Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of n-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline cas 16357-59-8 in China. Welcome to wholesale bulk high quality n-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline cas 16357-59-8 for sale here from our factory. Good service and reasonable price are available.
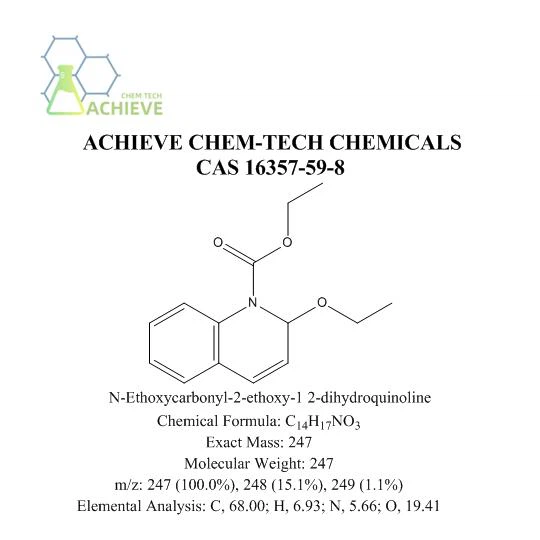
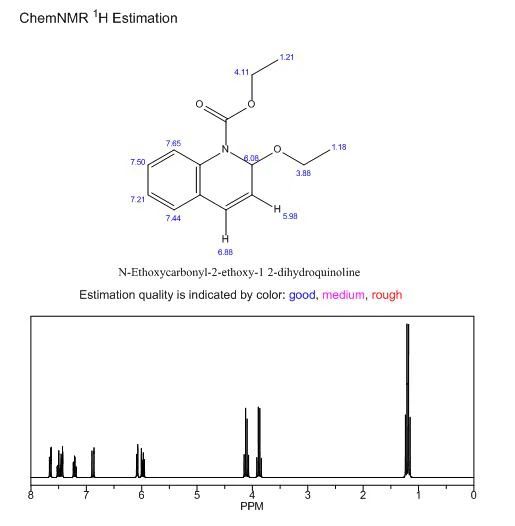
N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline(EEDQ) is an organic compound with the molecular formula C14H17NO3 and CAS 16357-59-8, belonging to quinoline derivatives. Usually a white or slightly yellowish crystalline solid. Organic intermediate, coupling agent in peptide synthesis, classic mixed anhydride method peptide constrictor, mild and effective. Irreversible membrane binding receptor antagonist, peptide synthesis condensation agent, commonly used for amino and carboxyl group condensation.
This appearance feature makes it easy to identify and distinguish in the laboratory. It has a certain solubility in common organic solvents, such as chloroform, methanol, ethanol, etc. This allows it to be synthesized and separated by solution method. The choice of solvent has a significant impact on its solubility. Different solvents may cause significant changes in their solubility, thereby affecting their operation and application in experiments. It has important application value in fields such as chemistry, medicine, and materials science. As an organic compound with unique chemical structure and biological activity, it has shown broad application prospects in various fields such as peptide synthesis, drug development, materials science, biological probes and labeling, and environmental science. With in-depth research on its properties and applications, it is believed that it will have more new uses discovered and applied in the future.

|
Chemical Formula |
C14H17NO3 |
|
Exact Mass |
247 |
|
Molecular Weight |
247 |
|
m/z |
C, 68.00; H, 6.93; N, 5.66; O, 19.41 |
|
Elemental Analysis |
H, 2.79; La, 32.08; N, 9.70; O, 55.42 |
|
|
|

Melting point 62-67 ° C (lit.), Boiling point 125-128 ° C (0.1 mmHg), Density 1.1173 (rough estimate), Refractive index 1.5300 (estimated), Flash point 125-128 ° C / 0.1mm, torage conditions 2-8 ° C, Acidity coefficient (PKA) - 1.27 ± 0.40 (predicted), Morphology crystal powder, Color white to Beige.

