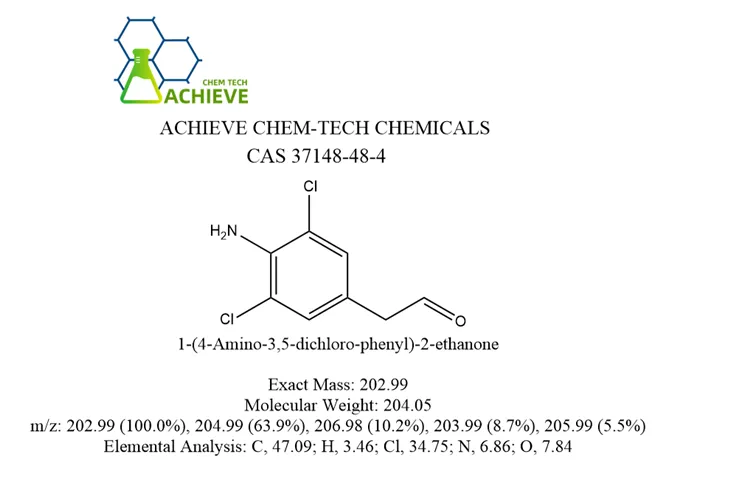
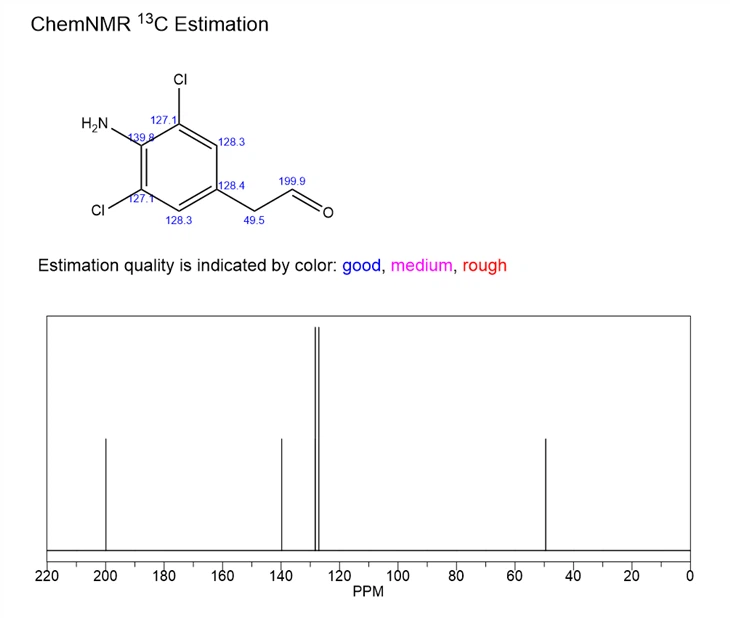
4-Amino-3,5-dichloroacetophenone is an organic compound with a specific molecular formula and molecular weight (C8H7Cl2NO, molecular weight 204.05), which usually exhibits beige or yellowish crystals. Its melting point ranges from 158-166°C, boiling point is about 351.5°C (at 760 mmHg pressure), and its density is about 1.4±0.1 g/cm³. In addition, it is insoluble in cold water, but slightly soluble in hot water, and soluble in organic solvents such as ethanol, ether, benzene and so on. 4-Amino-3,5-dihloroacetophenone is an important intermediate in the synthesis of the cough-relieving and asthma-relieving drug, Kerbasin. It can be used as pharmaceutical intermediates and animal feed additives. In addition, 4-Amino-3,5-dihloroacetophenone may also be used in the synthesis of other fine chemicals, such as dyes and spices.
|
|
|

|
|
|
| Chemical Formula | C8H7Cl2NO |
| Molecular Weight | 204.05 |
| Exact Mass | 202.99 |
| m/z | 202.99 (100.0%), 204.99 (63.9%), 206.98 (10.2%), 203.99 (8.7%), 205.99 (5.5%) |
| Elemental Analysis | C, 47.09; H, 3.46; Cl, 34.75; N, 6.86; O, 7.84 |
| Melting point | 162-166 °C(lit.) |
| Boiling point | 351.5±42.0 °C(Predicted) |
| Density | 1.2748 (rough estimate) |
| Refractive index | 1.5500 (estimate) |
| Storage conditions | Keep in dark place,Inert atmosphere,Room temperature |
| Solubility | DMSO (Slightly), Methanol (Slightly) |
| Form | Solid |
| Acidity Coefficient (pKa) | -1.72±0.10(Predicted) |
| Color | Light Tan to Brown |

