Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of palonosetron hcl cas 135729-62-3 in China. Welcome to wholesale bulk high quality palonosetron hcl cas 135729-62-3 for sale here from our factory. Good service and reasonable price are available.
Palonosetron hcl is a highly selective and potent second-generation 5-HT₃ receptor antagonist. Its unique isoquinoline ring system and thiazole skeleton in the molecular structure give it a kinetic advantage over the first-generation drugs. This drug binds to the 5-HT₃ receptor with high affinity through a allosteric regulatory mechanism, not only blocking the excitation transmission of peripheral vagal nerve terminals, but more importantly, it can directly inhibit the receptor activation of the vomiting center in the medulla oblongata, thereby achieving dual antiemetic control at both the central and peripheral levels. Its most significant characteristic is the negative synergy effect - when the drug binds to a receptor subunit, it induces a change in the entire receptor conformation, significantly reducing the binding ability of other agonists to adjacent sites. This "occupation locks out" mechanism ensures a persistent and stable antiemetic effect. Compared to short-acting drugs such as ondansetron, palonosetron has an elimination half-life of approximately 40 hours in the body, and it has potential cross-inhibition effects on the NK-1 neural pathway mediated by P substance. This makes its preventive effect on chemotherapy-induced delayed vomiting particularly prominent. A single 0.25mg intravenous administration can continuously exert blocking effects throughout the risk period (acute and delayed phases), and this excellent integration of pharmacodynamics and pharmacokinetics establishes its irreplaceable cornerstone position in highly emetogenic chemotherapy regimens.

|
|
|
|
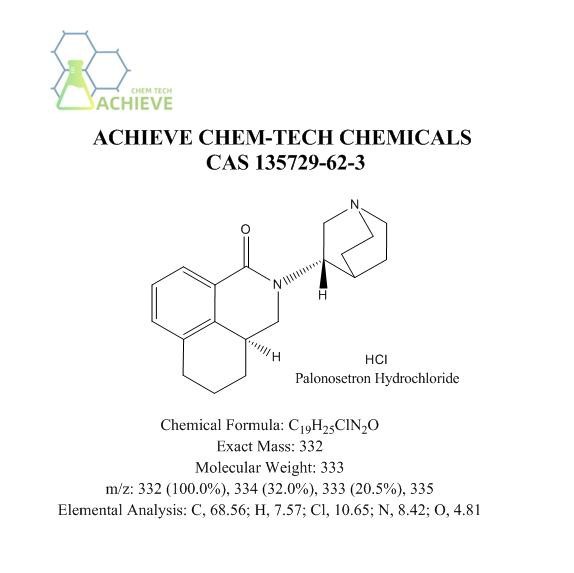
Chemical Formula |
C19H25ClN2O |
|
Exact Mass |
332 |
|
Molecular Weight |
333 |
|
m/z |
332 (100.0%), 334 (32.0%), 333 (20.5%), 335 |
|
Elemental Analysis |
C, 68.56; H, 7.57; Cl, 10.65; N, 8.42; O, 4.81 |

Palonosetron HCl, as a highly specific 5-HT3 (5-hydroxytryptamine 3) receptor antagonist, has demonstrated excellent efficacy in the medical field, particularly in the management of chemotherapy-induced nausea and vomiting (CINV). Its molecular structure is ingeniously designed to tightly bind and block 5-HT3 receptors. This strong affinity ensures that the drug can effectively inhibit the vomiting reflex mediated by 5-HT (5-hydroxytryptamine), while avoiding unnecessary interactions with non target receptors. Therefore, it has almost no affinity for other receptor types or only shows extremely low affinity, greatly reducing the risk of side effects.
5-HT3 receptors play a crucial role in the human body, mainly distributed in key areas of the nervous system responsible for regulating the vomiting reflex, including the vomiting center in the medulla oblongata and the chemoreceptor zone, as well as the peripheral vagus nerve endings. These areas are crucial for maintaining normal physiological functions, but they are also the main sites of action for chemotherapy-induced nausea and vomiting.