N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline (EEDQ), as an important organic compound, has demonstrated its wide application value in multiple fields.
In the field of peptide synthesis, EEDQ is widely used as an efficient condensing agent in the condensation reaction of amino and carboxyl groups. Its mild and effective catalytic effect makes the synthesis process of peptides more efficient and controllable.

Condensation agents in peptide synthesis
Application example:
Synthesis of complex peptides: In the synthesis of complex peptides such as glycocholic acid (GCA), EEDQ serves as a key condensing agent that can precisely control the sequence and structure of the peptides, thereby preparing peptide drugs or materials with specific biological activities.
Optimizing peptide synthesis conditions: By adjusting the dosage and reaction conditions of EEDQ, the peptide synthesis process can be optimized, and the purity and yield of the product can be improved.

EEDQ also has important application value in the field of drug development and research. As an irreversible membrane morphology receptor antagonist, it can be used to develop novel therapeutic drugs or diagnostic reagents.
Drug development and research
Application example:
Research on anti-tumor drugs: By regulating the structure and dosage of EEDQ, its potential mechanism of action in anti-tumor drug development can be explored. For example, EEDQ can bind to specific receptors on the surface of tumor cells, thereby inhibiting their growth and spread.
Anti inflammatory drug development: EEDQ can also serve as a candidate compound for anti-inflammatory drugs. By regulating its pharmacological activity, new drugs with anti-inflammatory effects can be developed for the treatment of inflammatory diseases.
Preparation of diagnostic reagents: By utilizing the binding ability of EEDQ to specific receptors, reagents for diagnosing diseases can be prepared. These reagents can bind to specific molecules in the patient's body, helping doctors accurately determine the type and severity of the disease.

In the field of polymer material modification, EEDQ has also demonstrated its unique advantages. By introducing EEDQ, the physical and chemical properties of polymer materials can be altered, thereby improving their performance.

Polymer material modification
Application example:
Improving heat resistance: When preparing heat-resistant polymer materials, an appropriate amount of N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline can be added. EEDQ can react with certain functional groups in polymer chains to form stable chemical bonds, thereby improving the heat resistance of materials.
Improving mechanical properties: By adjusting the dosage and reaction conditions of EEDQ, the mechanical properties of polymer materials can be improved. For example, the tensile strength and toughness of the material can be increased to make it more durable.
Preparation of functional polymer materials: By utilizing the specific chemical properties of EEDQ, polymer materials with special functions can be prepared. For example, conductive materials, magnetic materials, or optical materials can be prepared.

In the field of organic synthesis, EEDQ can also serve as a catalyst or intermediate for certain chemical reactions, promoting the progress of the reaction or improving the selectivity of the reaction.

Catalysts and intermediates in organic synthesis
Application example:
Catalytic esterification reaction: EEDQ can serve as a catalyst for esterification reactions, promoting esterification reactions between alcohols and acids. This reaction is very common in organic synthesis and can be used to prepare ester compounds.
Synthesis of heterocyclic compounds: By utilizing the specific chemical properties of EEDQ, various heterocyclic compounds can be synthesized. These compounds have wide application value in fields such as drug synthesis and pesticide preparation.
As an intermediate involved in complex organic synthesis: EEDQ can serve as a key intermediate in the reaction during complex organic synthesis processes. Organic compounds with specific structures and properties can be generated by reacting with other compounds.
EEDQ also has certain application value in the fields of wastewater treatment and environmental protection. It can react with certain pollutants in wastewater and convert them into harmless or low toxicity substances.

Wastewater Treatment and Environmental Protection
Application example:
Treating heavy metal ion wastewater: EEDQ can undergo complexation reactions with heavy metal ions in wastewater to remove them from the wastewater. This treatment method has the characteristics of high efficiency and environmental protection, and can be used to treat industrial wastewater containing heavy metal ions.
Degradation of organic pollutants: EEDQ can also react with organic pollutants in wastewater to degrade them into harmless or low toxicity substances. This treatment method can be used to treat wastewater containing organic pollutants, such as printing and dyeing wastewater, pharmaceutical wastewater, etc.


