Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of noradrenaline acid tartrate cas 69815-49-2 in China. Welcome to wholesale bulk high quality noradrenaline acid tartrate cas 69815-49-2 for sale here from our factory. Good service and reasonable price are available.
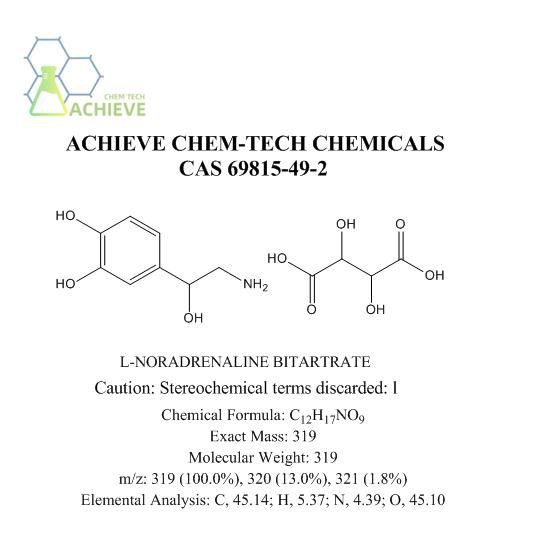
Noradrenaline acid tartrate(L-4-(2-Amino-1-hydroxyethyl)-1,2-benzenediol bitartrate), chemical formula C8H11NO3.C4H6O6.H2O, is a white or almost white crystalline powder. odorless, bitter, light, air perishable, soluble in water, slightly soluble in ethanol, insoluble in chloroform or ether. L-4-(2-Amino-1-hydroxyethyl)-1,2-benzenediol tartrate, chemical formula C8H11NO3 • C4H6O6) is a drug commonly known as bisoprolol free base tartrate. It is a white or almost white crystalline powder, commonly in the form of amorphous or crystalline solids. It has good solubility in water and can dissolve quickly. It can also be dissolved in organic solvents such as ethanol, methanol, and ether. Relatively stable within a certain temperature range, but decomposition may occur under high temperature conditions. The pH value depends on the concentration of its solution and the dissolution medium. Generally speaking, its aqueous solution is acidic. Display absorption peaks within the UV visible spectral range, with characteristic absorption wavelengths that can be used for analysis and quantitative determination.

|
Chemical Formula |
C12H17NO9 |
|
Exact Mass |
319 |
|
Molecular Weight |
319 |
|
m/z |
319 (100.0%), 320 (13.0%), 321 (1.8%) |
|
Elemental Analysis |
C, 45.14; H, 5.37; N, 4.39; O, 45.10 |
|
Melting point |
102-105 ° C |
|
Boiling point |
360 ° C |
|
Density |
1.082 ± 0.06 g / cm3 (predicted) |
|
Storage conditions |
2-8 ° C |
|
Specific rotation |
- 46 º (C = 0.5, HCl) |
|
Solubility H2O |
50 mg / ml |
|
|
|
The main component of heavy Noradrenaline acid tartrate is norepinephrine, which is an alpha receptor and beta 1 receptor agonist. In terms of pharmacological effects, it has a stronger effect on alpha receptors than adrenaline, but almost no effect on beta 2 receptors. This makes norepinephrine bitartrate have significant pharmacological effects in vasoconstriction and hypertension.
Norepinephrine bitartrate can cause extensive vasoconstriction, which is one of its core pharmacological effects. Specifically, it activates the alpha 1 receptor on the membrane of vascular smooth muscle cells, causing blood vessels to contract and increasing peripheral resistance, leading to an increase in blood pressure. This vasoconstrictive effect is not limited to skin, mucous membranes, and visceral blood vessels, but also includes coronary arteries and cerebral blood vessels.
In terms of coronary arteries, although norepinephrine can cause vasoconstriction, under certain conditions, it can also cause coronary artery dilation and increase cardiovascular and cerebrovascular blood flow. This may be due to the different effects of norepinephrine on different parts of the coronary artery, or its interaction with other vasoactive substances.
In terms of cerebral blood vessels, norepinephrine can reduce cerebral blood flow by constricting cerebral blood vessels, but in some cases, such as severe hypotension, it may also maintain normal brain tissue function by increasing cerebral blood flow.

