Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of cyclophosphamide monohydrate powder cas 6055-19-2 in China. Welcome to wholesale bulk high quality cyclophosphamide monohydrate powder cas 6055-19-2 for sale here from our factory. Good service and reasonable price are available.
Cyclophosphamide monohydrate powder, Molecular formula: C7H17Cl2N2O3P, molecular weight: about 279.1 g/mol, CAS 6055-19-2. Exists in the form of colorless or white crystalline solids. Its appearance is usually in powder or crystalline form. Moderate solubility in water. At room temperature, approximately 50-100 grams of Cyclophosphamide monohydrate can be dissolved per 100 milliliters of water. In addition, it can also be dissolved in ethanol, ether, dichloromethane and other organic solvents. Relatively stable under dry, dark, and low temperature conditions. However, it is sensitive to light, heat, and moisture, so it should be stored in sealed containers and avoid prolonged exposure to light or high temperatures. It is an electrophilic compound that can be hydrolyzed to 4-hydroxy Cyclophosphamide and Malondialdehyde. It is an unstable compound that is metabolized in the body and converted into its active form, thereby exerting anti-tumor activity. It is an important Nitrogen mustard compound, commonly used in cancer treatment.

|
|
|
|
Chemical Formula |
C7H15Cl2N2O2P |
|
Exact Mass |
260 |
|
Molecular Weight |
261 |
|
m/z |
260 (100.0%), 262 (63.9%), 264 (10.2%), 261 (7.6%), 263 (4.8%) |
|
Elemental Analysis |
C, 32.20; H, 5.79; Cl, 27.16; N, 10.73; O, 12.26; P, 11.86 |
The molecular structure of Cyclophosphamide monohydrate consists of the following main parts:
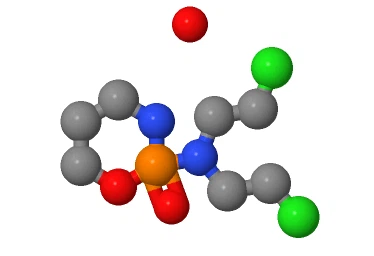
Cyclophosphamide group:
Cyclophosphamide is a compound containing cyclophosphamide group, which is formed by connecting a ring containing phosphorus and nitrogen with a carbonyl group (C=O). The Cyclophosphamide group endows Cyclophosphamide with anti-tumor and immunosuppressive activities.
Oxazole ring:
the ring of Cyclophosphamide group in Cyclophosphamide is also connected with an oxazole ring. The oxazole ring is composed of carbon, nitrogen and oxygen atoms, which together with the Cyclophosphamide group forms the core structure of Cyclophosphamide.
Monohydrate:
Cyclophosphamide monohydrate is a monohydrate, indicating that it binds to a water molecule (H2O). This water molecule typically interacts with functional groups in the Cyclophosphamide molecule in the form of hydrogen bonds.
The molecular structure of Cyclophosphamide monohydrate can be determined by various experimental and analytical methods, such as X-ray diffraction, nuclear magnetic resonance and mass spectrometry. These methods can provide information about the relative position of atoms in molecules and the connection mode of functional groups, thus revealing the molecular structure of Cyclophosphamide monohydrate.

Cyclophosphamide monohydrate powder (CTX), as a classic alkylating chemotherapy drug, has become an indispensable core drug in the fields of tumor treatment, immune suppression, and organ transplantation due to its unique "latent" design (acting after metabolic activation in the body) since its emergence in the 1950s.
1. Core drugs for hematological malignancies
It is a first-line chemotherapy drug for malignant lymphoma (including Hodgkin lymphoma and non Hodgkin lymphoma), often combined with doxorubicin, vincristine, and prednisone to form CHOP regimen, with a cure rate of 60% -70%. In the treatment of leukemia, it has significant therapeutic effects on acute lymphocytic leukemia (ALL) and chronic lymphocytic leukemia (CLL) by inducing cell cycle arrest (G1/S phase) and DNA cross-linking damage. Clinical data shows that adding cyclophosphamide to induction therapy for ALL patients increases the complete remission rate from 70% to 85% and reduces the recurrence rate by 30%. In addition, cyclophosphamide is a cornerstone drug for multiple myeloma (MM), and its combination with bortezomib and dexamethasone (VCD regimen) can prolong median survival to over 5 years.


