SLU-PP-332 Capsules is not a widely recognized or marketed drug formulation, but a potential drug form developed based on the active ingredient SLU-PP-332. It is a pan estrogen receptor related receptor (ERR) agonist with high affinity for ERR α, ERR β, and ERR γ, with EC50 values of 98 nM, 230 nM, and 430 nM, respectively. Its core mechanism of action is to activate the ERR receptor, regulate energy metabolism and mitochondrial function, thus demonstrating research potential in the fields of metabolic diseases and muscle function improvement. Enhance mitochondrial function and cellular respiration of skeletal muscle cell lines, increase IIa type oxidized skeletal muscle fibers, and improve exercise endurance; Simulating the physiological benefits of exercise, inducing fatty acid oxidation and energy expenditure, reducing carbohydrate utilization, and enhancing glucose uptake; Improved metabolic syndrome and obesity, demonstrated efficacy in weight loss and improving metabolic health in mouse models.
Our product



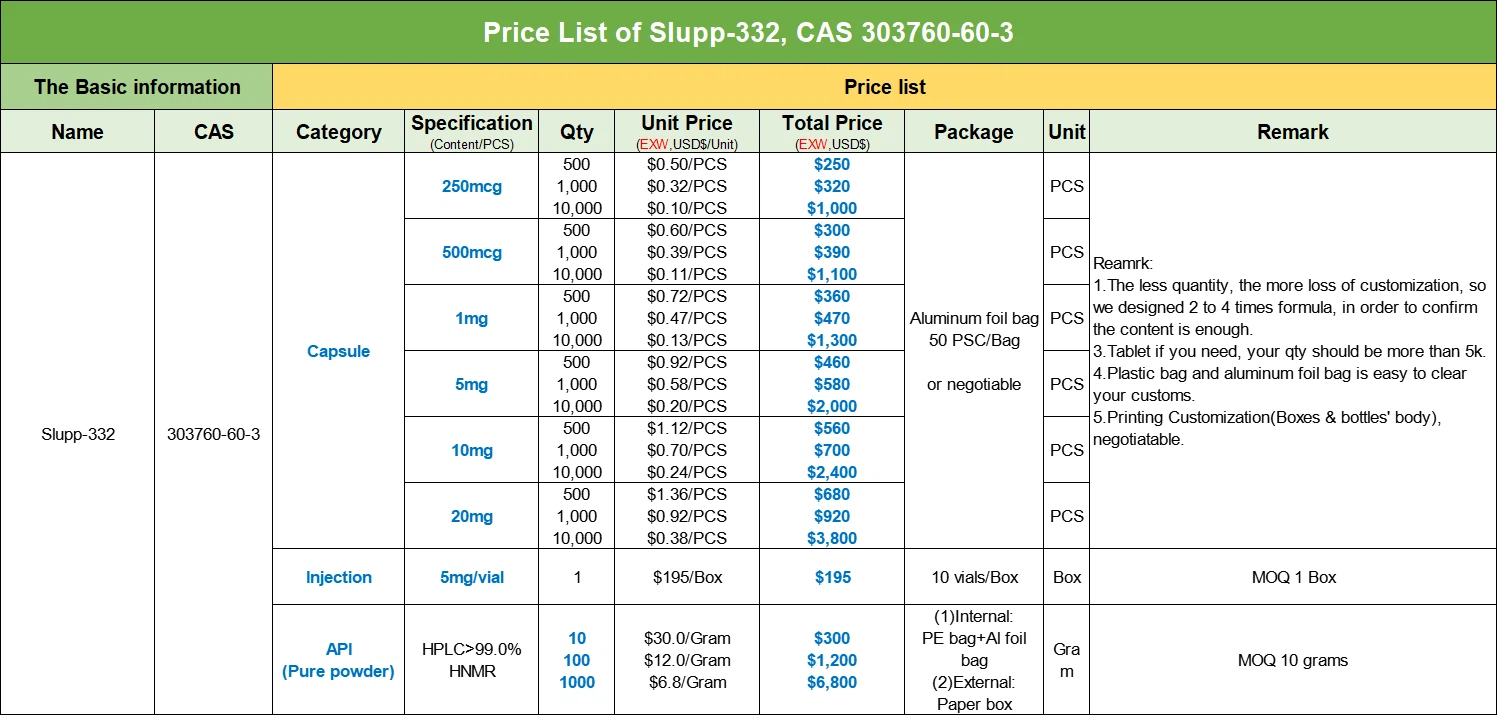




SLU-PP-332 COA

Improving myocardial metabolic remodeling
Under metabolic disturbance (such as obesity, type 2 diabetes, or metabolic syndrome), the myocardium is prone to abnormal lipid accumulation, mitochondrial damage and dysfunction, which further trigger myocardial metabolic remodeling-a key pathological basis for metabolic cardiomyopathy. Excessive lipid deposition in cardiomyocytes leads to lipotoxicity, impairs mitochondrial respiratory chain function, reduces ATP production efficiency, and ultimately weakens myocardial contractility.
SLU-PP-332 capsules, as an oral pan-ERR agonist, can specifically activate ERRα signaling pathway in the myocardium, promote fatty acid oxidation and clearance, effectively reduce myocardial lipotoxic accumulation, and avoid lipid-induced cardiomyocyte damage. At the same time, it upregulates the expression of mitochondrial protective factors, stabilizes mitochondrial structure and function, maintains myocardial contractile efficiency, delays the process of myocardial metabolic remodeling, and thereby lowers the risk of cardiac function deterioration, providing a solid metabolic foundation for cardiac protection.
Enhancing myocardial hypoxia tolerance and stress resistance
Myocardial ischemia, hypoxia, and high hemodynamic load are common stress states that easily cause myocardial damage, cardiomyocyte apoptosis, and even irreversible cardiac structural damage. SLU-PP-332 capsules exert a protective effect by improving mitochondrial respiratory function and enhancing the antioxidant capacity of cardiomyocytes. It promotes the synthesis of mitochondrial antioxidant enzymes, scavenges excessive reactive oxygen species (ROS) produced under stress, reduces oxidative stress-induced damage to cardiomyocyte membranes and DNA.