Other application areas
In addition to the aforementioned fields, N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline also has extensive application value in other areas. For example, in the field of electrochemistry, EEDQ can be used as an electrode modification material to improve the performance and stability of electrodes.
In the field of photochemistry, EEDQ can be used as photosensitizers or photocatalysts to participate in photochemical reaction processes; In the biomedical field, EEDQ can also serve as a biomarker or biological probe for biomedical research and diagnosis.


EEDQ, as an important organic compound, has particularly remarkable applications in the field of biomedicine. Especially as an irreversible membrane-bound receptor antagonist, EEDQ has demonstrated unique pharmacological activity and broad application prospects.
The concept and importance of irreversible membrane-bound receptor antagonists

Irreversible membrane-bound receptor antagonists are a special class of drug molecules that can tightly bind to receptors on the cell membrane and make this binding irreversible through the formation of covalent bonds. This characteristic gives irreversible membrane-bound receptor antagonists a unique advantage in drug development. They can occupy receptor sites for a long time, effectively blocking receptor-mediated signaling processes and achieving the goal of treating diseases.
The importance of irreversible membrane-bound receptor antagonists lies in their provision of a novel mechanism of drug action, offering new therapeutic strategies for diseases that are difficult to treat with traditional drugs. In addition, due to their irreversible binding properties, these drugs often have longer half lives and lower effective doses, thereby reducing the frequency of medication and the risk of side effects for patients.

The principle of EEDQ as an irreversible membrane-bound receptor antagonist
The reason why N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline can serve as an irreversible membrane-bound receptor antagonist is mainly attributed to its unique chemical structure and reactivity. The ethoxycarbonyl moiety in EEDQ molecules can undergo covalent reactions with functional groups such as amino or hydroxyl groups on the receptor, forming stable covalent bonds. The formation of this covalent bond enables EEDQ to firmly bind to the receptor and is not easily dissociated or displaced.
In addition, EEDQ also has good membrane penetration and receptor selectivity. It can effectively penetrate the cell membrane, reach the location of the target receptor, and specifically bind to the receptor. This selective binding ensures that EEDQ exerts pharmacological effects while having minimal impact on normal cells and tissues.

Application Examples of EEDQ as Irreversible Membrane Binding Receptor Antagonists
1. Application in neurological diseases
The application of EEDQ is particularly widespread in neurological diseases. For example, in the treatment of Parkinson's disease, EEDQ can act as an irreversible antagonist of dopamine D2 receptors, reducing symptoms in Parkinson's disease patients by blocking the signaling process mediated by dopamine D2 receptors. In addition, EEDQ can also be used to treat neurological disorders such as schizophrenia, by regulating the balance of neurotransmitters and improving patients' cognitive and behavioral disorders.
2. Application in cardiovascular diseases
Cardiovascular disease is one of the leading causes of death worldwide. The application of EEDQ in cardiovascular disease is mainly reflected in the regulation of cardiac ion channels. For example, EEDQ can act as an irreversible antagonist of sodium or calcium ion channels, by inhibiting the activity of ion channels and reducing the excitability of heart cells, thereby preventing and treating cardiovascular diseases such as arrhythmia.
3. Application in tumor treatment
Tumors are complex diseases that involve multiple signaling pathways in their occurrence and development. The application of EEDQ in tumor treatment is mainly reflected in the regulation of tumor related receptors. For example, EEDQ can act as an irreversible antagonist of certain tumor associated receptors, inhibiting the proliferation and invasion ability of tumor cells by blocking receptor-mediated signaling processes, thereby achieving the goal of treating tumors. In addition, EEDQ can also be used in combination with other anti-tumor drugs to improve treatment efficacy and reduce the risk of side effects.
4. Application in inflammatory diseases
Inflammatory diseases are a common type of chronic illness, including arthritis, asthma, inflammatory bowel disease, etc. The application of EEDQ in inflammatory diseases is mainly reflected in the regulation of inflammatory mediators. For example, EEDQ can act as an irreversible antagonist of certain inflammatory mediator receptors, inhibiting the release and activity of inflammatory mediators by blocking receptor-mediated signaling processes, thereby reducing inflammatory symptoms and promoting inflammation resolution.
5. Application in drug development
In addition to being directly used for treating diseases, N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline also plays an important role in drug development. For example, in drug screening and efficacy evaluation, EEDQ can be used as a tool drug to assess the affinity and selectivity of new drug molecules towards specific receptors. In addition, EEDQ can also serve as a probe for drug metabolism and pharmacokinetics research, used to investigate the metabolic pathways and excretion mechanisms of new drug molecules in vivo.

