We are well aware that technology is a direct manifestation of the company's hard power and the cornerstone for us to provide better Chemical Reagent, tetracaine powder, DIMETHYLPHOSPHINE OXIDE to customers. Only by meeting the needs of customers, customers will be willing to pay the bill, and enterprises can make profits. We focus on the main line of high-quality development, enhance our core competitiveness and continuously defend our advantages in global competition. Under the constantly changing conditions of the new economy, creating an innovative learning team is the fundamental weapon for an enterprise to succeed in the market. We attract talents with a relaxed employment environment and a broad development platform. We hope that more people with lofty ideals will join our team.
Methylamine hydrochloride powder is a white crystalline powder, an organic compound that is soluble in water and ethanol, but insoluble in non-polar solvents such as ether, acetone, and chloroform. It has hygroscopicity and irritability. Incompatible with strong oxidants, its molecular formula is CH5N · HCl, and its molecular weight is 67.52. Easy to deliquescent, has an irritating odor, and is highly corrosive. Almost insoluble in non-polar solvents such as benzene and toluene. It is an organic compound with strong irritant and corrosive properties. In addition to being easily soluble in polar solvents, it also has significant humidity sensitivity and odor characteristics. Special attention should be paid to safety and protective measures during preparation, storage, and use. It is relatively stable in dry air, but is prone to deliquescence and moisture absorption, and is prone to discoloration and degradation when exposed to moisture. Easy to react with strong bases, oxidants, or metal powders. It is an important organic reagent widely used in various fields such as organic synthesis, ionic liquid preparation, metal surface treatment, etc.

|
Chemical Formula |
CH6ClN |
|
Exact Mass |
67.02 |
|
Molecular Weight |
67.52 |
|
m/z |
67.02 (100.0%), 69.02 (32.0%), 68.02 (1.1%) |
|
Elemental Analysis |
C, 17.79; H, 8.96; Cl, 52.51; N, 20.75 |
|
Form |
Powder |
|
Color |
White crystalline |
|
Odor |
less |
|
PH |
5-7 (10g/ L, H2O, 20℃) |
|
Density |
1.43 (rough estimate) |
|
Flash point |
225-230 °C / 15 mm |


Remark: BLOOM TECH(Since 2008), ACHIEVE CHEM-TECH is the subsidiary of us.
The synthesis method of Methylamine Hydrochloride Powder will be introduced in detail:
1. Amination reaction method:
Amination reaction is a commonly used method for preparing Methylamine hydrochloride. The reaction principle is that sodium nitrite reacts with methylamine to generate DMA (N-nitrosomethylamine), and then hydrolyzes DMA in aqueous hydrochloric acid to generate Methylamine hydrochloride. The reaction process is as follows:
CH3NH2 + HNO2 → CH3N(NO)H2
CH3N(NO)H2 + HCl → CH3NH3Cl
In this way, high-purity Methylamine hydrochloride can be obtained.
2. Aldehyde Alkylation Reaction Method:
Formaldehyde and Methylamine hydrochloride react under acidic conditions to obtain methylamino alcohol or methylamine hydrochloride. The reaction process is as follows:
CH2O + CH3NH2·HCl → CH3NHOH·HCl
It should be noted that this reaction needs to be carried out under acidic conditions.
3. Sodium hydroxide hydrolysis method:
Throw methylaminopropanol into a tank catalyzed by copper tape, first heat and distill, then nitrate Methylamine hydrochloride through the solution to generate DMA (N-nitrosomethylamine), and then carry out hydrolysis reaction to obtain high-purity Methylamine hydrochloride:
CH3NH2·HCl + NaNO2 → CH3N(NO)H2·HCl + NaCl + H2O
CH3N(NO)H2·HCl + 2NaOH → CH3NH2 + NaNO2 + NaCl + 2H2O
This reaction is a simple synthetic method, but the product needs multiple crystallization and purification to obtain high-purity products.
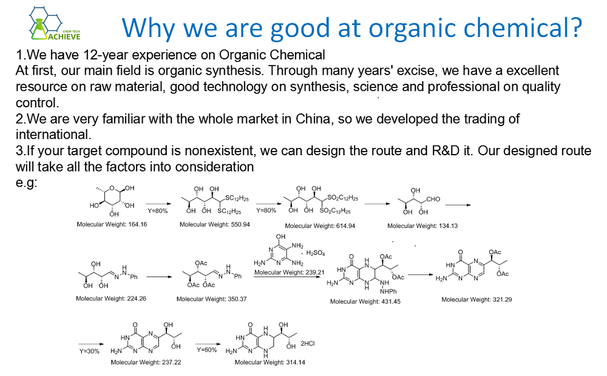
4. Pyrolysis method:
Methylamine hydrochloride can also be prepared by pyrolysis reaction. Methylamine hydrochloride is placed on a hot plate or suspended in molten salt for heating, and methylamine gas and anhydrous HCl can be obtained, and then methylamine gas and water react to generate Methylamine hydrochloride. The reaction is as follows:
CH3NH2 + H2O + HCl → CH3NH3Cl
This method is simple to operate, but requires temperature control and relatively harsh conditions.
5. Acid-base neutralization reaction method:
Methylamine and hydrochloric acid react under acidic conditions to obtain Methylamine hydrochloride. The reaction is as follows:
CH3NH2 + HCl → CH3NH3Cl
This method is simple and easy, but it needs to strictly control the pH of the reaction.
6. Improved amination reaction method:
Dissolve isopropylamine in hydrochloric acid, pass through sodium nitrite solution, react to generate DMA, and then react DMA and formaldehyde in aqueous solution to obtain high-purity Methylamine hydrochloride. The reaction process is as follows:
i-C3H7NH2 + HNO2 → i-C3H7N(NO)H2
i-C3H7N(NO)H2 + HCHO → CH3NH3Cl + i-C3H7OH
This method can improve the reaction yield and reaction selectivity, and the synthesis performance is relatively high.
In short, the above are several main synthetic methods of Methylamine hydrochloride. Different methods have their own advantages and disadvantages. Selecting the appropriate method needs to be considered comprehensively according to the specific situation in order to achieve high efficiency, energy saving, environmental protection and other purposes.

