Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of 1-methylpiperazine cas 109-01-3 in China. Welcome to wholesale bulk high quality 1-methylpiperazine cas 109-01-3 for sale here from our factory. Good service and reasonable price are available.
1-Methylpiperazine, chemical formula C5H12N2, CAS 109-01-3, Colorless liquid, easily soluble in water, ether, ethanol, soluble in any proportion in water, methanol, etc. The aqueous solution is weakly alkaline. In the pharmaceutical industry, the antibiotic drug methylphenidate and the antipsychotic drug triflurazine are synthesized by methylation reaction of piperazine hexahydrate, which are intermediates of organic synthesis. As a chemical with special properties, it has shown broad application prospects in the field of membrane separation technology. By acting as a chelating agent, additive, and participating in the separation and purification of specific substances, the separation efficiency and purity of the membrane can be significantly improved. However, its toxicity, cost, and technical challenges also limit its large-scale application.

|
Chemical Formula |
C5H12N2 |
|
Exact Mass |
100 |
|
Molecular Weight |
100 |
|
m/z |
100 (100.0%), 101 (5.4%) |
|
Elemental Analysis |
C, 59.96; H, 12.08; N, 27.97 |
|
|
|

The application of 1-Methylpiperazine in membrane separation technology is mainly due to its unique physical and chemical properties, especially its dual characteristics of hydrophilicity and lipophilicity, which enable it to play an important role in membrane separation processes. Membrane separation technology is an efficient and energy-saving separation technology widely used in various fields such as water treatment, gas separation, food processing, pharmaceutical manufacturing, etc. The core lies in utilizing the selective permeability of the membrane to achieve effective separation between different components. As a chemical with special properties, its application in membrane separation technology is gradually receiving attention.
Basic properties
The chemical formula is C5H12N2, which is a colorless to pale yellow liquid with a pungent odor. It is soluble in water and various organic solvents, with good solubility and stability. More importantly, it has the dual characteristics of hydrophilicity and lipophilicity, which enables it to play a unique role in membrane separation processes.
Application in membrane separation technology

1. As a chelating agent to improve separation efficiency
During membrane separation, it can act as a chelating agent to form stable complexes with the substance to be separated. The permeability of this complex on the membrane is different from that of the original substance, thus achieving effective separation. For example, when treating wastewater containing heavy metal ions, complexes can be formed with heavy metal ions, and then separated from the wastewater through the selective permeability of the membrane, achieving the goal of purifying water quality.
2. As an additive to improve membrane performance
It can also be added as an additive to membrane materials to improve the performance of the membrane. For example, when preparing reverse osmosis membranes, adding an appropriate amount can improve the membrane's anti fouling performance and permeate flux. This is because it can interact with certain functional groups in the membrane material, forming a more compact membrane structure, thereby reducing the adsorption and blockage of pollutants.


3. Promote the improvement of hydrophilicity on the membrane surface
For certain hydrophobic membrane materials, their hydrophilic properties can promote the improvement of membrane surface hydrophilicity. This helps to reduce membrane fouling and blockage during the separation process, improving membrane separation efficiency and stability. For example, when treating wastewater containing oil or protein, hydrophilic membrane surfaces can more effectively repel these pollutants, thereby achieving better separation effects.
4. Participate in the separation and purification of specific substances
It can also participate in the separation and purification process of specific substances. For example, in the field of pharmaceutical manufacturing, the separation and purification of certain drugs or their intermediates require specific membrane separation techniques. It can serve as an auxiliary agent in these processes, improving separation efficiency and purity through its interaction with the substance to be separated.