2. Elevated blood pressure

Norepinephrine bitartrate can rapidly and effectively increase diastolic blood pressure and pulse pressure, which is another important pharmacological effect. It stimulates alpha receptors to produce vasoconstriction, increasing peripheral resistance and thus raising blood pressure. At the same time, norepinephrine can also excite β 1 receptors, leading to increased myocardial contractility, faster heart rate, increased cardiac output, and further hypertension.
In clinical practice, norepinephrine bitartrate is commonly used to treat various hypotensive states, such as acute myocardial infarction, hypotension caused by extracorporeal circulation, and shock caused by insufficient blood volume. In these cases, norepinephrine can rapidly increase blood pressure, ensuring blood supply to important organs and maintaining stable vital signs.
3. Enhance myocardial contractility
Tartrate norepinephrine can enhance the contractility of myocardial cells and improve cardiac function by exciting β 1 receptors. This effect is particularly important in cardiac resuscitation. When a patient experiences cardiac arrest, in addition to using cardiac compressions, artificial respiration, and other cardiopulmonary resuscitation measures, it is also necessary to take norepinephrine bitartrate and adrenaline, dilute them appropriately with physiological saline, and inject them directly intravenously. By stimulating the alpha and beta receptors, the responsiveness of the myocardium is increased, which is beneficial for cardiac arrest and improves the success rate of rescue.


4. Improve shock state
Shock is a severe pathophysiological state characterized by decreased blood pressure, inadequate tissue perfusion, and organ dysfunction. Norepinephrine bitartrate can significantly improve shock state by constricting blood vessels, increasing blood pressure, and enhancing myocardial contractility. It can not only increase peripheral resistance and raise blood pressure, but also contract visceral and skin blood vessels, reduce visceral blood flow and increase cardiovascular and cerebrovascular blood supply, helping to maintain blood supply to important organs such as the brain and heart.
5. Maintain vital signs
After cardiac arrest resuscitation, norepinephrine bitartrate can be used to maintain blood pressure, stabilize circulatory system function, and ensure stable vital signs. In addition, it can also be used for the treatment of hypotension during spinal canal block, as well as for the adjuvant treatment of shock, hypotension caused by insufficient blood volume or hypotension after pheochromocytoma resection.


This is our advanced product Noradrenaline Acid Tartrate
Remark: BLOOM TECH(Since 2008), ACHIEVE CHEM-TECH is the subsidiary of us.

The laboratory synthesis of L-4- (2-Amino-1-hydroxyethyl) -1,2-benzenediol bitartrate can usually be carried out through the following steps:
Chemical equation:
C8H8O2+phenylhydrazine mesylate → p-hydroxyacetophenone phenylhydrazine
P-hydroxyacetophenone phenyl hydrazine+H2+catalyst → L-4-(2-Amino-1-hydroxyethyl)-1,2-benzenediol
Chemical equation:
L-4-(2-Amino-1-hydroxyethyl)-1,2-benzenediol+Cl2OS+base → L-4-(2-Amino-1-hydroxyethyl)-1,2-benzenediol phenylhydrazine sulfite
Chemical equation:
L-4-(2-Amino-1 hydroxyethyl)-1,2-benzenediol phenylhydrazine sulfite+C8H11NO → L-4-(2-Amino-1 hydroxyethyl)-1,2-benzenediol phenylhydrazine
Chemical equation:
L-4-(2-Amino-1 hydroxyethyl)-1,2-benzenediol phenylhydrazine+C4H6O6 → L-4-(2-Amino-1 hydroxyethyl)-1,2-benzenediol bitartrate
Please note that the above are only the basic steps and corresponding chemical equations of the synthesis method for L-4- (2-Amino-1-hydroxyethyl) -1,2-benzenediol bitartrate. In actual synthesis, it may be necessary to optimize and adjust reaction conditions, solvents, and catalysts. In addition, to ensure safety and success rate, please follow the correct chemical experimental procedures during laboratory operations, and conduct further literature research and experimental design based on specific experimental situations.

