Articaine HCl, the chemical formula is C13H20N2O3S·HCl, CAS 23964-57-0. Is a local anesthetic widely used in dental and other surgical procedures. It is the hydrochloride salt form of Articaine and has a unique set of chemical properties. It is a four-ring structure consisting of a benzene ring, a thiophene ring, a methylpropylaminocarboxyl group and an alkyl group. Possesses a chiral carbon atom, so two enantiomers exist: (S)-Articaine HCl and (R)-Articaine HCl. It is in the form of hydrochloride, so it is acidic. When Articaine HCl is dissolved in water, it completely dissociates into positively charged Articaine cations and chloride ions (Cl-). This dissociation process is a reversible reaction that depends on the acid-base balance in the solution. Different enantiomers have different optical activity properties, among which (S)-Articaine HCl has specific optical activity. Solubility may vary at different pH values. Under acidic conditions, the solubility may decrease slightly, while in alkaline environments, the solubility may be higher. Therefore, the pH value of the solution needs to be adjusted according to the specific needs and conditions when the drug is formulated and used.
(Product link1:https://www.bloomtechz.com/synthetic-chemical/api-researching-only/articaine-hcl-powder-cas-23964-57-0.html
(Product link2:https://www.bloomtechz.com/synthetic-chemical/api-researching-only/articaine-hydrochloride-powder-cas-23964-57-0.html)
Articaine HCl has anesthetic and analgesic properties. Its molecular structure is as follows:
1. Hydrogen and ionic bonds:
There are hydrogen bonds and ionic bonds in the molecule of Articaine HCl. Hydrogen bonds are weak bonds formed by hydrogen atoms in Articaine and electronegative atoms in other molecules (such as oxygen, nitrogen, etc.). These hydrogen bonds facilitate the interaction between Articaine and other molecular structures, such as drug binding to receptors. At the same time, the cationic moieties in Articaine HCl form ionic bonds with the anionic moieties such as chloride ions.
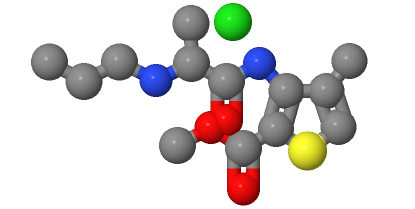
2. Structural features:
The molecular structure of Articaine HCl can be analyzed from two aspects: the organic part and the cationic part.
- Organic section:
The organic part of Articaine HCl consists of a benzene ring, a thiophene ring, a methylpropylaminocarboxyl group, and an alkyl group. A methyl group and a propylaminocarboxyl group are attached to the benzene ring. There is a methyl group attached to the thiophene ring. The alkyl group is attached to a carbon atom on the thiophene ring. These organic groups endow Articaine HCl with pharmaceutical activity and biological properties.
- Cationic part:
Articaine HCl is the hydrochloride salt formed by the reaction of Articaine with hydrochloric acid. It contains a positively charged Articaine cation and a chloride ion (Cl-). This cationic moiety is responsible for interacting with the anesthetic target sites, thereby producing a local anesthetic effect.
3. Three-dimensional structure:
Articaine HCl molecules contain chiral carbon atoms, so there are two enantiomers: (S)-Articaine HCl and (R)-Articaine HCl. These two isomers are mirror images in spatial configuration and have the same chemical composition, but their chemical and biological properties may differ slightly.
Articaine HCl is a local anesthetic drug that produces an anesthetic effect and provides pain relief through local application. Pharmacokinetics describes the process of absorption, distribution, metabolism and excretion of drugs in the body.
1. Absorption:
Articaine HCl is mainly absorbed through topical anesthesia and infiltration anesthesia of local tissues. It penetrates quickly through nerve tissue and other tissues and reaches the site of action quickly. Absorption is rapid, typically reaching peak concentration within minutes.

2. Distribution:
Articaine HCl is widely distributed in the body, including nervous tissue, muscle tissue and other tissues. It can cross the blood-brain barrier and enter the central nervous system. The larger volume of distribution (Vd) of Articaine HCl may be related to its lipophilicity.
3. Metabolism:
Articaine HCl is metabolized in the liver into several metabolites. The main metabolic pathway is the conversion of Articaine HCl to articainic acid methyl ester and other metabolites through the action of esterases such as cholinesterase in the liver. These metabolites may be further metabolized by enzyme systems in the liver.
4. Excretion:
Articaine HCl and its metabolites are mainly excreted through the kidneys and excreted through urine. About 80-90% of the dose is excreted in the urine in unmetabolized form. The excretion half-life (t1/2) of unmetabolized Articaine HCl in urine is approximately 1.5 to 2.5 hours.
5. Influencing factors:
Several factors can affect the pharmacokinetics of Articaine HCl, including individual differences, age, sex, liver function, renal function, and interactions with other drugs. Individual differences may lead to differences in the absorption and clearance of Articaine HCl in different patients. Age and gender can affect drug metabolism and excretion. Hepatic and renal insufficiency can affect the rate of metabolism and excretion of Articaine HCl. Interactions with other drugs may alter the pharmacokinetics of Articaine HCl.
|
|
|
Articaine HCl is a drug widely used in dental procedures and local anesthesia. It belongs to the amide class of local anesthetics and has unique pharmacological properties compared to other commonly used local anesthetics such as lidocaine and procaine.
1. Pharmacological mechanism:
The pharmacological effects of Articaine HCl are mainly realized by blocking the sodium channel on the nerve cell membrane. It blocks the conduction of nerve impulses by inhibiting the entry of sodium ions into nerve cells, thereby producing a local anesthetic effect. Compared with traditional local anesthetics, Articaine HCl has a stronger affinity for sodium channels, faster onset and longer duration of action.
2. Narcotic effects:
The main anesthetic effects of Articaine HCl include local anesthesia, infiltration anesthesia and topical anesthesia. The local anesthetic effect is achieved by direct injection of Articaine HCl into the target area, commonly used in various dental procedures and treatments. The infiltration anesthesia effect is achieved by injecting Articaine HCl into the target area around the local tissue, suitable for minor surgeries and treatments. The topical anesthetic effect is achieved by spraying or smearing Articaine HCl on the mucosal surface, which is commonly used for topical anesthesia of the gums and oral mucosa.
3. Pharmacokinetic characteristics:
Articaine HCl is characterized by rapid absorption and rapid anesthetic effect. It penetrates rapidly through tissues and reaches peak concentration within minutes. Articaine HCl has a high bioavailability of about 90%. It has a low protein binding rate of only 20%, which means that it exists in the blood in a free form. Articaine HCl is mainly metabolized by the liver, converted into multiple metabolites, and excreted through the kidneys.
4. Safety and adverse reactions:
Articaine HCl is generally considered a safe and effective local anesthetic. It has relatively few adverse effects, and most of them are temporary and reversible. Common adverse reactions include local irritation, numbness, muscle tremors, dizziness, nausea and vomiting. Severe allergic and neurotoxic reactions are very rare.
5. Uses and applications:
Articaine HCl is widely used in dental procedures and treatments. It can be used in tooth extraction, root canal treatment, restoration and periodontal surgery etc. Articaine HCl may also be used in combination with other drugs such as vasoconstrictors and epinephrine to prolong the duration of local anesthesia and enhance hemostasis.
6. Special population considerations:
For special populations such as children, the elderly, and pregnant women, the use of Articaine HCl requires special consideration and caution. When administering local anesthesia to children, the dose should be adjusted according to age and weight, and adverse reactions should be closely monitored. The elderly and pregnant women need to be especially careful when using Articaine HCl and must use it under the guidance of a doctor.