When chemotherapy drugs enter the body, they stimulate chromaffin cells in the small intestine to release large amounts of 5-HT. These released 5-HTs subsequently bind to 5-HT3 receptors located on the afferent fibers of the vagus nerve, activating a series of complex neurophysiological responses, ultimately leading to the occurrence of the vomiting reflex. This process not only brings great discomfort to patients, but may also affect the compliance and efficacy of chemotherapy.
Palonosetron hydrochloride, through its potent and selective 5-HT3 receptor antagonism, can effectively prevent the binding of 5-HT to these receptors, thereby inhibiting the initiation and maintenance of the vomiting reflex, providing effective antiemetic protection for patients undergoing chemotherapy. In addition, due to its low side effects, palonosetron hydrochloride can also help improve patients' quality of life, enabling them to complete chemotherapy courses more smoothly.

Synthesis methods
Route 1
It is a brief introduction to the synthesis of palonosetron hydrochloride from 1,8-naphthalene dicarboxylic anhydride as the starting material.
1,8-naphthalene dicarboxylic anhydride+H2 → 1,8-naphthalene dicarboxylic acid
1,8-naphthalene dicarboxylic anhydride first reacts with dilute hydrochloric acid in a mixed solution of ethanol water (v/v=3:2). After adding an appropriate amount of palladium (Pd/C) catalyst and dispersing it on carbon, a hydrogenation reaction is carried out to obtain the corresponding 1,8-naphthalene dicarboxylic acid (NPA). This step is mainly aimed at reducing the carbonyl group of anhydrides to the carbonyl group of aldehydes.
C12H8O4 + NH3 → 1,8-naphthylacetic acid
Dissolve the NPA obtained in step 1 in a mixed solution of ammonia water/isopropanol and react under high pressure to obtain the corresponding amino acid (NAP). The control of reaction temperature and time has a significant impact on the yield and product system.
1,8-naphthylacetic acid+SOCl2 → 1,8-naphthylacetyl chloride
React the NAP obtained in step 2 with chlorine sulfite (SOCl2) to obtain the corresponding sulfonyl chloride complex. During this process, water should be avoided from entering the reaction system to avoid affecting the reaction.
1,8-naphthylacetyl chloride+C6H5CH=CH2 → phenylmethylene N-sulfonyl-N - (1-naphthyl) formamide
The sulfonyl chloride obtained in step 3 is condensed with phenylethylene to obtain the corresponding phenylmethylene N-sulfonyl-N - (1-naphthyl) formamide compound.
Phenylmethylene N-sulfonyl-N - (1-naphthyl) formamide+NaOH → C19H25ClN2O
The compound obtained in step 4 is reacted with sodium hydroxide (NaOH) to undergo nucleophilic addition and internal acylation reactions to obtain palonosetron hydrochloride.
Route 2
It is to use tetrahydronaphthalene as the starting material, and the specific steps are as follows:
C10H12+O2 → C10H10O+H2O
Firstly, tetrahydronaphthalene undergoes an oxidation reaction with oxygen. Commonly used oxidants include ammonium persulfate (NH4) 2S2O8 or potassium persulfate K2S2O8. This reaction will generate the corresponding tetrahydronaphthalene ketone.
C10H10O+SOCl2 → tetrahydronaphthalenesulfonyl chloride+HCl
React the tetrahydronaphthalene ketone obtained in step 1 with chlorine sulfite (SOCl2), and perform sulfonation reaction under appropriate solvents and conditions to obtain the corresponding tetrahydronaphthalene ketone sulfonyl chloride complex.
Tetrahydronaphthone sulfonyl chloride+R-NH2 → Tetrahydronaphthone sulfonamide R+HCl
Perform a condensation reaction between the tetrahydronaphthalenesulfonyl chloride obtained in step 2 and the corresponding amine compound. In this reaction, amine compounds can be part of the precursor molecules of Palonosetron or compounds with similar structures. Condensation reactions are usually carried out under alkaline conditions, and commonly used bases include sodium carbonate (Na2CO3) or sodium bicarbonate (NaHCO3).
Tetrahydronaphthalenesulfonamide-R+NaOH → C19H25ClN2O+Na2SO3+H2O
Expose the condensation product obtained in step 3 to appropriate alkaline conditions, such as using sodium hydroxide (NaOH) or sodium bicarbonate (NaHCO3) solution for cyclization reaction. This cyclization reaction will lead to nucleophilic addition and endoacylation reactions within the molecule, ultimately forming Palonosetron HCl.
Toxicological research
Carcinogenicity
In a 104 week carcinogenicity study of CD-1 mice, animals were orally treated with palonosetron at doses of 10, 30, and 60 mg/kg/day, and the results showed that palonosetron was non carcinogenic. The systemic exposure level (plasma AUC) of palonosetron generated by the highest experimental dose is 150-289 times the recommended exposure level for humans after intravenous administration of 0.25mg palonosetron (AUC=29.8ng. h/ml). In a 104 week carcinogenicity study in SD rats, male and female rats were orally administered palonosetron at doses of 15, 30, and 60 mg/kg/day, and 15, 45, and 90 mg/kg/day, respectively. The highest dose produced a palonosetron systemic exposure (plasma AUC) that was 137-308 times the recommended human dose exposure. The use of palonosetron increased the incidence rate of benign adrenal pheochromocytoma and mixed benign and malignant pheochromocytoma in male rats, the incidence rate of pancreatic islet cell adenoma, mixed adenoma and sarcoma, and pituitary adenoma in male rats, while in female rats, it led to the occurrence of hepatic cellular adenoma and sarcoma, and the incidence rate of thyroid C-cell adenoma, mixed adenoma, and sarcoma.
Mutability
Bacterial Ames test, Chinese hamster ovary cell mutagenicity test, in vitro liver cell disordered DNA synthesis (UDS) test, or mouse micronucleus test showed that palonosetron has no mutagenic toxicity. However, palonosetron has chromosomal aberrations in Chinese hamster ovary cells.
Reproductive toxicity
When orally administered to rats at a dose of 60mg/kg (approximately 1894 times the recommended intravenous dose for humans based on body surface area), it has no effect on the fertility and reproductive ability of male and female rats.
Palonosetron HCl, belongs to the class of serotonin 5-HT3 receptor antagonists. It exhibits high efficacy and affinity for the 5-HT3 receptor, with an IC50 value of 0.24 nM for 5-HT3A and a pKi value of 10.4. This medication demonstrates a slow dissociation rate, effectively inhibiting 5-HT-induced enhancement of cellular responses to substance P both in vitro and in vivo.
It is administered as an injection, typically in a dose of 0.25 mg for adults to prevent CINV, given intravenously over 30 seconds, approximately 30 minutes before the start of chemotherapy. For postoperative nausea and vomiting, a lower dose of 0.075 mg is administered intravenously over 10 seconds immediately before the induction of anesthesia.
The storage should be at a controlled temperature of 20-25°C (68-77°F), with short-term travel allowing temperatures of 15-30°C (59-86°F), while preventing freezing and protecting from light.
In summary, Palonosetron HCl is an effective and crucial medication for managing nausea and vomiting associated with chemotherapy, improving patients' quality of life during treatment.