2. Combination chemotherapy core for solid tumor treatment
In the field of solid tumors, it is often combined with other drugs to form a combination chemotherapy regimen. For example:
Breast cancer: CAF scheme is composed with doxorubicin and fluorouracil. Postoperative adjuvant chemotherapy can increase the 5-year survival rate from 50% to 70%.
Ovarian cancer: Combination with paclitaxel (TC regimen) is the standard first-line treatment for advanced ovarian cancer, with an objective response rate of 75% and a median progression free survival extended to 18 months.
Small cell lung cancer: When combined with etoposide and cisplatin in an EP regimen, the median survival of extensive stage small cell lung cancer is extended from 8 months to 12 months.
Soft tissue sarcoma: The combination with doxorubicin (AI regimen) increases the 5-year survival rate to 60% for Ewing's sarcoma and rhabdomyosarcoma.
3. Precise application of special tumor types
Has unique therapeutic effects on certain special tumors:
Testicular tumors: As the core components of the BEP regimen (bleomycin, etoposide, cyclophosphamide), the cure rate of early testicular germ cell tumors exceeds 95%.
Neuroblastoma: The combination of topotecan and cisplatin (TC regimen) increased the 3-year survival rate of high-risk neuroblastoma from 30% to 55%.
Retinoblastoma: In combination with intravenous chemotherapy and ophthalmic artery intervention chemotherapy (IAC), it can penetrate the blood retinal barrier, increasing the eye protection rate from 40% to 70%.

Immunosuppressive therapy: a powerful tool for regulating autoimmune diseases

1. Classic treatments for autoimmune diseases
By inhibiting T lymphocyte proliferation and antibody production, it has become the standard treatment for various autoimmune diseases:
Systemic lupus erythematosus (SLE): For patients with severe lupus nephritis (type IV), monthly intravenous pulse therapy (0.5-1g/m ²) can reduce the risk of renal function deterioration by 50% and increase the 5-year survival rate from 60% to 85%.
Rheumatoid arthritis (RA): For refractory RA that is ineffective with traditional DMARDs, the combination of methotrexate can increase the ACR50 remission rate from 20% to 45%.
Vasculitis: The standard induction therapy for ANCA associated vasculitis (cyclophosphamide+corticosteroids) can achieve a disease remission rate of 80% and a 40% reduction in recurrence rate.
2. Prevention of organ transplant rejection reactions
By inhibiting T cell activation, the risk of acute rejection after organ transplantation can be reduced. In kidney transplantation, a triple immunosuppressive regimen consisting of calcineurin inhibitor (tacrolimus) and anti metabolite (mycophenolate mofetil) can increase the one-year survival rate of transplanted kidneys from 75% to 90%. In addition, it can also be used for pre-treatment before bone marrow transplantation, by clearing host immune cells and reducing the incidence of graft-versus-host disease (GVHD).
3. Local treatment of skin immune diseases
Cyclophosphamide eye drops are used for the treatment of erosive corneal ulcers after corneal transplantation. By inhibiting local immune response, the corneal healing rate is increased from 30% to 70%, and the survival time of the transplant is extended to more than 2 years.

Application expansion in special clinical scenarios

1. Treatment of hematological diseases
Idiopathic thrombocytopenic purpura (ITP): For ITP patients who are resistant to glucocorticoids or have relapsed, oral cyclophosphamide (1-2mg/kg/d) can restore platelet count to normal with a rate of 60% and a long-term remission rate of 30%.
Aplastic anemia: Combined with anti thymocyte globulin (ATG) immunosuppressive therapy, the 5-year survival rate of severe aplastic anemia can be increased from 50% to 70%.
2. Immune regulation of infectious diseases
In severe infections with immune overactivation (such as sepsis and COVID-19), low-dose cyclophosphamide (0.5g/m ²) can reduce the incidence of multiple organ dysfunction syndrome (MODS) by inhibiting excessive inflammatory response. Clinical studies have shown that it can reduce the 28 day mortality rate of sepsis patients from 40% to 28%.
3. Exploratory treatment for rare diseases
Scleroderma: For rapidly progressing diffuse cutaneous scleroderma, cyclophosphamide monohydrate powder intravenous pulse therapy can improve the skin sclerosis score by 30% and reduce the risk of interstitial lung disease progression by 40%.
Pemphigus vulgaris: Combined treatment with glucocorticoids can reduce the steroid dosage of bullous pemphigus vulgaris by 50% and lower the recurrence rate by 35%.

