Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of sodium cyanoborohydride solution cas 25895-60-7 in China. Welcome to wholesale bulk high quality sodium cyanoborohydride solution cas 25895-60-7 for sale here from our factory. Good service and reasonable price are available.
Sodium cyanoborohydride, also known as NaBH3CN, is a versatile organic composite with a CAS number of 25895-60-7. Its molecular formula is CH2BNNa or commonly written as NaBH3CN, and it has a molecular weight of approximately 62.83-62.85 g/mol. This white or slightly yellow solid powder is widely recognized as a gentle and selective reducing agent in organic chemistry.

|
|
|
|
Chemical Formula |
CH3BNNa |
|
Exact Mass |
63.03 |
|
Molecular Weight |
62.84 |
|
m/z |
63.03 (100.0%), 62.03 (24.8%), 64.03 (1.1%) |
|
Elemental Analysis |
C, 19.11; H, 4.81; B, 17.20; N, 22.29; Na, 36.58 |
It is soluble in water and various polar organic solvents such as methanol, ethanol, tetrahydrofuran (THF), and acetic acid. Its solubility in water at 29°C is 2120 g/L, and it exhibits varying solubility in THF depending on temperature (e.g., 372 g/L at 28°C, 410 g/L at 46°C, 422 g/L at 62°C). It is also soluble in diethylene glycol dimethyl ether but is only slightly soluble in ethanol and insoluble in ethers, benzene, and hexane.
It is stable in solutions with a pH of around 3 but can decompose when heated, with a melting point range of 240-242°C (decomposition). It is hygroscopic and should be stored under dry conditions, preferably under an inert atmosphere such as nitrogen or argon.

Sodium cyanoborohydride (chemical formula NaBH3CN) is an important inorganic composite that has shown wide application value in multiple fields due to its unique chemical properties.
Reducing agents in organic synthesis

As a mild reducing agent, it plays a crucial role in organic synthesis. Although its reducibility is slightly weaker than that of sodium borohydride, it can remain stable under specific conditions (such as acidic environments at pH 3), which gives it a unique advantage in reduction reactions.
Reduction of aldhydes and ketosis: It can effectively reduce aldhydes and ketosis to their corresponding alcohols. This reaction is extremely common in organic synthesis and is an important means of constructing carbon carbon and carbon oxygen bonds.
For example, in drug synthesis, the reduction reactions of aldhydes and ketosis are commonly used to prepare alcohol composites with specific biological activities.
Reductive amination: It performs well in the reductive amination reaction of ketosis. It can reduce the imine intermediate genrated by the reaction of ketosis and amines to amines, thereby constructing new carbon nitrogen bonds.


This reaction is of great significance in drug synthesis and total synthesis of natural products, and is an effective method for preparing nitrogen-containing heterocyclic composites and amine composites.
Selective reduction: exhibits high selectivity in reduction reactions. It can selectively reduce specific functional groups without interfering with other functional groups. For example, in molecules containing functional groups such as esters, lactones, nitro groups, halides, or epoxy composites, sodium cyanoborohydride can selectively reduce aldhydes, ketosis, or imines while retaining other functional groups unchanged.
Reduction of special functional groups
In addition to the reduction of aldhydes and ketosis, it can also reduce a series of special functional groups, providing more possibilities for organic synthesis.
Reduction of imines and amonium salts: capable of reducing imines and amonium salts to genrate corresponding amine composites. This reaction is of great significance in drug synthesis and total synthesis of natural products, and is an effective method for preparing nitrogen-containing heterocyclic composites and amine composites.


Reduction of oxime and oxime derivatives: capable of reducing oxime and its derivatives to genrate corresponding amine or alcohol composites. This reaction is commonly used in organic synthesis to prepare composites with specific biological activities.
Reduction of hydrazones and enamines: capable of reducing hydrazones and enamines to genrate corresponding amines or hydrocarbon composites. This reaction is of great significance in constructing carbon nitrogen and carbon carbon bonds.
Reduction of polarized olefins, alkyl halides, epoxides: It can also reduce functional groups such as polarized olefins, alkyl halides, and epoxides to genrate corresponding hydrocarbons or alcohol composites. This reaction is commonly used in organic synthesis to prepare composites with specific structures.

