Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of pralidoxime chloride powder in China. Welcome to wholesale bulk high quality pralidoxime chloride powder for sale here from our factory. Good service and reasonable price are available.
Pralidoxime Chloride Powder is an effective acetylcholinesterase (AChE) reactivation agent, belonging to the oxime class of compounds, mainly used for the treatment of organophosphate poisoning.

The appearance is usually yellow white crystalline powder or off white powder, with high solubility in water (640mg/ml at 25 ℃), and ultrasound assisted dissolution is required in DMSO. Pralidoxime Chloride can restore the activity of acetylcholinesterase, thereby alleviating these symptoms. This substance is an effective medication for treating organophosphate poisoning. It can reactivate acetylcholinesterase inhibited by organic phosphorus compounds, alleviate toxic symptoms such as breathing difficulties, muscle tremors, and pupil constriction. Multiple studies have shown that Pralidoxime Chloride has significant therapeutic effects in treating organophosphate poisoning. For example, reports have shown that in patients with organophosphate insecticide poisoning, after intramuscular injection of Pralidoxime Chloride, clinical symptoms of poisoning mostly disappear within 30 minutes to 1 hour, and blood cholinesterase activity returns to over 70% of normal values.
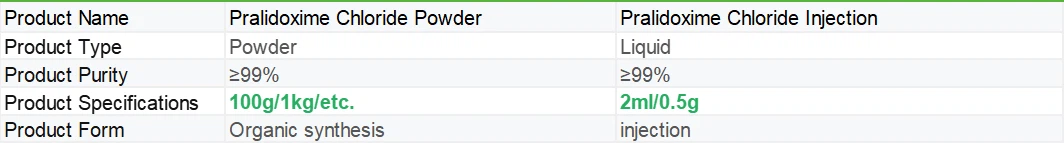
At the same time, our company not only provides pure powders, but also tablets and injections. If needed, please feel free to contact us at any time.
 |
 |

Additional information of chemical compound:
|
|
|
Pralidoxime Chloride COA


Pralidoxime Chloride Powder is an important pharmaceutical raw material, with the chemical name 2-pyridylaldehyde oxime methyl chloride, CAS number 51-15-0, molecular formula C7H9ClN2O, and molecular weight 172.61. This substance has a wide range of applications in the pharmaceutical field, especially playing a key role in the detoxification treatment of organophosphate poisoning. Here is a detailed analysis of its usage:
The pharmacological effects of Pralidoxime Chloride are mainly based on its properties as an activator of acetylcholinesterase (AChE). After binding with acetylcholinesterase, organic phosphorus compounds form phosphorylated acetylcholinesterase, causing the enzyme to lose its ability to hydrolyze acetylcholine, which in turn leads to the accumulation of acetylcholine in neural synapses and produces toxic symptoms. Pralidoxime Chloride removes the phosphoryl group from the enzyme through its oxime group's nucleophilic attack on the phosphate center of the binding nerve agent, restoring the activity of acetylcholinesterase, thereby decomposing acetylcholine and relieving poisoning symptoms. By binding with phosphorylated acetylcholinesterase, Pralidoxime Chloride can release the inhibition of acetylcholinesterase by organic phosphorus compounds, restoring the enzyme's ability to hydrolyze acetylcholine. The recovery of acetylcholinesterase activity helps to reduce the accumulation of acetylcholine in neural synapses, thereby alleviating toxic symptoms. By restoring acetylcholinesterase activity, Pralidoxime Chloride can improve nerve conduction function and promote normal functioning of the nervous system.

clinical application

Pralidoxime Chloride has shown significant therapeutic effects in clinical applications. Multiple studies have shown that it can effectively improve the symptoms of patients with organophosphate poisoning, increase blood cholinesterase activity, and reduce mortality. In the treatment of acute organophosphate poisoning, Pralidoxime Chloride is often used in combination with drugs such as atropine. Atropine can counteract the accumulated acetylcholine in the body, while Pralidoxime Chloride restores the activity of acetylcholinesterase. The synergistic effect of the two can better alleviate the symptoms of poisoning. The usage and dosage of Pralidoxime Chloride Powder are adjusted according to the degree of poisoning.
Mild poisoning patients can receive medication through intramuscular injection or slow intravenous injection; Patients with moderate poisoning may require a larger initial dose and may require multiple doses or maintenance of medication; Severe poisoning patients require larger doses and more frequent administration. Pralidoxime Chloride has a significant effect on nicotine like symptoms caused by organophosphate poisoning, but a weaker effect on muscarinic symptoms, and a less significant effect on central nervous system symptoms. It has poor detoxification effect on acetylcholinesterase that has been "aged" by organic phosphate ester inhibition for more than 36 hours, and has no revival effect on acetylcholinesterase inhibited by chronic organophosphate insecticide poisoning.