All the uses of Methylamine hydrochloride will be described in detail:
1. Synthesis of ionic liquids:
Ionic liquids, a class of ions with weak hydrogen bonds that remain liquid at room temperature, have attracted much attention for their applications in a variety of fields. Methylamine hydrochloride can be coordinated with some ligands such as sodium hydroxide, acetone and acetaldehyde to form an ionic liquid [CH3NH3]+[X]− (X- is the corresponding hydrochloric acid).
2. Amination reaction:
Methylamine hydrochloride can be used in amination reaction to generate methylamine (CH3NH2). For example, Methylamine hydrochloride can react with sodium nitrite to produce N-nitrosomethylamine (or DMA):
CH3NH2·HCl + NaNO2 → CH3N(NO)H2·HCl + NaCl + H2O
The product DMA can also be used as a precursor for the synthesis of other compounds.
3. Aldehyde Alkylation:
Methylamine hydrochloride can react with aldehydes to generate corresponding methylaminoalcohols or methylamines. For example, formaldehyde hydrate (formaldehyde) can react with Methylamine hydrochloride to produce methylaminoalcohol and water:
CH3NH2·HCl + CH2O → CH3NHOH·HCl
4. Synthesis of α-azophenone derivatives:
Methylamine hydrochloride can be used to synthesize derivatives of α-azobenzophenone, which is an important class of organic compounds and can be used as raw materials in the fields of dyes and photosensitizers. For example, it can react with phenylhydrazine to form phenylhydrazino-α-azophenone:
C6H5NHNH2 + C6H7NOCl → C6H5N=NC6H4NOH + HCl
5. Drug synthesis:
Methylamine hydrochloride can be used to prepare a variety of drugs, such as bifuran, dopamine, and drugs for treating chronic obstructive pulmonary disease. The most common of these is the preparation of Phenylephrine hydrochloride, a compound often used as a vasoconstrictor and a topical decongestant to relieve nasal congestion caused by allergies.
6. Other purposes:
Methylamine Hydrochloride Powder can also be used as a component of corrosion inhibitor and buffer in daily chemicals, food and cosmetics to maintain the stability and quality of these products. In addition, it can also be used in metal surface treatment to improve its corrosion resistance.
In conclusion, Methylamine hydrochloride, as an organic synthesis reagent with various applications, is widely used in ionic liquids, amination reactions, aldehyde alkylation reactions, synthesis of α-azophenone derivatives, drug synthesis and other fields .
What are the precautions?
Here are some important safety precautions when using Methylamine Hydrochloride Powder:
1.Personal protection:
- Appropriate protective gloves and clothing should be worn during operation to prevent skin contact.
- Safety glasses or goggles should be worn to protect the eyes from irritation or injury.
- In environments where dust may be generated, respiratory protective equipment such as P95 (USA) or P1 (EU) particulate respirators should be used.
2.Operational handling:
- Avoid contact with skin and eyes, and do not inhale dust.
- Suitable exhaust equipment should be provided in areas where dust is generated.
- Operators should receive specialized training and strictly follow operating procedures.
- Keep away from sources of fire and heat, and smoking is strictly prohibited in the workplace.
3.Storage conditions:
- It should be stored in a dry, cool, and well ventilated place.
- Keep the container tightly sealed to avoid moisture absorption.
- Store in a cool and shady place, and the storage temperature should not exceed 37 ° C.
4.Emergency measures:
- After skin contact, it should be immediately washed with soap and plenty of water.
- After eye contact, rinse thoroughly with plenty of water for at least 15 minutes and seek medical assistance.
- After inhalation, move the patient to fresh air. If breathing stops, perform artificial respiration and seek medical assistance.
- After ingestion, do not feed anything to unconscious individuals by mouth, rinse with water, and seek medical assistance.
5.Firefighting measures:
- Use water mist, dry powder, foam or carbon dioxide extinguishing agent to extinguish the fire.
- Firefighters must wear gas carrying respirators, full body firefighting suits, and extinguish fires in the upwind direction.
6.Emergency response to leakage:
- When there is a leak, use personal protective equipment to avoid dust generation and ensure adequate ventilation.
- Do not let the product enter the sewer.
- The collection and removal methods of leaked chemicals, as well as the disposal materials used, should avoid generating dust. They should be swept and shoveled off, and placed in suitable sealed containers for disposal.
7.Disposal of waste:
- Waste chemicals should be recycled as much as possible. If they cannot be recycled, incineration methods should be used for disposal.
- Do not dispose of this product by discharging it into the sewer.
8.Transportation information:
According to transportation regulations, methylamine hydrochloride is not classified as a hazardous material.
We love an incredibly fantastic standing amid our consumers for China Factory Methylamine Hydrochloride CAS 593-51-1 Powder Supplier 593 51 1. We warmly welcome your patronage and will serve our clients both at home and abroad with products of superior quality and excellent service geared to the trend of further development as always. Our customers mainly distributed in the North America, Africa and Eastern Europe.