Other uses in industrial applications

In addition to the field of organic synthesis, it has also demonstrated other applications in industrial settings.
Foaming agents for plastics, rubber, and their adhesives: can be used as foaming agents for plastics, rubber, and their adhesives. It can decompose under heating conditions to produce gas, causing the material to expand and form a porous structure, thereby improving the physical and processing properties of the material.
Ethylene polymerization catalyst: One of the components that can be used as an ethylene polymerization catalyst.
It can participate in the polymerization reaction of ethylene, regulate the molecular structure and properties of polymers.
Color tone improver for paper and fiber: can be used as a color tone improver for paper and fiber. It can react with certain components in paper or fibers to improve their color and appearance quality.
Mercury containing wastewater treatment agent: can be used as a mercury containing wastewater treatment agent. It can react with mercury ions to genrate stable precipitates, thereby removing mercury ions from wastewater and reducing their toxicity.


Preparation of sodium cyanoborohydride solution:
According to the different raw materials used, the synthesis methods of NaBH4 can be divided into four categories:
Borane or organic boron as raw materials;
Boron trihalide and sodium tetrafluoroborate are used as raw materials;
Borate ester as raw material;
The raw materials are boron oxide, boron phosphate and borate.
The commonly used method is the sodium cyanoborohydride solution method:
Add boric acid and an appropriate amount of methanol to the distillation pot, slowly heat, and completely reflux at 54 ° C for 2 hours, then collect the azeotrope of boric acid methane and methanol.
After sulfuric acid treatment and distillation, a relatively pure product can be obtained.
Sodium hydride, produced by the reaction of hydrogen and sodium, is sent into a condensation reaction tank and heated to about 220 ° C while stirring. Then, methyl borate is added and heating stops at 260 ° C. The feed temperature should be controlled below 280 ° C. Continue stirring after feeding to allow it to fully react.
After the reaction is complete, cool to below 100 ° C and centrifuge to produce a filter cake of condensed products. Add an appropriate amount of water to the hydrolysis machine, slowly place the filter cake into the hydrolysis machine, control the temperature below 50 ° C, increase the temperature to 80 ° C after feeding is complete, and centrifuge for separation.
Then feed the hydrolysis solution into the layering machine and let it stand for 1 hour before automatic layering. The lower hydrolysis solution is a sodium borohydride solution.
The reaction formula is as follows:

Note: Hydrocyanic acid is used for this preparation, and the preparation and reaction of co solution should be carried out in the fume hood.
Install a constant pressure drip funnel, a mechanical stirrer and a reflux condenser on a clean and dry 2L three port flask. The outlet of the condenser is connected to a washing bottle containing 1mol sodium hydroxide solution to remove the escaping hydrocyanic acid vapor, and then a gas meter is introduced to measure the amount of hydrogen genrated.
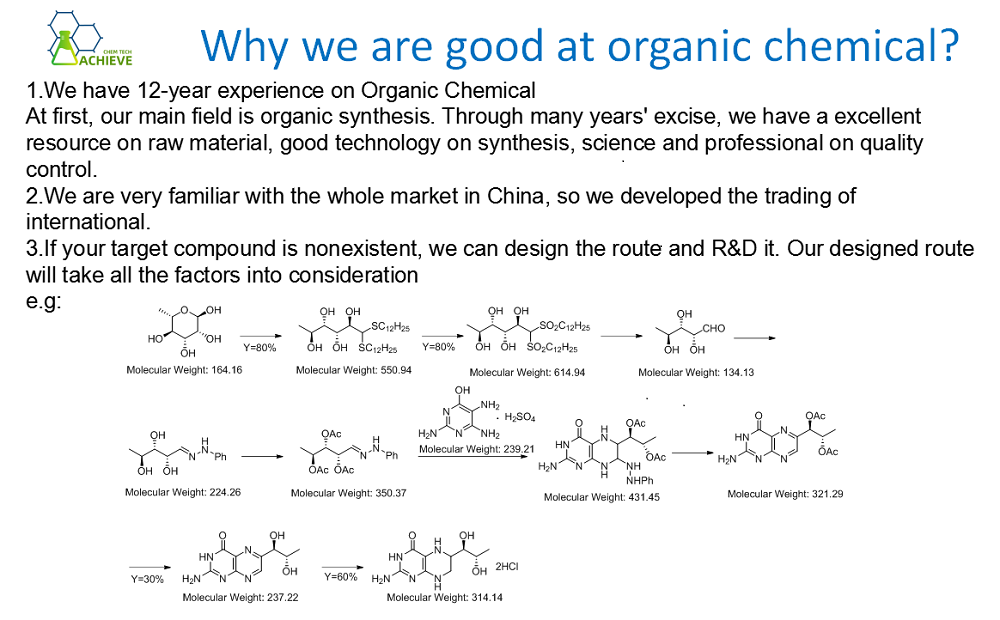
Add 1000mL tetrahydrofuran (dried with CaH2 in advance and filtered before use) and 80.2g (2.09mol) 98.5% sodium borohydride into the flask. Clean the flask with dry nitrogen. The dropping funnel contains 16.7% (weight ratio) solution of hydrocyanic acid tetrahydrofuran (294g, containing 58.8g or 2.33mol 98% hydrocyanic acid). This system simply cleans again. Slowly add the hydrocyanic acid solution to the sodium borohydride slurry at room temperature to release hydrogen slowly and immediately.
![]()
As the reaction is slightly exothermic, the flask is kept at 25 ℃ by cooling in a water bath. After stirring for 1h, the mixture is gradually heated to reflux until the hydrogen is discharged. After the reaction mixture is cooled to suffocation temperature, clean it with nitrogen repeatedly and then evacuate it. A small amount of insoluble solids shall be filtered with filter paper lined with activated carbon. Concentrate and dry the transparent light yellow filtrate in the vacuum rotary evaporator. Before heating, remove about half of the solution, and finally dry it under vacuum at 60 ℃ to obtain 120g of white solid sodium cyanobohydride with a yield of 91%. The product may contain B (OH) 3 impurities through element analysis. Dissolve the crude product in tetrahydrofuran (20% V/V), filter, add 4 times of excess dichloromethane, and recrystallize twice to obtain the pure product.
FAQ
What is sodium cyanoborohydride used for?
Sodium cyanoborohydride (CBH) is commonly used as a mild reducing agent in the reductive amination of aldehydes and free amines. Within the pharmaceutical industry, this reaction is employed in the bioconjugation of proteins and peptides.
Is sodium cyanoborohydride toxic?
Poison by ingestion. Causes gastrointestinal tract burns. Ingestion may result in symptoms similar to cyanide poisoning which is characterized by asphyxiation.
What is the difference between NaBH4 and NaBH3CN?
Sodium cyanoborohydride (NaBH3CN) is a mild reducing agent that is commonly used in reductive aminations. The presence of the electron-withdrawing cyano (CN) group makes it less reactive than sodium borohydride (NaBH4).
What do NaBH4 and NaOH do?
At lower pH levels, sodium borohydride reacts exothermically with water to generate flammable hydrogen gas. The heat may ignite the hydrogen, the solvent, and surrounding combustible materials. SODIUM BOROHYDRIDE AND SODIUM HYDROXIDE SOLUTION is a powerful basic reducing agent.
Hot Tags: sodium cyanoborohydride solution cas 25895-60-7, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, N N Dimethylformamide pure, Hydroxylamine hydrochloride powder, Basic Chemicals, Solvent, propylene oxide market, Raw Materials