In a standard laboratory procedure, we first prepare a clean 100 milliliter round bottom flask and then precisely add the specified amount of quinoline - an organic compound with 97% high purity and a mass of 1.26 grams, equivalent to 9.5 millimoles. Next, we added 3.2 milliliters of ethanol, with a substance amount of 47 millimoles, as one of the solvents, and 0.4 milliliters of water, with a substance amount of 21 millimoles, to adjust the polarity of the reaction system. In addition, 1.1 grams of sodium bicarbonate (NaHCO3) was added in an amount of 13 millimoles as a base in the reaction to neutralize potential acidic byproducts.
Quickly cool the obtained pale yellow mixed solution to 0 degrees Celsius to ensure that the subsequent reaction can proceed smoothly under low temperature conditions. While maintaining this temperature, we used a dropping funnel to slowly and evenly add newly distilled ethyl chloroformate (1.08 g, exactly 10 mmol) dropwise to the reaction mixture over a period of 20 minutes. This step requires careful operation to avoid local overheating or excessive reaction.
Let the reaction mixture continue to stir at 0 degrees Celsius for 1 hour to ensure that all reactants are fully in contact and the expected chemical reaction occurs. Subsequently, in order to quickly stop the reaction, we added 10 milliliters of water to the system, a process called "quenching".
Using a separatory funnel, we extracted the aqueous solution three times with 15 milliliters of dichloromethane to extract the organic phase containing the target product. After each extraction, collect the organic layer and finally merge all the organic phases. To remove any residual moisture, we dried the combined organic layers using anhydrous sodium sulfate (Na2SO4).
The organic solution is concentrated under reduced pressure using a rotary evaporator until an amorphous sol like substance is obtained. In order to further improve the purity of the product and obtain a more suitable form for further research, we dissolved this sol in an appropriate amount of hexane and obtained pure the product crystals through recrystallization. This product not only has high purity, but also has a clear structure, providing a solid foundation for subsequent research and applications.
Unexpected Discovery of Pharmacological Activities (1968–1970s)
The discovery of EEDQ originated from technological breakthroughs in peptide synthesis chemistry in the 1960s. In 1968, chemist Bernard Belleau and his team member George Malek from McGill University, Canada, published pioneering research in the Journal of the American Chemical Society (J. Am. Chem. Soc.), in which they successfully synthesized and reported EEDQ (CAS: 16357-59-8) for the first time.At that time, peptide synthesis relied on coupling agents such as DCC, which suffered from drawbacks including easy racemization, numerous side reactions, and complicated work-up procedures. Starting from quinoline, Belleau's team precisely constructed a stable dihydroquinoline heterocyclic structure through ethoxylation and carbamation reactions, and named the compound N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline, abbreviated as EEDQ.This reagent required no additional base catalysis, efficiently activated carboxyl groups, and selectively formed amide bonds. Its only byproducts were quinoline and ethanol, which could be easily removed by acid washing. EEDQ quickly became a revolutionary coupling agent in the field of peptide synthesis.
Patent Portfolio and Early Industrialization (1968–1969)