We are the supplier of 4-Amino-3,5-Dichloroacetophenone.
Remark: BLOOM TECH(Since 2008), ACHIEVE CHEM-TECH is our subsidiary of us.
The following are the detailed steps and corresponding chemical equations for obtaining 3,5-dichloro-4-aminoacetophenone by chlorinating p-aminoacetophenone:
Reaction of p-aminoacetophenone with hydrochloric acid: C6H4CH2CH2N++HCl → C6H4CH2CH2NH2+HCl
Preparation of p-aminoacetophenone hydrochloride: C6H4CH2CH2OH+HCl → C6H4CH2CH2Cl+H2O
Preparation of imine based compounds: C6H4CH2CH2Cl+CH3OH → C6H4CH(CH3)CH2OH+HCl
Preparation of pyridine ring compounds: C6H4CH(CH3)CH2OH+O2 → C6H4CH(CH3)COOH+H2O
Hydrolysis reaction: C6H4CH(CH3)COOH+H2O → C6H4CH(CH3)COO(-)+H(+)+HCOOH
Washing and drying: C6H4CH(CH3)COO(-)+anhydrous ethanol → C6H4CH(CH3)COOH+(CH3) 2CHOH
Experimental steps
Place para aminoacetophenone (AR) and anhydrous hydrochloric acid (AR) separately in dry and clean beakers for later use.
Preparation of hydrochloric acid solution: Add an appropriate amount of concentrated hydrochloric acid to a beaker containing a small amount of water, stir well, and cool to room temperature.
Preparation of p-aminoacetophenone hydrochloride: Slowly add p-aminoacetophenone to the above hydrochloric acid solution, stir while adding until completely dissolved. Then let the solution stand for a period of time to fully separate the precipitate.
Preparation of imine based compounds: After filtering out the insoluble solution of p-aminoacetophenone hydrochloride obtained in the previous step, add an appropriate amount of solvent such as methanol or ethanol dropwise to convert it into imine based compounds. Pay attention to controlling droplet acceleration to avoid excessive oxidation leading to the generation of by-products.
Preparation of pyridine ring compounds: Slowly add the imine compound obtained in the previous step to a solvent containing pyridine (such as pyridine, pyridine, etc.) under stirring, and maintain the temperature not exceeding 60 ℃. After the dripping is completed, continue stirring for a certain period of time to complete the reaction.
Hydrolysis reaction: Hydrolyze the pyridine ring compound obtained in the previous step into a mixture of pyridine ring compounds and pyridine ketones. The specific operation is to add the mixture to an appropriate amount of water, heat it to around 80 ℃, and keep stirring for a certain period of time until all pyridine ketones are converted into pyridine.
Washing and drying: Filter out excess water from the product obtained in the previous step, wash it once with anhydrous ethanol, and then place it in a well ventilated area to air dry naturally to obtain the target product 3,5-dichloro-4-aminoacetophenone.

4-Amino-3,5-Dichloroacetophenone (CAS number: 37148-48-4) is an important pharmaceutical intermediate with wide applications in the pharmaceutical field. The following is a detailed exploration of its use in the pharmaceutical field, aiming to comprehensively and deeply analyze its importance and potential value.
As an intermediate of the cough and asthma suppressant ketoconazole
The most well-known use is as an intermediate in the synthesis of cough and asthma relieving drugs, such as Quechuanxin (also known as Chuankening, Chuanshu tablets, etc.). Ketamine is a highly effective new drug mainly used to treat respiratory diseases such as tracheitis and asthma, especially for refractory asthma and elderly asthma.

(1) Pharmacological effects of ketoconazole
Ketamine mainly alleviates asthma symptoms by inhibiting the contraction of bronchial smooth muscle, dilating the bronchi. It can also reduce airway reactivity, decrease the release of inflammatory mediators, and thus alleviate airway inflammation and edema. In addition, ketoconazole also has anti allergic effects, which can inhibit the release of allergic mediators and reduce the occurrence of allergic reactions.
(2) The role in the synthesis of clenbuterol
As a key intermediate, it participates in a series of chemical reactions in the synthesis process of chlortetracycline. These reactions include substitution, addition, condensation, etc., ultimately resulting in the production of chlortetracycline, which has cough and asthma relieving effects. Therefore, its quality and purity have a significant impact on the synthesis and efficacy of chlortetracycline.

Application of other pharmaceutical intermediates
In addition to serving as an intermediate for clenbuterol, it can also be used to synthesize other pharmaceutical intermediates that play important roles in the pharmaceutical manufacturing process.