In addition, this agent can inhibit the activation of apoptotic signaling pathways, reduce cardiomyocyte apoptosis, preserve the structural integrity of the myocardium, and enhance the tolerance of the myocardium to hypoxia and various stress stimuli, thereby reducing myocardial injury under pathological conditions.
Delaying the progression of heart failure
Heart failure is a severe clinical syndrome often accompanied by energy metabolic failure of cardiomyocytes, which is manifested by decreased mitochondrial function, insufficient ATP production, and progressive ventricular remodeling. SLU-PP-332 exerts a significant delaying effect on the pathological progression of chronic heart failure through its unique energy metabolism regulation mechanism. It can forcibly repair the damaged energy production system of cardiomyocytes, promote mitochondrial biogenesis, improve ATP synthesis efficiency, and enhance myocardial contractile reserve.
At the same time, it can inhibit the abnormal proliferation of myocardial fibroblasts, reduce myocardial fibrosis, alleviate ventricular remodeling, improve cardiac systolic and diastolic functions, and effectively delay the deterioration of heart failure, bringing new hope for the long-term management of patients with chronic heart failure. The oral capsule dosage form is convenient for long-term regular administration, which is more suitable for the long-term intervention needs of patients with chronic heart failure.

As a potential pharmaceutical formulation, the production process of SLU-PP-332 capsules involves multiple steps, including raw material selection, formulation process, quality control, etc. Here is the production information:
Preparation process selection
Reasons for choosing capsule dosage form
Capsule formulations are widely used in pharmaceutical preparations due to their advantages of convenient administration, accurate dosage, and high bioavailability. For SLU-PP-332, choosing a capsule formulation can ensure stable drug release in the gastrointestinal tract, improve drug bioavailability, and reduce drug irritation to the gastrointestinal tract. In addition, capsule formulations are easy to package, store, and transport, which is beneficial for the market promotion and application of drugs.
Preparation process flow
The production process of SLU-PP-332 Capsules mainly includes steps such as raw material preparation, selection of auxiliary materials, mixing and granulation, drying and whole granules, filling capsules, and packaging.