Only one carboxyl group in a molecule of tartaric acid is dehydrogenated and salted with norepinephrine, which is known as bariterate or bitartrate. The aqueous solution has certain acidity. The reason for using tartaric acid to salt norepinephrine is the product of chiral resolution.
Noradrenline acid tartrate method-neutralization titration method. Method principle : The test sample was placed in a conical flask, dissolved with acetic acid, added crystal violet indicator solution, titrated with perchloric acid titration solution ( 0.1 mol / L ) until the solution showed blue and green. Read out the amount of perchloric acid titration solution and calculate the content of norepinephrine tartrate.
L-4- (2-Amino-1-hydroxyethyl) -1,2-benzenediol tartrate is a compound widely used in the pharmaceutical field.
L-4- (2-Amino-1-hydroxyethyl) -1,2-benzenediol bitartrate is often used as a β Receptor blockers are used to treat cardiovascular diseases such as hypertension, arrhythmia, coronary heart disease, and heart failure. It suppresses β The activity of receptors reduces the contractility and heart rate of the heart, thereby reducing the burden on the heart.
Bisoprolol free base tartrate can be used as a first-line drug for controlling hypertension. It helps to lower blood pressure levels and reduce the risk of cardiovascular complications by reducing the contractile force and heart rate of the heart, lowering blood pressure.
L-4-(2-Amino-1-hydroxyethyl)-1,2-benzenediol bitartrate is widely used in the treatment of myocardial infarction. It can reduce the risk of recurrence of myocardial infarction and reduce the incidence of cardiovascular events.
This compound can be used to alleviate symptoms of angina (a type of chest pain caused by insufficient coronary artery blood supply). By reducing cardiac load and reducing cardiac oxygen demand, it can help alleviate chest pain and discomfort caused by angina.
Bisoprolol free base tartrate has a certain therapeutic effect on controlling arrhythmia such as atrial fibrillation and ventricular fibrillation. It suppresses β The activity of receptors regulates cardiac electrical activity, improves heart rhythm, and achieves the effect of treating arrhythmia.
L-4-(2-Amino-1-hydroxyethyl)-1,2-benzenediol bitartrate can be used as a preventive drug to reduce the frequency and severity of migraine attacks in patients. It plays a role in reducing migraine by regulating neurotransmitters and dilating blood vessels.
Bisoprolol free base tartrate is sometimes used to treat hyperthyroidism. It can inhibit the effects of thyroid hormone on β The stimulating effect of receptors reduces the responsiveness of the cardiovascular system and the impact of thyroid hormones.
In addition to the main applications mentioned above, Noradrenline acid tartrate has also been used for research and applications in other medical fields, such as the treatment of anxiety disorders and the study of neuroprotective effects.

The molecular formula of Noradrenaline Acid Tartrate is C8H11NO3 · C4H6O6, with a molecular weight of 337.28. It consists of two parts: norepinephrine and bitartrate. Norepinephrine is the main bioactive component, while tartaric acid acts as an acidifier to increase its water solubility.
From a chemical structure perspective, norepinephrine bitartrate has a benzene ring structure, which is connected to two hydroxyl groups (- OH) and one amino group (- NH2). In addition, it also has a side chain with a methyl group (- CH3) and a hydroxyl group (- OH) on the side chain. These functional groups endow norepinephrine bitartrate with unique biological activity and pharmacological effects.
2, Molecular structure analysis
1. Benzene ring structure: The benzene ring is the core of the molecular structure of norepinephrine bitartrate. The π electron cloud density on the benzene ring is high, which gives the benzene ring strong stability and conjugation effect. These characteristics enable norepinephrine bitartrate to bind to various receptors in the organism, producing a wide range of physiological effects.
2. Functional group analysis:
Hydroxyl (-OH):
The molecule of norepinephrine bitartrate contains two hydroxyl groups, located on the benzene ring and side chains, respectively. These hydroxyl groups can form hydrogen bonds with water molecules in living organisms, thereby increasing their water solubility. At the same time, hydroxyl groups also have a certain degree of acidity and can undergo acid-base reactions with alkaline substances in organisms, further expanding their biological activity.
Amino group (-NH2):
Amino group is another important functional group in the molecule of norepinephrine bitartrate. Amino groups can react with various substances in living organisms, such as binding with protons to form ammonium salts, and forming amides with carboxylic acids. These reactions not only affect the pharmacological effects of norepinephrine bitartrate, but are also closely related to its metabolism and excretion in the organism.
Methyl (-CH3):
Methyl is a simple hydrocarbon group on the side chain. Its existence distinguishes norepinephrine bitartrate structurally from catecholamine compounds such as adrenaline. The presence of methyl groups may affect the affinity of norepinephrine bitartrate with receptors, as well as its stability and metabolism in living organisms.
Stereoscopic structure:
The stereostructure of norepinephrine bitartrate has a significant impact on its biological activity. For example, the relative positions of substituents on the benzene ring and functional groups on the side chains in space may affect their binding mode and affinity with receptors. Therefore, the study of the stereostructure of norepinephrine bitartrate contributes to a deeper understanding of its pharmacological mechanism of action.
Hot Tags: noradrenaline acid tartrate cas 69815-49-2, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, cholesterol powder, tetramisole hcl powder, epinephrine powder, articaine hydrochloride powder, tetramisole hydrochloride powder, procaine powder