The molecular structure of S-Allyl-L-cysteine (link:https://www.bloomtechz.com/synthetic-chemical/additive/s-allyl-l-cysteine-cas-49621-03-6.html) can be determined by analyzing its composition and chemical bonds. The molecular formula of S-Allyl-L-cysteine is C6H11NO2S, and the chemical structure is as follows:
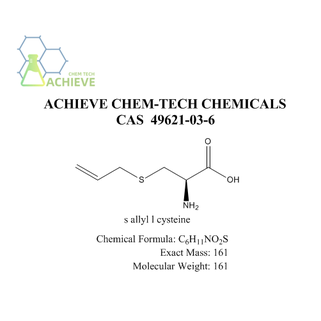
S-Allyl-L-cysteine consists of several functional groups:
1. Cysteine group: Cysteine group is composed of one cysteine molecule. It contains a carboxyl group (-COOH) and an amino group (-NH2), which are linked by an amide bond on a carbon atom.
2. Allyl substituents: Allyl substituents are composed of one allyl molecule. It consists of three carbon atoms forming a continuous chain structure. There is a double bond (C=C) on the carbon atom in the middle, and a hydrogen atom is attached to the carbon atoms on both sides.
3. Sulfur atom: The sulfur atom (S) is located on the carbon atom of the cysteine group and is connected to the carbon atom by a single bond. Sulfur atom is an important component of S-Allyl-L-cysteine, which plays an important role in chemical and biological activities.
By analyzing the molecular structure of S-Allyl-L-cysteine, its chemical properties and possible reaction pathways can be understood. The cysteine group in it gives it the characteristics of an amino acid, including the ability to undergo condensation or addition reactions with other molecules. The allyl substituent makes it have certain reactivity and special stereo configuration.
In addition, the molecular structure of S-Allyl-L-cysteine also determines its physical properties, such as solubility, polarity and molecular weight. These physical properties have important implications for understanding and using the compound for applications in medicine, food and other fields.
It should be noted that the molecular structure described above is a simplified representation. In order to more accurately understand the molecular structure characteristics of S-Allyl-L-cysteine, it may be necessary to use experimental techniques such as X-ray diffraction, nuclear magnetic resonance, etc. for detailed analysis. structural analysis and confirmation.
S-Allyl-L-cysteine (S-allyl-L-cysteine) is an organic compound whose name relates to its molecular structure and chemical properties.
1. Molecular structure: The molecule of S-Allyl-L-cysteine is composed of multiple parts, including a cysteine group and an allyl substituent. Among them, "S" means that the sulfur atom in cysteine is located at the S position of the chiral center, and it has two forms of stereoisomers R- and S-. Whereas "Allyl" means an allyl substituent and refers to an allyl group consisting of three carbon atoms.
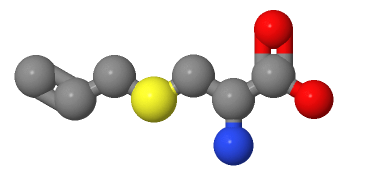
2. Group priority: According to regulations, the naming priority of the chiral center is higher than that of the substituent. Therefore, when naming, it is necessary to first determine the naming of the cysteine group.
3. Cysteine group naming: Cysteine is a natural amino acid, and its naming follows the common naming rules of amino acids. In the nomenclature of the cysteine group, "cysteine" means glycine, denoting the carboxyl and amino moieties therein.
4. Naming of the allyl group: after the naming of the cysteine group, the naming of the allyl substituent needs to be added. The allyl group is composed of three carbon atoms. According to the IUPAC naming rules, it can be called "prop-2-enyl", which means a single-bonded alkyl group composed of three carbon atoms, in which there is a double bond on C2.
5. Combination naming: Combine the cysteine group and the allyl substituent together to obtain the complete compound name S-Allyl-L-cysteine. Among them, "S" indicates that the sulfur atom is located at the S position of the chiral center, "Allyl" indicates an allyl substituent, and "L" indicates a left-handed property.
 S-Allyl-L-cysteine (S-Allyl-L-cysteine) is a thioamino acid with unique chemical reactivity properties.
S-Allyl-L-cysteine (S-Allyl-L-cysteine) is a thioamino acid with unique chemical reactivity properties.