5. Development of gas separation membranes
In the field of gas separation, it has also demonstrated potential application value. Due to its unique chemical structure, it can form stable complexes with certain gas molecules, thereby achieving effective separation between gas molecules. This provides the possibility for developing new and efficient gas separation membranes.
Advantages and Challenges in Membrane Separation Technology
Advantage
(1) Multifunctionality:
It has the dual characteristics of hydrophilicity and lipophilicity, allowing it to play multiple roles in membrane separation processes.
(2) Efficiency:
As a chelating agent and additive, it can significantly improve the separation efficiency and purity of the membrane.
(3) Stability:
It has good chemical stability and can maintain its performance unchanged under various conditions.
Challenge
(1) Toxicity issue:
It is irritating to the eyes, skin, and upper respiratory tract, so safety precautions should be taken during use. This increases the complexity of its application in membrane separation technology.
(2) Cost issue:
Although membrane separation technology has potential application value, its relatively high cost may limit its large-scale application.
(3) Technical challenge:
Further research and exploration are needed to effectively apply N-methylpiperazine in membrane separation technology. For example, in-depth research is needed on how to optimize its addition amount and how to combine it with other membrane materials.

The invention relates to a chemical synthesis method of chemical products, in particular to the synthesis process of 1-methylpiperazine using piperazine, formaldehyde and hydrogen as raw materials. Technical background N-methylpiperazine is one of the derivatives of piperazine and an important fine chemical product. It can be used in medicine to synthesize antibacterial drugs such as ofloxacin, levofloxacin, fleroxacin and other psychoactive drugs such as clozapine and olanzapine; It is also widely used in rubber, plastics and other polymer chemical industries.
At present, the product is still in the stage of development in China. There are two main methods of synthesis developed in China according to the literature:
1. The piperazine and hydrochloric acid are first reacted in anhydrous ethanol to produce piperazine hydrochloride. The piperazine hydrochloride is separated by evaporation of ethanol, and then reacted with formaldehyde and formic acid to obtain N-methylpiperazine hydrochloride. Then the product is neutralized with sodium hydroxide. After the separation of sodium chloride, the product is distilled.
The process of this method is long, and the highly corrosive hydrochloric acid and formic acid are used in the production. The pollution is serious, the operating environment is poor, and the yield is only 49%.
2. Using methanol as the methylation reagent and solvent, piperazine and methanol are vaporized and then reacted through the fixed bed catalyst layer. Some raw materials generate N-methylpiperazine. The high-temperature gaseous reaction mixture is condensed and collected, and then rectified to obtain the product. The reaction temperature of this method is as high as 300 ° C, the energy consumption is large, and the single-pass conversion rate can only reach 50%, there are many by-products, and the product purity is low; The fixed-layer catalyst used in this production method is not easy to be replaced. As the catalyst activity decreases, the conversion rate becomes lower and lower, and the energy consumption becomes higher and higher. At a certain time, it can only be replaced completely. The replacement cost is very high, and the production cost is difficult to reduce.