Palonosetron hydrochloride is a highly selective and affinity 5-HT3 receptor antagonist, belonging to the second-generation antiemetic drugs. Since its emergence in the early 21st century, it has become an important drug for preventing chemotherapy-induced nausea and vomiting (CINV) and postoperative nausea and vomiting (PONV). Chemotherapy induced nausea and vomiting (CINV) has long been one of the main side effects in cancer treatment, seriously affecting patients' quality of life and treatment compliance. In the late 1980s to early 1990s, the successful development of first generation 5-HT3 receptor antagonists such as Ondansetron, Granisetron, and Dolasetron marked an important breakthrough in the treatment of CINV. However, clinical practice has gradually exposed the obvious limitations of the first generation drugs: their half-life is short (usually 3-9 hours), their effectiveness in treating delayed CINV (nausea and vomiting that occurs within 24 hours to several days after chemotherapy) is limited, and some patients may still experience breakthrough vomiting symptoms. These shortcomings have prompted the pharmaceutical industry to seek new 5-HT3 receptor antagonists with longer lasting efficacy and higher receptor affinity. In this context, Helsinn Healthcare (now Helsinn Group) in Switzerland has launched a research and development program for a new antiemetic drug. The company has extensive experience in supportive tumor therapy and recognizes the clinical value of developing a new generation of 5-HT3 receptor antagonists with longer duration of action and stronger efficacy. This strategic decision directly led to Palonosetron's discovery.
Hot Tags: palonosetron hcl cas 135729-62-3, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale