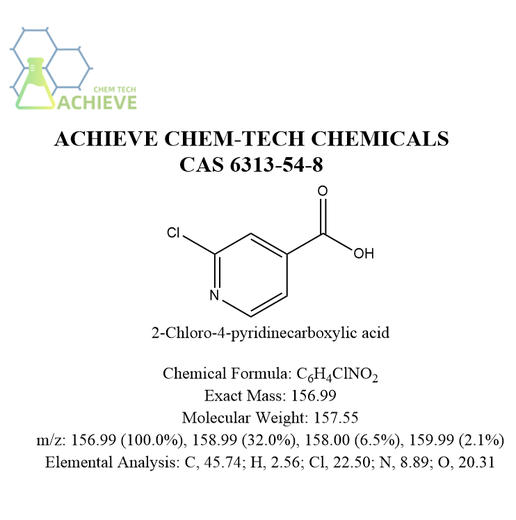
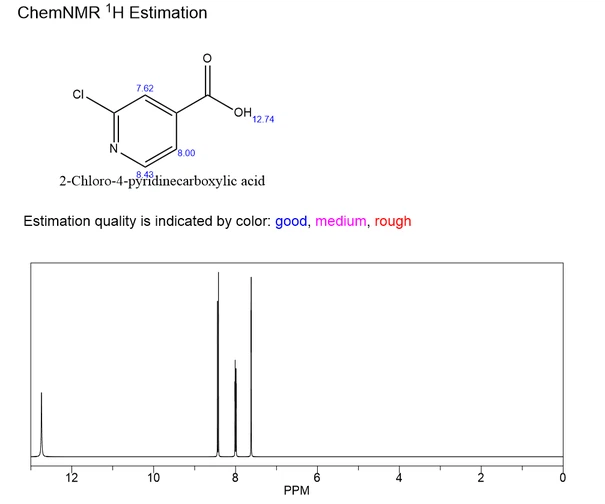
2-Chloro-4-pyridinecarboxylic acid is an organic compound with CAS 6313-54-8 and chemical formula C6H4ClNO2. Usually white or light yellow powder with a slight irritating odor. Slightly soluble in water, slightly soluble in ethanol, insoluble in ether. Stable at room temperature, but may decompose under high temperature or exposure to light. The structure contains a free carboxyl unit and a chlorine atom. Due to the electron deficiency of the pyridine ring, this substance can undergo a series of nucleophilic substitution reactions under the attack of strong nucleophilic reagents, resulting in a series of dechlorinated functionalized products. It is a pyridine derivative that can combine with acidic substances to form salts. It can be used as an intermediate in organic synthesis and pharmaceutical chemistry, and is often used for structural modification and synthesis of drug molecules and bioactive molecules. For example, relevant literature has reported that this substance can be used in the synthesis of high camptothecin derivatives with anticancer activity.

|
Chemical Formula |
C6H4ClNO2 |
|
Exact Mass |
156.99 |
|
Molecular Weight |
157.55 |
|
m/z |
156.99 (100.0%), 158.99 (32.0%), 158.00 (6.5%), 159.99 (2.1%) |
|
Elemental Analysis |
C, 45.74; H, 2.56; Cl, 22.50; N, 8.89; O, 20.31 |
| Melting point |
246 ° C (Dec.) (lit.) |
|
Boiling point |
417.7 ± 25.0 ° C (predicted) |
|
Density |
1.470 ± 0.06 g / cm3 (predicted) |
|
|
|

2,6-Dichloroisoniacin is obtained through chlorination reaction, followed by chlorination reaction to obtain 2-chloroisoniacin, which is then synthesized through directed dechlorination reaction.
Step 1: Chlorination reaction to obtain 2,6-dichloroisononicotinic acid:
(1) Under appropriate reaction conditions, react isoniacin with chlorinating agents (such as sulfoxide chloride). The function of a chlorinating agent is to replace the hydrogen atom in isoniacin with the chlorine atom.
