Lead acetate trihydrate is a chemical substance with the molecular formula C4H6O4Pb.3 (H2O), CAS 6080-56-4, Also known as lead acetate (II) trihydrate. White crystalline or flaky powder, industrial products often have large brown or gray blocks. Flammable. When exposed to air, it absorbs carbon dioxide and becomes an insoluble substance in water. Slightly vinegar flavored. Easily soluble in water, slightly soluble in alcohol, and easily soluble in glycerol. Relative density 2.55 (25/4 ℃), melting point 75 ℃ (rapid heating), solubility 55.04g/100g of water, water loss at 75 ℃. Decompose at 200 ℃. Anhydrous products have a melting point of 280 ℃ and a relative density of 3.25 (20/4 ℃). 44.3g can be dissolved in 100g of water at 20 ℃. In addition, lead acetate decahydrate is a white orthogonal crystal with a relative density of 1.69 (25/4 ℃), which is prone to weathering and dehydration at room temperature. Mainly used for preparing other lead compounds, such as lead oxide, lead carbonate, lead nitrate, etc. It can be used as a catalyst or precursor for catalytic reactions in organic synthesis. In addition, it is widely used in industries such as pigments, dyes, rubber, textiles, cosmetics, etc. There are also some other applications available. For example, it can be used to prepare pyroelectric thin films, ceramic materials, glass and other products. In the process of preparing pyroelectric thin films, it can be used as a raw material to participate in the sol gel reaction process and prepare pyroelectric thin films with excellent performance. In the preparation process of ceramic materials and glass, it can be used as an additive or flux to improve the performance and appearance quality of products.

|
|
|
|
Chemical Formula |
C4H12O7Pb |
|
Exact Mass |
380 |
|
Molecular Weight |
379 |
|
m/z |
380 (100.0%), 378 (46.0%), 379 (42.2%), 381 (4.3%), 376 (2.7%), 379 (2.0%), 380 (1.8%), 382 (1.4%) |
|
Elemental Analysis |
C, 12.67; H, 3.19; O, 29.52; Pb, 54.62 |
The chemical formula of Lead Acetate Trihydrate is Pb (C2H3O2) 2-3H2O. This compound is composed of lead, acetate ions, and water molecules. We will gradually analyze the various components of its molecular structure.

1. Lead ion (Pb2+)
It is the center of the compound. The electronic structure of lead ions is [Xe] 4f ^ 14 5d ^ 10 6s ^ 2, which means they have a valence electron that can form chemical bonds. In this compound, lea ions are surrounded by six oxygen atoms in a tetrahedral coordination environment.
2. Acetate ion (CH3COO-)
Adopting advanced technology and concepts of the industrial internet, it helps manufacturing enterprises create a unified digital system covering the entire process of production and management.
3. Water molecules (H2O)
There are three water molecules present in the product. These water molecules combine with lea acetate to form trihydrates. They form hydrogen bonds with lead ions or other water molecules through oxygen atoms. Water molecules play an important role in stabilizing the crystal structure of compounds.

Lead acetate trihydrate (chemical formula: Pb (CH3COO) 2 · 3H2O) is a white or slightly yellow crystal with hygroscopicity, easily soluble in water and acetic acid. As an important chemical substance, it has a wide range of applications in industry, scientific research, and other fields.

1. Pigment industry
It occupies an important position in the pigment industry. As a commonly used white pigment, it has good covering power and luster, and is therefore widely used in fields such as coatings, paints, plastics, etc. Lea acetate pigment has durability and stability, making it an ideal choice for indoor and outdoor decoration, building coatings, and other projects.
Specifically, it can be mixed with other pigments and additives to prepare coatings with different colors and properties. In the process of coating manufacturing, it can provide good covering power and gloss, making the coating have better decorative effect and durability. In addition, due to the stability of lea acetate pigments, they can maintain bright colors for a long time and are not easily faded.
2. Battery manufacturing
It also has important applications in the field of battery manufacturing. It can be used as a material for battery plates to provide electronic conductivity. Lead acetate batteries are widely used in small electronic devices, backup power sources, and other fields due to their low self discharge rate and long lifespan.
During the battery manufacturing process, it undergoes a series of chemical reactions to form lea-acid salts with good conductivity. These lead-acid salts can serve as active substances for the battery plates and participate in the charging and discharging processes of the battery. Due to its low self discharge rate and long lifespan, lead-acid batteries can maintain stable electrical energy output for a long time, meeting the energy needs of various electronic devices.


3. Electrochemical research
In electrochemical research, it also plays an important role. It can be used as an electrode material or electrolyte additive to prepare high-performance electrochemical devices such as batteries and supercapacitors. By optimizing the preparation process and electrochemical performance of product, the energy density, power density, and cycle stability of electrochemical devices can be further improved.
Specifically, it can be prepared into electrode materials or electrolyte additives through chemical reaction processes such as dissolution and precipitation. These materials have good electrochemical performance and stability, and can be used to prepare high-performance electrochemical devices such as batteries and supercapacitors. By optimizing the composition and structure parameters of materials, the performance and stability of electrochemical devices can be further improved.
4. Synthetic industry
In the synthetic industry, it also has important application value. It can participate in various chemical reaction processes as a catalyst, intermediate, etc., to prepare compounds with specific structures and properties.
For example, it can act as a catalyst to participate in chemical reaction processes such as esterification and oxidation. By optimizing the composition and structure parameters of the catalyst, the conversion rate and selectivity of chemical reactions can be further improved. In addition, it can also serve as a reaction intermediate in the preparation of various organic and inorganic compounds and other products.


Lead acetate trihydrate is a white powder with a monoclinic crystal structure. In the spatial group P21/c, the cell parameters are a=0.8449 nm, b=1.0767 nm, and c=1.4099 nm, β= 90.92 °. Its structure consists of tetrahedral coordinated lead ions, acetate ions, and water molecules, with each lea ion surrounded by six oxygen atoms, forming an octahedral coordination environment. Acetate ions and water molecules combine with lea ions through a double bridge connection, forming a three-dimensional network structure.
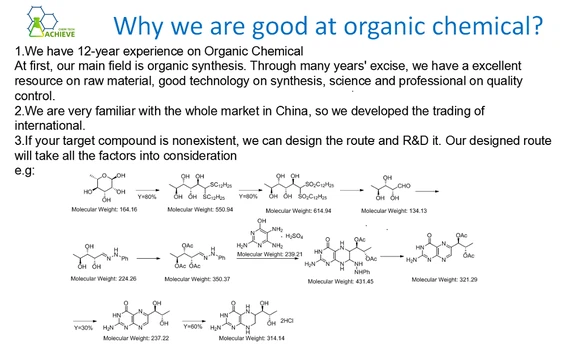
1. It is obtained by reacting lead oxide with acetic acid. Dissolve lead oxide in 80% hot acetic acid until soaked, filter, add a small amount of acetic acid to the filtrate, and evaporate to a relative density of 1.40. Cool, shake filter, and dry to obtain lead acetate. The purity of industrial lead acetate can generally reach over 98%. After purification, 1% acetic acid solution can be used for recrystallization, or lead trihydrate acetate can be dissolved in water and hydrogen sulfide is introduced to precipitate lead sulfide and other impurities. After filtration, fluorescent gallium reagents (complexing agents of Co, Al, Cu, etc.) are added to the filtrate, a small amount of activated carbon is added, and EDTA sodium salt is added for treatment to obtain extremely pure reagent grade products.
2. Dissolve lead oxide (II) in 50% hot acetic acid until saturated. Filter, add a small amount of acetic acid to the filtrate, evaporate on a water bath to a relative density of 1.40, let stand, and crystals precipitate. Filter and dry between layers of filter paper to obtain pure product. Place this product in the air. It will absorb gases such as carbon dioxide and ammonia in the air, generating carbonates or alkaline salts. It can be purified by recrystallization with a 2% to 3% acetic acid aqueous solution. 100g of lead acetate needs to be dissolved in 100mL of 2% to 3% acetic acid aqueous solution. After heating, the insoluble matter is filtered out while it is hot. After the filtrate is cooled, it is allowed to stand until the crystals precipitate, and the crystals are filtered and dried at room temperature. Concentrate the mother liquor to half the original volume, add a small amount of acetic acid, and cool it to obtain some more products.
3. Dissolve lead oxide in hot acetic acid, filter, add acetic acid to the filtrate for reaction, and evaporate to a relative density of 1.40. Cool, separate crystals, and dry to obtain pure peoduct.
4. Boil 100kg of distilled water with steam for 15 minutes to remove carbon dioxide, then add 0.5kg of industrial acetic acid and 100kg of industrial lead acetate. Continuously steam until the lead acetate is completely dissolved, then add a small amount of industrial activated carbon, stir evenly, and filter while hot (with three layers of filter paper). The resulting filtrate is cooled, crystallized, centrifuged, and dried. It is then laid flat and dried in a 30-40 ℃ oven, occasionally flipped and crushed. The mother liquor can be recycled.
FAQ
Is lead acetate banned?
On October 31, 2018, FDA repealed the regulation which provides for the use of lead acetate in hair dyes because there is no longer a reasonable certainty of no harm from the use of this color additive. Of note, temporary tattoos marketed as "black henna" contain PPD and may increase your risk of allergy to hair dyes.
What is another name for lead acetate?
It is also known as Plumbous acetate, salt of Saturn, sugar of lead, Goulard's powder or lead diacetate. Its systematic IUPAC name is Lead(II) ethanoate. Lead acetate is also toxic like other lead compounds.
What is acetate most commonly used for?
Known for its luxurious, silky texture, acetate is commonly used in fashion for its smooth finish and elegant drape. Often compared to silk, acetate fabric offers a cost-effective yet visually appealing alternative, making it a popular choice in the production of formalwear and linings.
What is lead acetate trihydrate used for?
Application. Lead(II) acetate trihydrate can be used as a reagent to synthesize lead-based coordination polymers by using isomeric phenylenediacetic acids. It can also be used as a precursor to synthesize lead sulfide nanoparticles.
What is lead acetate solution used for?
Lead(II) acetate is also used as a mordant in textile printing and dyeing, and as a drier in paints and varnishes. It was historically used as a sweetener and preservative in wines and in other foods and for cosmetics.
Is lead acetate harmful?
Lead Acetate is a PROBABLE CARCINOGEN in humans. There is some evidence that inorganic Lead compounds cause lung, brain, stomach, and kidney cancer in humans and they have been shown to cause kidney cancer in animals. a carcinogen. has been shown to be a teratogen in animals.
Is lead acetate still used in cosmetics?
Effective from 6 January 2022, the Food and Drug Administration has amended the colour additive regulations to no longer provide for the safe use of lead acetate in cosmetics intended for colouring hair on the scalp.
Hot Tags: lead acetate trihydrate cas 6080-56-4, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID, Cerium sulfate powder, CHICAGO SKY BLUE 6B, Consumable, Iridium III chloride