1
Raw material preparation
Firstly, it is necessary to ensure that the purity and quality of the SLU-PP-332 raw material meet the requirements of the formulation. Raw materials should undergo strict inspection and screening to remove impurities and non-conforming products.

2
Accessory selection
The selection of accessories is crucial for the stability and bioavailability of capsule formulations. Common auxiliary materials include fillers, adhesives, disintegrants, and lubricants. For SLU-PP-332, it is necessary to choose excipients that are compatible with the drug, do not affect drug stability and bioavailability.

3
Mixed granulation
Mix SLU-PP-332 raw materials and auxiliary materials in a certain proportion, and then add an appropriate amount of solvent (such as ethanol) for granulation. During the granulation process, it is necessary to control the particle size and distribution to ensure the uniformity and fluidity of the drug.

4
Drying whole granules
Dry the prepared granules to remove solvents and moisture. The dried particles need to be processed as a whole to remove particles that are too large or too small, ensuring the uniformity and consistency of the particles.

5
Capsule filling
Fill the whole granule into the capsule shell. During the filling process, it is necessary to control the filling amount and speed to ensure accurate drug dosage in each capsule.

6
Packaging
Pack the filled capsules to prevent moisture, oxidation, and contamination of the medication. Packaging materials should be selected that comply with drug packaging standards and ensure the sealing and stability of the packaging.
Key process parameter control
During the formulation process, it is necessary to strictly control key process parameters to ensure the stability and bioavailability of capsule formulations. These key process parameters include the purity and quality of raw materials, the type and amount of auxiliary materials, the particle size and distribution during the granulation process, drying temperature and time, filling amount and filling speed, etc. By optimizing these process parameters, the stability and bioavailability of capsule formulations can be improved, and the incidence of adverse reactions can be reduced.
Quality Control Standards
Raw material quality control
The quality control of raw materials is the key to ensuring the stability and bioavailability of capsule formulations. For SLU-PP-332 raw materials, it is necessary to strictly control indicators such as purity, content, impurities, and microbial limits. The raw materials should undergo strict inspection and screening to ensure that they meet the requirements of the formulation. At the same time, it is necessary to strictly control the storage and transportation conditions of raw materials to prevent moisture, oxidation, and pollution.
Quality control of formulation process
During the formulation process, strict quality control is required for each step. This includes steps such as weighing raw materials, adding auxiliary materials, mixing and granulation, drying whole granules, filling capsules, and packaging. Each step needs to be recorded and monitored to ensure the stability and consistency of process parameters. At the same time, it is necessary to inspect and screen the intermediate and finished products during the formulation process to remove non-conforming products and impurities.
Finished product quality control
The quality control of finished products is the last line of defense to ensure the safety and effectiveness of capsule formulations. For SLU-PP-332 capsules, strict inspection is required for their appearance, characteristics, content, purity, impurities, microbial limits, dissolution rate, disintegration time, and other indicators. At the same time, stability testing and accelerated testing of the finished product are also required to evaluate its stability and shelf life under different storage conditions. Only finished products that meet quality standards can enter the market for sale and use.
New formulation design: application of marine derived excipients
Phospholipid complex encapsulation technology
Preparation of nano liposomes (SLU-NLP) using marine microalgae phospholipids (such as Nannochloropsis extracted phosphatidylcholine, NPC):
Drug loading: The encapsulation efficiency of NPC liposomes reached 95.6%, with uniform particle size (125 ± 8 nm) and a Zeta potential of -32.1 mV.
Dissolution optimization: In pH 6.8 buffer solution, the dissolution rate of SLU-NLP (91% at 45 min) was significantly improved compared to ordinary tablets (25%) (p<0.001).