(2) After the reaction is completed, 2,6-dichloroisonotinic acid is obtained through appropriate separation and purification methods (such as distillation, crystallization, etc.). The product of this step is 2,6-dichloroisonotinic acid.
C6H3Cl2NO2 + HCl → 2,6-Dichloroisonicotinic acid hydrochloride
Step 2: Chlorination reaction to obtain 2-chloroisoniatinic acid:
(1) React the 2,6-dichloroisonotinic acid obtained in the previous step with a chlorinating agent again. This time, the action of the chlorinating agent is to selectively replace one chlorine atom in 2,6-dichloroisononicotinic acid with another chlorine atom, thereby forming 2-chloroisonicotinic acid.
(2) After the reaction is completed, 2-chloroisonicotinic acid is also obtained through appropriate separation and purification methods. The product of this step is 2-chloroisonicotinic acid.
2,6-Dichloroisoniazid hydrochloride + HCl → C6H4ClNO2
Step 3: Preparation of 2-chloroisonicotinic acid through directed dechlorination reaction:
(1) React the 2-chloroisonicotinic acid obtained in the previous step with a directed dechlorination agent. The function of directional dechlorination agent is to selectively remove chlorine atoms from 2-chloroisoniacin, thereby obtaining the target product 2-chloroisoniacin.
(2) After the reaction is completed, pure 2-chloroisonicotinic acid is obtained through appropriate separation and purification methods. The product of this step is the target 2-Chloro-4-pyridinecarboxylic acid.
C6H4ClNO2 + directional dechlorination agent → C6H4ClNO2
We are the factory of 2-Chloro 4-pyridinecarboxylic acid. Remark: BLOOM TECH(Since 2008), ACHIEVE CHEM-TECH is the subsidiary of us. Our transportation modes include sea transportation, air transportation and land transportation. We prepare various ways to meet the different needs of customers, so as to better serve customers and achieve a win-win situation.

The transportation of chemical products usually requires following a series of safety regulations and procedures to ensure safety and efficiency during the transportation process. Here are some basic steps and precautions for chemical transportation:
1. Understanding transportation regulations: Before starting transportation, it is necessary to understand and comply with relevant transportation regulations. These regulations may involve regulations on the transportation of dangerous goods, as well as transportation requirements specific to specific types of chemicals.
2. Choose the appropriate transportation method: Choose the appropriate transportation method based on the nature, quantity, and transportation distance of the chemical product. The common modes of transportation include land, sea, and air.
3. Prepare transportation documents: A detailed transportation document needs to be prepared, including a detailed description of the goods, quantity, destination, transportation method, and carrier information. In addition, it is necessary to provide proof of the safety and stability of the goods.
4. Packaging Chemicals: Chemicals typically require special packaging to ensure safety during transportation. The packaging should comply with the hazardous material packaging and labeling requirements specified by the International Air Transport Association (IATA).
5. Comply with transportation regulations: During transportation, it is necessary to comply with all transportation regulations, including loading, securing, protecting, and preventing leakage of goods. In addition, specific traffic rules and speed restrictions need to be followed.
6. Maintain communication: Throughout the transportation process, it is necessary to maintain communication with the carrier and destination in order to promptly address any potential issues.
7. Recording and reporting: During transportation, all activities and events need to be recorded and reported to relevant departments as needed.
Please note that these steps are only general guidance and cannot replace specific regulations and rules. Before starting transportation, it is best to consult a professional transportation company or organization to ensure compliance with all regulations and rules.

2-Chloro-4-pyridinecarboxylic acid has a wide range of applications in chemistry. The following is a detailed description of all its uses in chemistry:
1. Organic synthesis
It is an important intermediate in organic synthesis. It can participate in various organic reactions, such as esterification, amidation, alkylation, etc., thereby constructing various complex organic molecules. By transforming and modifying its functional groups, compounds with different structures and properties can be synthesized, providing rich synthesis routes and strategies for organic synthesis.
