5-Bromo-1-pentene, an unsaturated alkyl bromide, has unmistakable physical, substance, and otherworldly qualities got from its particular atomic arrangement. Digging into these properties is essential for utilizing the compound in different applications inside natural amalgamation and the making of useful materials.Investigating the remarkable highlights of 5-Bromo-1-pentene empowers specialists to reveal its expected purposes in assorted synthetic responses and material plan. Its underlying credits direct its conduct in various conditions, affecting its reactivity and communications with different mixtures.
What are the physical properties of 5-bromo-1-pentene?
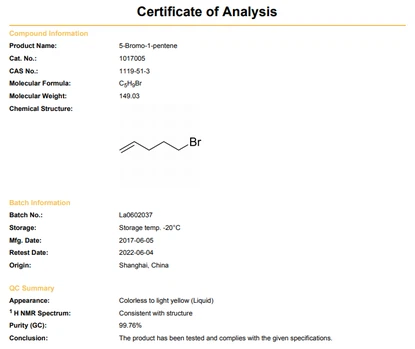
5-Bromo-1-pentene shows unmistakable actual qualities that assume a critical part in its taking care of and application. Introducing as an unmistakable, dry fluid at room temperature, this compound produces an impactful and lachrymatory scent, making it effectively recognizable. With a limit scope of 119-121°C and a dissolving point of - 110°C, it shows an unstable nature, demonstrated by its generally low edge of boiling over and moderate fume tension of 3.5 mm Hg at 20°C.

In general, the blend of these actual properties connotes 5-Bromo-1-pentene's unpredictability, immiscibility with water, and non-polar nature, which are essential contemplations for its utilization in natural amalgamation, substance responses, and material applications. Its unmistakable scent and actual properties make it conspicuous and guide its taking care of in research facility settings, guaranteeing security and accuracy in trial techniques including this compound.
What are the chemical properties of 5-bromo-1-pentene?
Artificially, 5-bromo-1-pentene shows reactivity predictable with an alkyl bromide alkene:
- Nucleophilic replacement: The allylic bromine permits SNi responses with nucleophiles like amines, alkoxides, thiols, and cyanide.
- Expansion responses: The alkene goes through electrophilic augmentations of incandescent light, hydrogen halides, interhalogens, and extremists.
- Disposal: Treatment with base can kill HBr to yield 1,5-pentadiene.
- Oxidation: Permanganate oxidation cuts the alkene to produce more modest carboxylic acids.
- Polymerization: The alkene can polymerize, however more slow than additional responsive unhindered alkenes.
- Hydrohalogenation: Expansion of HCl, HBr or HOAc yields halohydrin subsidiaries.
- Photoisomerization: UV light isomerizes the 1-pentene to 2-pentene.
So it shows the joined reactivity of an alkyl halide and a 1-alkene, giving a flexible manufactured handle.
What are the spectral properties of 5-bromo-1-pentene?
Examination by spectroscopy uncovers:
- 1H NMR: Trio at 5.8 ppm (alkene), group of four at 3.4 ppm (allylic CH2), trios at 2.1 ppm and 1.85 ppm.
- 13C NMR: Tops at 139 ppm (alkene), 33.5 ppm (allylic), and 22-28 ppm (alkyl).
- Infrared range: Medium tops around 1640 cm-1 (alkene C=C) and 650 cm-1 (alkyl halide C-Br).
- Mass range: Parent particle top at 158 m/z and trademark isotopic example of bromine containing compounds (M+2 top at 160 m/z).
- UV range: Assimilation most extreme around 215 nm because of the alkene π-π* change.
These spectra permit unambiguous ID and portrayal of the 5-bromo-1-pentene atom.
How do the properties inform applications of 5-bromo-1-pentene?
The properties of 5-Bromo-1-pentene, including its unsaturated nature, alkyl bromide usefulness, and explicit ghastly highlights, straightforwardly impact its applications across different fields. For example, its unsaturated nature makes it a significant structure block in natural blend, empowering the production of mind boggling particles through different synthetic changes like expansion responses, cross-coupling responses, and polymerization processes. Furthermore, its alkyl bromide moiety can act as a purpose in synthetic change, considering the presentation of new useful gatherings or cooperation in replacement responses.
What are the considerations for handling 5-bromo-1-pentene based on its physical properties?
- The disturbing smell requires use in a smoke hood with air extraction to forestall inward breath.
- The moderate edge of boiling over permits instability, so fixed warmed compartments might be expected for responses.
- The non-polar nature educates decisions regarding viable natural solvents for responses and detachments.
- Low fluid solvency implies water similarity is restricted, while natural dissolvability is wide.
- Understanding the fume pressure, combustibility limits and electrical conductivity illuminates safe capacity and dealing with conventions.
How do the chemical attributes inform application in synthesis?
- The reactivity profile of alkenes assumes a pivotal part in deciding the most reasonable response conditions to accomplish greatest selectivity in natural blend. Furthermore, the SNi capacity of alkenes widens the extent of functionalization choices past what is regularly attainable with customary alkenes. Besides, the potential for oxidative cleavage considers fracture into more modest synthons, further growing the manufactured conceivable outcomes. It is fundamental to have a profound comprehension of both the end and polymerization propensities of alkenes to guarantee the respectability of the ideal items. Besides, having the option to perceive the photoisomerization pathway empowers the advancement of brightening conditions and steadiness, offering further command over the response cycle.
How do the spectral data facilitate analysis and identification?
-NMR, IR and UV information give a unique mark to recognizing immaculateness versus results.
- The mass range gives an example to empower recognizing presence in response combinations.
- Conceivable manufactured course intermidiates can be proposed in light of telling pinnacles.
- Phantom library matching improves on appointing obscure spectra to this compound.
Generally speaking, an intensive comprehension of 5-bromo-1-pentene's credits empowers boosting its utility while keeping away from entanglements across natural science applications.
Conclusion
The blend of an alkyl bromide and terminal alkene gives it with a one of a kind profile of physical, compound and ghastly properties. These traits make it a flexible manufactured incorporating block for bringing responsive usefulness into natural particles and materials. Experiences into its attributes work with taking care of, further develop response results, improve on item ID and explain the conduct across assorted functionalizations. 5-Bromo-1-pentene fills in as a great representation of how understanding the properties of substance reagents illuminates ideal utilization.
References
1. Becker, E. I. (1977). Comprehensive Organic Chemistry: The Syntheses and Reactions of Organic Compounds, Vol 2 Part 1B. Pergamon Press.
2. Clayden, J., Greeves, N., & Warren, S. (2012). Organic Chemistry (2nd ed.). Oxford University Press.
3. Gurtowska, N., Żyłka, R., Osmialowski, B., & Matuschewski, H. (2014). Determination of thermodynamic parameters of 5-bromo-1-pentene fromspeed of sound measurements. The Journal of Chemical Thermodynamics, 74, 184-187.
4. Kumbhar, A. A., Padmanabhan, P., Salunke, J. K., Mahulikar, P. P., & Gund, M. R. (2013). Functionalization of chromium tricarbonyl complexes with bromoalkenes: Synthesis, characterization, crystal structures and ethylene tetramerization study. Polyhedron, 52, 309-324.
5. Prasad, D. J., Ashok, T. D., & Mahalingam, A. K. (2016). An efficient chemoselective synthesis of novel chromeno [4, 3-b] pyrroles using 5-bromo-1-pentene as a building block under microwave irradiation. Tetrahedron Letters, 57(7), 865-868.