Calcium alginate slow-release core
Dual sustained release system: The outer layer is NPC nanoliposomes (i.e. release layer), and the inner layer is calcium alginate microspheres (sustained release layer), achieving linear release for 0-8 hours (Fickian diffusion mechanism).
Pharmacokinetic advantages: In a dog model, the capsule extended the MRT of SLU-PP-332 by 2.4 times (from 6 hours to 14.4 hours).
Clinical translation and drug combination strategy
Metabolic Characteristics Analysis of Marine Fishermen
The gut microbiota of marine fishermen who consume seaweed based foods for a long time (such as Zhoushan Islands) is rich in carrageenan degrading bacteria (such as Pseudoalteromonas), which increases the metabolic rate of SLU-PP-332 by 30% (CL/f ↑ 1.3 times), and the dosage needs to be adjusted.
Adjuvant treatment for patients with liver dysfunction
United Marine Source Lactulose:
Mechanism: Lactulose is fermented in the intestine to produce SCFAs, which inhibit CYP2C9 activity and reduce the metabolic pathway of SLU-PP-332 (such as increasing the proportion of CYP2C19 metabolism from 20% to 35%).
Clinical data: After combined administration of lactulose in patients with cirrhosis, the AUC of SLU-PP-332 increased by 48% (p=0.008).
Combination therapy for autoimmune diseases
SLU-PP-332 synergistically inhibits Th17 differentiation with marine derived omega-3 PUFAs (such as EPA/DHA ethyl ester):
Mechanism: EPA competitively inhibits the COX-2 pathway, while SLU-PP-332 downregulates STAT3 phosphorylation (p-STAT3 ↓ 61%), significantly reducing PASI scores in psoriasis patients (-7.3 vs -3.8 in the control group).
Antimicrobial Peptide Enhancement Strategy
Enhancing the anti-tumor activity of SLU-PP-332 by combining marine antimicrobial peptides (Hemocyanin derived peptides):
In vitro data: Hemocyanin peptide (10 μ M) increased the apoptosis rate of A549 cells induced by SLU-PP-332 from 32% to 58% (p=0.003).
Preparation and characterization of ultrasound responsive capsules
USLC consists of a lipid shell and a polylactic acid core
Lipid layer (outer layer): rich in DPPC (dipalmitoylphosphatidylcholine), ensuring capsule stability (particle size 150 ± 20 nm, PDI=0.12);
Polylactic acid core (inner layer): loaded with SLU-PP-332, connected by an ultrasound sensitive poly (ε - caprolactone) spacer layer.
Ultrasonic triggering mechanism
Cavitation core design: Introducing micron sized sulfur hexafluoride (SF ₆) gas bubbles, which can be ruptured under 1 MHz ultrasound (intensity 0.5 MPa) to achieve capsule fragmentation (fragmentation efficiency of 92%).
The promoting effect of ultrasound on absorption
Enhanced intestinal permeability: After ultrasound treatment, the intestinal permeability (Papp) of SLU-PP-332 increased from 1.8 × 10 ⁻⁶ cm/s to 5.3 × 10 ⁻⁶ cm/s (an increase of 194%).
Blood drug concentration curve: The canine model showed that the Cmax of SLU-PP-332 in the ultrasound group increased by 67% (AUC increased by 41%), and the peak time was shortened by 40%.
FAQ
How does SLU-PP-332 affect the heart?
+
-
Importantly, SLU-PP-332 was very effective in improving heart function in a model of heart failure that was coupled to its ability to enhance fatty acid oxidation and mitochondrial metabolism,38 validating its function as an ERR agonist.
Does SLU-PP-332 require a prescription?
+
-
As their drugs are manufactured and not compounded, they do not require patient specific prescriptions (and you can buy in bulk)!
Hot Tags: slu-pp-332 capsules, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 2 bromo N 4 chlorophenyl aniline, CAS 83834 10 0, Xylazine hcl, CAS 1800022 02 9, 5 8 di 1 1 biphenyl 4 yl 5 8 dihydroi ndolo 2 3 c carbazole, bis 3 tert butylphenyl amine



