2. Analytical Chemistry
It also has applications in analytical chemistry. It can be used as a fluorescent probe, chromatographic separator, etc. for chemical analysis. For example, by utilizing the fluorescence properties of 2-Chloro 4-pyridinecarboxylic acid, highly sensitive fluorescent probes can be designed for detecting pollutants in the environment, metabolites in living organisms, and more. In addition, it can also serve as a chromatographic separation agent for the separation and analysis of complex samples.
3. Electrochemistry
It also has certain applications in the field of electrochemistry. It can be used as a battery material, capacitor material, etc. for electrochemical energy storage and conversion. For example, by utilizing the redox properties of 2-Chloro 4 pyridinecarboxylic acid, high-performance battery materials can be designed to improve the energy storage density and cycling stability of the battery. In addition, it can also be used to synthesize capacitor materials, improving the performance and service life of capacitors.
2-Chloro-4-Pyridinecarboxylic Acid is far more than a laboratory curiosity-it is a linchpin of modern chemistry, enabling breakthroughs in medicine, agriculture, and materials. Its unique structure, coupled with its reactivity and versatility, ensures its place in the pantheon of essential organic compounds.
As researchers push the boundaries of synthesis and application, 2-Cl-4-PCA will continue to evolve, proving that even the simplest molecules can drive profound scientific and industrial advancements. Whether in the fight against disease, the protection of crops, or the development of cutting-edge materials, this pyridine derivative exemplifies the power of chemistry to transform our world.
Industrial Applications: From Pharmaceuticals to Agrochemicals
► Pharmaceutical Intermediates
2-Cl-4-PCA is a key building block in anti-inflammatory, antiviral, and anticancer drugs.
Case Study: COX-2 Inhibitors
Cyclooxygenase-2 (COX-2) inhibitors, used to treat arthritis, often incorporate pyridine derivatives for enhanced potency. 2-Cl-4-PCA serves as a precursor to:
Celecoxib (Celebrex®): A selective COX-2 inhibitor.
Rofecoxib (Vioxx®, withdrawn): Another COX-2 inhibitor.
Synthesis Pathway:
2-Cl-4-PCA is converted to its acid chloride (2-Cl-4-PCA-Cl) using SOCl₂.
The acid chloride reacts with an arylamine (e.g., 4-methylsulfonylphenylamine) to form an amide.
The amide undergoes cyclization to yield the COX-2 inhibitor core.
► Agrochemical Development
Pyridine derivatives are widely used in herbicides, insecticides, and fungicides. 2-Cl-4-PCA contributes to:
Herbicide Synthesis
Fluroxypyr: A broadleaf herbicide used in cereal crops.
2-Cl-4-PCA is esterified with 3,4,5-trifluorophenol to form fluroxypyr.
Insecticide Intermediates
Imidacloprid: A neonicotinoid insecticide.
2-Cl-4-PCA is converted to a nitroimine derivative, a key intermediate in imidacloprid synthesis.
► Material Science: Coordination Polymers and Catalysis
The carboxylic acid group of 2-Cl-4-PCA enables its use in metal-organic frameworks (MOFs) and catalysts.
MOF Synthesis
2-Cl-4-PCA can coordinate with transition metals (e.g., Zn²⁺, Cu²⁺) to form porous MOFs for gas storage or separation.
Heterogeneous Catalysis
Palladium complexes of 2-Cl-4-PCA are used in cross-coupling reactions (e.g., Suzuki-Miyaura coupling).
► Specialty Chemicals
Dyes and Pigments: The pyridine ring can be functionalized to create chromophores.
Corrosion Inhibitors: 2-Cl-4-PCA derivatives form protective films on metal surfaces.
We actively improve our independent research, development and innovation capabilities, and hope to create the best quality 2-Chloroisonicotinic Acid CAS 6313-54-8 2-Chloro-4-Pyridinecarboxylic Acid. Advocating professionalism, producing first-class products and providing first-class service is the guarantee for our company to develop. Our company has a large inventory, complete varieties, stable supply, timely supply, won the praise of our customers.