Drug interactions and combination therapy strategies

1. The synergistic effect of enhancing chemotherapy efficacy
The combination with doxorubicin can enhance self metabolic activation by upregulating the expression of cytochrome P450 enzyme (CYP2B6), resulting in a 2-fold increase in the concentration of phosphoramide nitrogen mustard in tumor cells and a 40% increase in anti-tumor effect. In addition, the combination with paclitaxel can increase the sensitivity of ovarian cancer cells to paclitaxel by threefold by inhibiting the expression of P-glycoprotein and reducing the efflux of paclitaxel by tumor cells.
2. Synergistic regulation of immune suppression
In organ transplantation, the combination with mycophenolate mofetil can inhibit T cell activation through different mechanisms: it mainly acts on the early stage of T cell proliferation, while mycophenolate mofetil inhibits purine synthesis. The synergy of the two reduces the incidence of acute rejection from 25% to 10%.
3. Interactions in drug metabolism
Cyclophosphamide is a substrate of CYP3A4, and when combined with rifampicin (CYP3A4 inducer), it can reduce the blood concentration of cyclophosphamide by 50% and weaken its therapeutic effect; Combined use with ketoconazole (CYP3A4 inhibitor) can increase blood drug concentration by 2 times and increase the risk of toxicity. Therefore, close monitoring of blood drug concentration is necessary when using combination therapy.


The synthesis of Cyclophosphamide monohydrate usually involves three main steps: the introduction of oxazole ring, amidation reaction, and hydration reaction. The following is a description of the specific steps:
In the presence of oxazole Procaine (Proca ï n oxazole), oxazole ring was introduced into the compound 2-chloroethyl Sulfonamide. First, 2-chloroethyl Sulfonamide and oxazol Procaine undergo SN2 Substitution reaction to generate N - (2-chloroethyl sulfonyl) - N '- (4-oxazolyl) hydrazine. Subsequently, after deprotection reaction, the product was obtained after the introduction of the oxazole ring.
In the product obtained in the first step, N - (2-chloroethylsulfinyl) - N '- (4-oxazolyl) oxamide is formed by amidation with Sulfinic acid derivatives. This reaction usually uses di N-Methylformamide (DMF) as solvent, and Sulfinic acid derivatives and base catalysts, such as triethylamine, are added. After the reaction, the product is obtained through extraction and crystallization purification.
The product after amidation reacts with water to form Cyclophosphamide monohydrate. In this reaction, the product is dissolved in water and undergoes hydrolysis to form a crystalline substance with one water molecule.
It should be noted that the reagents and conditions used in the synthesis process of cyclophosphamide monohydrate powder need to be strictly controlled to ensure the purity and yield of the product. In addition, during the synthesis process, multiple crystallization and purification steps are required to obtain high-purity Cyclophosphamide monohydrate.
To sum up, the synthesis of Cyclophosphamide monohydrate includes the introduction of oxazole ring, amidation reaction, hydration reaction and other Committed step. Through these steps, the starting substance can be converted into the target product, resulting in Cyclophosphamide monohydrate. However, due to the complexity of the synthesis method, the process of synthesizing Cyclophosphamide monohydrate requires strict control of reaction conditions and purification process to ensure the quality and purity of the product.

Melting point:
The melting point of Cyclophosphamide monohydrate is approximately 48-52 degrees Celsius. This means that within this temperature range, it will transition from solid to liquid.
Molecular weight:
The molecular weight of Cyclophosphamide monohydrate is approximately 279.1 g/mol. Molecular weight refers to the total mass of atoms in a molecule.
Density:
The density of Cyclophosphamide monohydrate is approximately 1.27 g/cm ³. This value represents the mass per unit volume.
Refractive index:
The refractive index of Cyclophosphamide monohydrate is 1.572. The refractive index is a measure of the degree to which light deviates when passing through a material interface.
PH value:
The pH value of a Cyclophosphamide monohydrate solution is usually in the neutral range (about 7), but it depends on the concentration of the solution and preparation conditions.
Nature:
Cyclophosphamide monohydrate is an electrophilic compound that can be hydrolyzed to 4-hydroxy Cyclophosphamide and Malondialdehyde. It is an unstable compound that is metabolized in the body and converted into its active form, thereby exerting anti-tumor activity.
Hot Tags: cyclophosphamide monohydrate powder cas 6055-19-2, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, pure benzocaine powder, Procaine Hydrochloride, tetrahydrobiopterin synthesis, API Researching Only , tetracaine hcl powder, IPTG reagent