1. Oxidation reaction: S-Allyl-L-cysteine is a compound that is easily oxidized. In air or in the presence of oxygen, S-Allyl-L-cysteine can be oxidized to S-Allyl-L-cysteine sulfoxide and S-Allyl-L-cysteine sulfone. These oxidation products may play an important role in the storage and processing of garlic, and also affect the pharmacological activity of garlic.
2. Chelation reaction: Since S-Allyl-L-cysteine contains a thiol functional group and an amine functional group, it can form a coordination bond and undergo a chelation reaction with metal ions. The complexes formed by S-Allyl-L-cysteine and metal ions may affect the stability and activity of metal ions in vivo.
3. Carboxylation reaction: S-Allyl-L-cysteine can be converted into its related ester compounds through carboxylation reaction. For example, reaction with an anhydride can form the corresponding ester.
4. Amino metabolism reactions: The amine groups in S-Allyl-L-cysteine can participate in various amino metabolism reactions. For example, it can be converted into derivatives such as S-Allyl-L-cysteine sulfenic acid and S-Allyl-L-cysteine sulfenic acid.
5. Adjacent group reaction: The allyl functional group of S-Allyl-L-cysteine can participate in different types of reactions through adjacent group reactions. For example, it can undergo an addition reaction with an electrophile, a Michael addition reaction with an enzyme-catalyzed substrate, and the like.
6. Alkylation reaction: S-Allyl-L-cysteine can undergo alkylation reaction with some electrophilic reagents (such as alkylating reagents) to form corresponding alkyl derivatives.
In general, S-Allyl-L-cysteine, as a thioamino acid, has various chemical reactivity properties. These reactions involve reaction types such as oxidation, chelation, carboxylation, amino metabolism, proximity group, and alkylation. These reactions provide an important basis for further research on the biological activity, metabolic pathway and pharmacological effects of S-Allyl-L-cysteine.
The development history of S-Allyl-L-cysteine (S-Allyl-L-cysteine) can be traced back to the research on the active ingredient in garlic (Allium sativum).
1. The medicinal value of garlic: Garlic has been widely used in food seasoning and traditional herbal medicine throughout human history. Garlic has been recognized for its various health and medicinal effects since ancient times. Garlic contains many bioactive compounds, including sulfur compounds and non-sulfur compounds.
2. Extraction and identification of active ingredients: In the 1960s, scientists began to study the active ingredients in garlic. Through extraction and separation techniques, the researchers identified a series of biologically active compounds, including sulfur compounds, phenols, and polysaccharides.
3. Discovery of S-Allyl-L-cysteine: S-Allyl-L-cysteine is a sulfur compound found in garlic. In 1964, Japanese scientists such as Suzuki isolated S-Allyl-L-cysteine from garlic for the first time, and found that it had certain pharmacological activity.
4. Pharmacological research: Subsequent research revealed a variety of health effects of S-Allyl-L-cysteine. It is believed to have functions such as antioxidant, antitumor, anti-inflammatory, antibacterial, lipid-lowering and immune regulation. These findings have aroused the interest of the scientific community in further research on S-Allyl-L-cysteine.

5. Mechanism research: Through in vitro and in vivo experiments, researchers began to explore the molecular mechanism of S-Allyl-L-cysteine. Studies have shown that S-Allyl-L-cysteine can exert its pharmacological effects through a variety of ways, including scavenging free radicals, regulating signal pathways, affecting gene expression and improving cell apoptosis.
6. Medical application: The pharmacological activity of S-Allyl-L-cysteine makes it have a wide application potential in the medical field. Studies have shown that S-Allyl-L-cysteine can be used for the treatment and prevention of diseases such as cardiovascular disease, cancer, neurodegenerative disease, liver disease and diabetes.
7. Pharmaceutical products: As a natural product, S-Allyl-L-cysteine has been used in some pharmaceutical products. For example, some garlic-derived medicines or health supplements contain S-Allyl-L-cysteine as an active ingredient.
8. Further research: Although S-Allyl-L-cysteine has attracted extensive research interest, there are still many aspects that need to be further explored. Including its pharmacokinetics, toxicology, safety and effectiveness of clinical research, etc.
In general, S-Allyl-L-cysteine, as the active ingredient in garlic, was discovered and received extensive attention in the past decades of research. Its pharmacological activity and potential medical application make it a compound with important research value and development potential. Future research will continue to deeply explore the mechanism, application fields and interactions of S-Allyl-L-cysteine with other compounds.