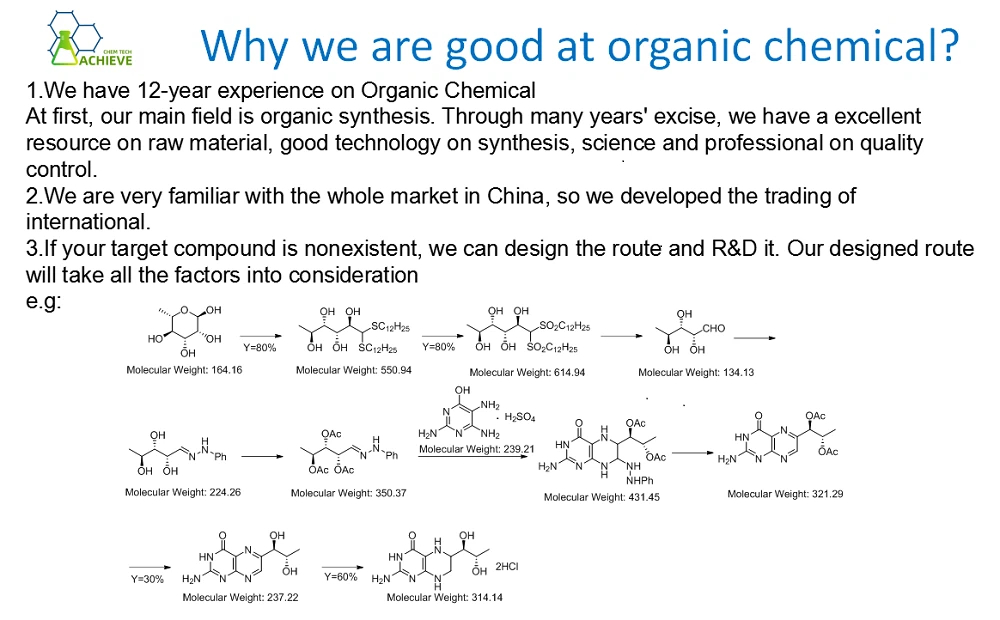
The purpose of this method is to provide a synthetic method of 1-methylpiperazne which is environmentally friendly, high conversion of raw materials, good product selectivity, simple process and suitable for industrialization.
The invention comprises the following steps:
1) The condensation reaction takes methanol as solvent, piperazine and formaldehyde are fed in a molar ratio of 1:0.81.6, and the condensation reaction is carried out in the reactor;
2) After condensation, add catalyst into the same reactor, and replace it with nitrogen and hydrogen. After that, the hydrogen pressure rises to 16Mpa, control the material temperature in the reactor to 70100 ° C, and keep stirring until the hydrogenation reaction is over; The amount of catalyst added is 412% of the mass of piperazine;
3) Cool the material to normal temperature, stop mixing, discharge and filter, and recover the catalyst;
4) Distill the filtrate, recover methanol and unreacted piperazine, and then collect 137 "C fraction to obtain N-methylpiperazine. The single-pass yield of this method can be as high as 73.83%, which greatly reduces the energy consumption compared with the existing two methods, so the production cost is also greatly reduced.
The current method requires several separation operations, a large number of equipment and a long process flow. All reactions in the invention are completed in one reactor, and the equipment investment and maintenance amount are small, so the process has obvious progressiveness; In addition, the current method 1 produces a large amount of waste water, which has a certain impact on the environment. There are basically no three wastes in this production process; The purity of N-methylpiperazine prepared by the current method can only reach 99.0%, and the purity of the product can reach 99.95% by this method.
A synthesis method of 1-methylpiperazine, which is characterized by comprising the following steps:
1) The condensation reaction takes methanol as solvent, piperazine and formaldehyde are fed at a molar ratio of 1 ∶ 0.8~1.6, and the condensation reaction is carried out in the reactor;
2) After condensation, add catalyst into the same reactor, and replace it with nitrogen and hydrogen. After that, the hydrogen pressure rises to 1~6Mpa, control the temperature of materials in the reactor to 70~100 ℃, and keep stirring until the hydrogenation reaction is over; The amount of catalyst added is 4~12% of the mass of piperazine;
3) Cool the material to normal temperature, stop mixing, discharge and filter, and recover the catalyst;
4) Distill the filtrate, recover methanol and unreacted piperazine, and collect the fraction at 137 ℃ to obtain N-methylpiperazine.
This method has the advantages of short process flow, low equipment investment, high one-way yield and no pollutant discharge.