Following the successful synthesis of EEDQ, chemist Weinberg from Bristol-Myers (the predecessor of Bristol-Myers Squibb) rapidly followed up. Between 1968 and 1969, two U.S. patents (US 3389142 and US 3452140) were successively granted, covering the preparation process of EEDQ and its application in peptide synthesis, laying the foundation for its intellectual property protection.In the same period, Belleau's team further validated its performance: EEDQ reacted mildly at room temperature with extremely low racemization, was compatible with amino acid protection and fragment condensation, and was especially suitable for the synthesis of complex peptides and small-molecule pharmaceutical intermediates. Although the early synthetic route was cumbersome and costly, its advantages of high selectivity, easy purification, and low side reactions allowed it to rapidly replace some traditional coupling agents and enter research pipelines of chemical laboratories and pharmaceutical companies worldwide.
Materials of Metric Roller Chain Sprocket
When first synthesized in 1968, EEDQ was only designed as a peptide coupling reagent. However, in the same year, Belleau's team unexpectedly discovered its irreversible α-adrenergic receptor antagonistic activity and central nervous system depressant effects, opening up new pharmacological value.From 1970 to 1973, academic research further confirmed that EEDQ could irreversibly inactivate various membrane receptors including muscarinic cholinergic receptors and 5-hydroxytryptamine receptors, making it a unique tool drug in neuroscience research. Its mechanism of action involves selective alkylation of receptor active sites, with long-lasting efficacy and high specificity. It has been widely used to establish receptor-deficient animal models, study neural signaling pathways, and screen drugs for psychosis and hypertension.
Academic Advancement and Global Popularization (1970–1990s)
In 1971, Japanese chemists Yajima and Kawatani optimized the synthetic process of EEDQ, improving its purity and yield and promoting its application in the pharmaceutical and chemical industries in Asia.During the 1970s and 1980s, researchers worldwide continued to expand its applications. Beyond peptide synthesis, EEDQ was also employed in organic reactions such as selective hydroxyl protection, ester synthesis, and heterocyclic compound construction, establishing itself as a versatile carboxyl-activating reagent.In 1983, Hamblin and Creese demonstrated its ability to block central dopamine receptors, further consolidating its key role in neuropharmacological research. It was included in the standard reagent catalogs of universities and pharmaceutical companies globally.
Modern Applications and Enduring Value (2000–Present)
Entering the 21st century, EEDQ maintains an irreplaceable position in the synthesis of peptide drugs, antibody-drug conjugates (ADCs), and biomaterials due to its high stability, strong compatibility, and favorable cost-performance ratio.Despite competition from new coupling agents such as HATU and PyBOP, its advantages of minimal racemization, simple work-up, and low cost still make it the first choice for small- to medium-scale synthesis and basic research.Meanwhile, as an irreversible receptor antagonist, it continues to be used in brain science and precision medicine research, supporting investigations into the mechanisms of neurological diseases and the development of new drugs.Since its birth in 1968, EEDQ has stood the test of half a century, remaining a classic reagent in organic chemistry and pharmacology, and witnessing the evolution of peptide synthesis and neuroscience.

EEDQ is a versatile reagent with broad applications in organic synthesis, pharmaceutical chemistry, and material science. Its ability to form amide bonds efficiently, dehydrate alcohols, and modify biomolecules makes it indispensable in modern chemistry. While traditional methods rely on organic solvents, emerging green chemistry approaches promise to enhance its sustainability. Future research will focus on expanding its utility in bioconjugation, prodrug design, and advanced material synthesis, solidifying EEDQ's role as a cornerstone of synthetic chemistry.

Hot Tags: n-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline cas 16357-59-8, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, myrcene powder, 1 4 Phenylenebisboronic Acid CAS 4612 26 4, squaric acid treatment, hydroxyethyl urea powder, 4 4 Dimethylbiphenyl CAS 613 33 2, 2 Phenylacetamide CAS 103 81 1