Storage and stability
Pralidoxime Chloride Powder, as an important pharmaceutical raw material, is mainly used to treat organophosphate poisoning. Its storage and stability are crucial for ensuring the quality and efficacy of drugs. The following will elaborate on the storage and stability of Pralidoxime Chloride:
1.Storage conditions
Temperature control
Pralidoxime Chloride is sensitive to temperature and is typically recommended for storage at low temperatures. The generally recommended storage temperature is between -20 ° C and 0 ° C. Within this temperature range, the chemical stability of the drug is good and can effectively extend its shelf life. High temperature can accelerate the decomposition reaction of drugs, leading to reduced or even ineffective efficacy. Therefore, high temperature environments should be avoided during storage to ensure that the medication is under suitable temperature conditions.
Light control
Pralidoxime Chloride is sensitive to light, which may trigger photochemical reactions of the drug, leading to its degradation. Therefore, drugs should be stored in a dark environment, such as brown glass bottles or opaque containers, to reduce the impact of light on drug stability.
Humidity control
Excessive humidity may cause drugs to absorb moisture, thereby affecting their stability and solubility. Therefore, the storage environment should be kept dry to prevent drugs from getting damp. Desiccants or dehumidification devices can be used to control the humidity of the storage environment.
Packaging requirements
Medications should be packaged in a sealed manner to prevent the invasion of air, moisture, and microorganisms. Common packaging forms include aluminum foil bags, aluminum cans, cardboard boxes or cartons, etc. These packaging materials can effectively isolate the external environment and protect the stability of drugs. For drugs that require frequent use, it is recommended to divide them into small dose reserve solutions to avoid the impact of repeated freezing and thawing on drug stability. At the same time, the packaged reserve solution should also be stored under low temperature conditions and used up as soon as possible.
2.Stability
Chemical stability
Under suitable storage conditions, the shelf life of Pralidoxime Chloride is usually 2 to 3 years. However, the actual validity period may vary due to storage conditions, packaging materials, and other factors. Therefore, the expiration date of the medication should be checked before use to ensure that it is used within the expiration date. During storage, Pralidoxime Chloride may undergo decomposition reactions, producing impurities or degradation products. These decomposition products may affect the efficacy and safety of drugs. Therefore, it is very important to regularly conduct quality testing on drugs to ensure that they meet quality standards.
Physical stability
During storage, the appearance of the medication may change, such as darkening color, clumping, etc. These changes may indicate that the drug has undergone physical or chemical changes that have affected its stability and efficacy. Therefore, the appearance of the medication should be carefully observed before use, and any abnormalities should be promptly addressed. Pralidoxime Chloride should have good solubility so that it can be fully dissolved when preparing injections. However, improper storage conditions may lead to a decrease in drug solubility, affecting the effectiveness of formulation. Therefore, the solubility of the drug should be checked before preparing the injection to ensure that it meets the requirements.
3.Influencing factors
Temperature
Temperature is one of the main factors affecting the stability of Pralidoxime Chloride. High temperature can accelerate the decomposition reaction of drugs, leading to reduced or even ineffective efficacy. Therefore, temperature should be strictly controlled during storage and transportation to ensure that the medication is under suitable temperature conditions.
Light
Light is also an important factor affecting drug stability. Long term exposure to light may trigger photochemical reactions of drugs, leading to their degradation. Therefore, light avoidance measures should be taken during storage and transportation to reduce the impact of light on drug stability.
Humidity
Excessive humidity may cause drugs to absorb moisture, thereby affecting their stability and solubility. Therefore, during storage and transportation, the environment should be kept dry to prevent drugs from getting damp.
Packaging materials
The choice of packaging materials also has a significant impact on the stability of drugs. High quality packaging materials can effectively isolate the external environment and protect the stability of drugs. Therefore, when choosing packaging materials, factors such as sealing, light avoidance, and moisture resistance should be considered.
4. Measures to improve stability
Optimize storage conditions
By strictly controlling storage temperature, lighting, and humidity conditions, the stability of Pralidoxime Chloride Powder can be effectively improved. For example, storing drugs in a low temperature, dark, and dry environment can prolong their shelf life and maintain their efficacy.
Improve packaging materials
Choosing high-quality packaging materials is also one of the important measures to improve drug stability. For example, using packaging materials with good sealing, light avoidance, and moisture resistance can effectively isolate the influence of the external environment on drugs and protect their stability.
Strengthen quality inspection
Regular quality testing of drugs is an important means to ensure their stability and efficacy. By detecting indicators such as appearance, solubility, and content of drugs, quality issues can be promptly identified and corresponding measures taken.
Standardize usage and operation
When using Pralidoxime Chloride, strictly follow the instructions in the manual. For example, suitable solvents and preparation methods should be used when preparing injection solutions to avoid degradation or deterioration of drugs due to improper handling.
Hot Tags: pralidoxime chloride powder, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, tetracaine hcl powder, Sapropterin Hydrochloride, IPTG reagent, 99 9 pure lidocaine powder, API Researching Only , articaine hydrochloride powder