1-Methylpiperazine, as an important organic compound, has a wide range of applications in fields such as medicine, pesticides, and materials science. This article systematically reviews the discovery process of 1-methylpiperazine, from its early background, chemical structure identification to the development process of industrial production, and deeply analyzes the contributions of key scientists and institutions. Research has found that the discovery of 1-methylpiperazine is a product of the development of organic chemistry in the 19th century, and its structural elucidation and improved synthesis methods have laid a solid foundation for subsequent applications.
The research on piperazine compounds can be traced back to the mid-19th century. In 1849, French chemist Auguste Cahours first isolated piperazine from the degradation products of piperine, marking the beginning of research on this type of heterocyclic compound. In the following decades, chemists began to systematically study the properties and reactions of piperazine and its derivatives.
The discovery of 1-methylpiperazine is closely related to the rapid development of organic chemistry in the late 19th century. In 1887, German chemist Arthur Rudolf Hantzsch first reported the preparation of 1-methylpiperazine while studying the synthesis methods of nitrogen heterocyclic compounds. Hantzsch's method involves reacting N-methylethylenediamine with formaldehyde under acidic conditions, laying the foundation for subsequent research. It is worth noting that at that time, Hantzsch did not fully recognize the structural characteristics of this compound and only described it as "a methylated piperazine like substance".
In the late 19th and early 20th centuries, with the development of organic structure theory, chemists gradually deepened their understanding of 1-methylpiperazine.
In 1895, Swiss chemist Alfred Werner proposed the coordination theory, providing a new perspective for understanding the structure of nitrogen-containing heterocyclic compounds. Under this theoretical framework, the structure of 1-methylpiperazine is more clearly elucidated: it is a six membered nitrogen-containing heterocyclic ring, with a methyl substituent attached to one nitrogen atom.
The early 20th century was the golden age of organic structural chemistry, with various new analytical techniques and theoretical methods constantly emerging. In this context, the chemical structure of 1-methylpiperazine has been more accurately confirmed.
In 1912, British chemist William Henry Perkin Jr. used emerging ultraviolet spectroscopy techniques to study the optical properties of piperazine and its derivatives, providing important evidence for the structural confirmation of 1-methylpiperazine.
In the 1920s, the development of X-ray crystal diffraction technology brought revolutionary breakthroughs to the study of organic molecular structures.
In 1928, German chemist Kathleen Lonsdale first applied X-ray diffraction technology to determine the structure of hexamethylenetetramine, which provided a methodological reference for studying 1-methylpiperazine with similar structures.
In 1935, the resonance theory proposed by American chemist Linus Pauling further explained the delocalization phenomenon of lone pair electrons of nitrogen atoms in 1-methylpiperazine.
The emergence of nuclear magnetic resonance (NMR) technology has brought new breakthroughs to the study of the structure of 1-methylpiperazine. In 1953, American chemist Martin Packard first observed the proton NMR signal of 1-methylpiperazine, which not only confirmed its structure but also provided a new tool for studying its conformational dynamics.
In the 1960s, with the development of 13C NMR technology, scientists were able to more comprehensively analyze the electronic structure and substituent effects of 1-methylpiperazine.
The synthesis method of 1-methylpiperazine has undergone an evolutionary process from laboratory preparation to industrial production. Early synthesis mainly relied on Hantzsch's method, which was prepared by the condensation reaction of N-methylethylenediamine and formaldehyde. Although this method is feasible, the yield is low and there are many by-products.
In the 1930s, German chemist Walter Reppe developed acetylene chemistry, providing a new approach for the synthesis of 1-methylpiperazine. In 1940, Reppe reported a new process for the one-step synthesis of 1-methylpiperazine using acetylene, formaldehyde, and methylamine, greatly improving yield and purity. This method was widely adopted by German chemical companies during World War II.
In the 1950s, with the rise of petrochemicals, synthetic routes using ethylene and propylene as raw materials gradually became mainstream. In 1956, American chemist Herbert C. Brown developed a two-step synthesis method using ethylene oxide and methylamine as raw materials. This process has the advantages of easy availability of raw materials and mild reaction conditions, and has been adopted by many chemical companies.
Modern industrial production mainly adopts catalytic amination process. In 1990, Japanese chemist Ryoji Noyori developed an efficient chiral catalyst, making enantioselective synthesis of 1-methylpiperazine possible. At present, the annual production of 1-methylpiperazine worldwide has exceeded 50000 tons, with major producers including BASF from Germany, Dow Chemical from the United States, and Zhejiang Xinhecheng from China.
Hot Tags: 1-methylpiperazine cas 109-01-3, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, gs 441524 remdesivir, 3 Nitrobenzonitrile, 2 6 Pyridinedicarboxylic acid, BENZENE D6, 2 5 Dihydroxybenzaldehyde, DIMETHYLPHOSPHINE OXIDE