(1) Synthesize other cough and asthma suppressants
In addition to ketoconazole, it can also be used to synthesize other cough and asthma relieving drugs. These drugs have pharmacological effects similar to clenbuterol, but with different chemical structures and mechanisms of action. By introducing it, more cough and asthma suppressants with unique therapeutic effects and lower side effects can be developed.
(2) Synthesize other types of drugs
It can also be used to synthesize other types of drugs, such as anti-inflammatory drugs, anti-tumor drugs, etc. In the synthesis process of these drugs, as key intermediates, they participate in chemical reactions and ultimately generate drug molecules with specific pharmacological effects.
Application in drug development
It also has potential application value in drug development. Its unique chemical structure and properties make it one of the important candidate compounds in drug development.
(1) As the starting point for new drug development
It can serve as a starting point for new drug development, and through chemical modification and modification, new drugs with novel pharmacological effects and lower side effects can be developed. This new drug may have a wider range of treatments and higher efficacy.
(2) As a model compound for drug screening
It can also serve as a model compound for drug screening. By binding to target proteins or receptors, their efficacy and affinity can be evaluated, providing strong support for new drug development.

Other applications in the field of medicine
In addition to the above applications, it also has other potential application values in the pharmaceutical field.

(1) As an analytical reagent
Can be used as an analytical reagent for chemical analysis and detection. Its unique chemical properties make it a key reagent in certain chemical reactions, helping researchers to more accurately understand and analyze the properties of substances.
(2) As a research subject in drug metabolism
It can also be used as a research object for drug metabolism. By studying its metabolic processes in the body, we can understand the absorption, distribution, metabolism, and excretion of drugs in the body, providing strong support for new drug development.
It has wide application value in the field of medicine. As a key intermediate of the cough and asthma suppressant ketoconazole, it also plays an important role in new drug development, drug metabolism research, and other aspects. In the future, with the continuous advancement of pharmaceutical technology and the deepening of new drug research and development, the application of 4-Amino-3,5-Dichloroacetophenone in the pharmaceutical field will be more extensive. At the same time, it is also necessary to strengthen environmental protection and safety measures to ensure safety and sustainability during production and use.

3,5-dichloro-4-aminoacetophenone (CAS number: 37148-48-4), as an important aromatic ketone compound, has a research and development history dating back to the mid-20th century. It has gone through a critical stage from basic synthesis research to industrial application, gradually becoming an indispensable intermediate in fields such as medicine, pesticides, and materials science.
The synthesis initially originated from the research demand for chlorinated aromatic ketone compounds. Early synthesis methods were mainly based on electrophilic substitution reactions, by introducing chlorine atoms and amino groups on the benzene ring, followed by the introduction of acetyl groups. For example, in the 1950s, researchers used 4-aminoacetophenone as a raw material and introduced chlorine gas into a solution of glacial acetic acid for chlorination reaction. By controlling the reaction conditions (such as temperature and chlorine gas injection rate), 3,5-dichloro-4-aminoacetophenone was successfully prepared. The yield of this method is about 50%, although the efficiency is relatively low, it lays the foundation for subsequent research.
In the 1970s, with the advancement of organic synthesis technology, researchers began to explore more efficient synthesis routes. For example, starting from 2,6-dichloroaniline, introducing acetyl groups through acylation reaction, and then purifying by recrystallization, high-purity products can be obtained. This method not only simplifies the operation steps, but also significantly improves the yield (up to 80% or more), becoming an important reference for industrial production.
In the 1980s, with the increasing demand for this substance in industries such as medicine and pesticides, its industrial production became a research focus. Domestic chemical enterprises, such as Zhongshan Dixin Chemical Co., Ltd. and Jiangsu Pulesi Biotechnology Co., Ltd., have achieved large-scale production by optimizing reaction conditions and improving equipment design. For example, by using a continuous chlorination reaction device, the amount of chlorine gas introduced can be precisely controlled to reduce the generation of by-products; By using a solvent recovery system, production costs can be reduced and environmental pollution can be minimized.
At the same time, researchers have also developed green synthesis processes. For example, using copper bromide instead of liquid bromine as the bromination reagent avoids the use of highly toxic bromine and improves reaction safety; By replacing traditional reducing agents with catalytic hydrogenation reduction, waste emissions have been reduced. These innovations make their production more environmentally friendly and promote the sustainable development of the industry.
Entering the 21st century, the application fields are constantly expanding. In the field of medicine, as a key intermediate for the synthesis of cough and asthma suppressant ketoconazole, its demand continues to grow. For example, 4-amino-3,5-dichloro-alpha-bromoacetophenone prepared by bromination reaction can further synthesize stable isotope labeled clenbuterol compounds for drug metabolism research.
In the field of materials science, its aromatic ketone structure makes it an important monomer for preparing functional polymer materials. For example, its derivatives can be used to synthesize photoluminescent materials, liquid crystal display materials, etc., and have potential application value in display technology, optical sensing, and other fields.
Hot Tags: 4-Amino-3,5-Dichloroacetophenone CAS 37148-48-4, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 2 6 Pyridinedicarboxylic acid, Methylamine hydrochloride powder, N N N Trimethylethylenediamine, 3 methyl 4 piperidone, 3 Nitrobenzaldehyde 99 , 2 5 Dihydroxybenzaldehyde