Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of 6-nitro-1-diazo-2-naphthol-4-sulfonic acid cas 50412-00-5 in China. Welcome to wholesale bulk high quality 6-nitro-1-diazo-2-naphthol-4-sulfonic acid cas 50412-00-5 for sale here from our factory. Good service and reasonable price are available.
6-Nitro-1-Diazo-2-Naphthol-4-Sulfonic Acid, yellow or yellow-brown crystal, is easily oxidized in the air and the color gradually deepens. It begins to decompose when heated to 203 °C, and there is a sharp decomposition phenomenon during combustion. It is very easy to dissolve in water, and its saturated aqueous solution can be recrystallized in hot water. Adding hydrochloric acid or sulfuric acid can also precipitate crystallization. It is a dye intermediate for the manufacture of acid-mordant black dyes and metal complex dyes, such as acid-mordant black T, black A, etc.6-Nitrate is an important dye intermediate, which is mainly used to manufacture high-grade reactive dyes. So its market prospect is broad. With the progress of society, cotton textiles dyed with direct, vulcanized, and dispersed dyes in the past will be gradually dyed with reactive dyes.

|
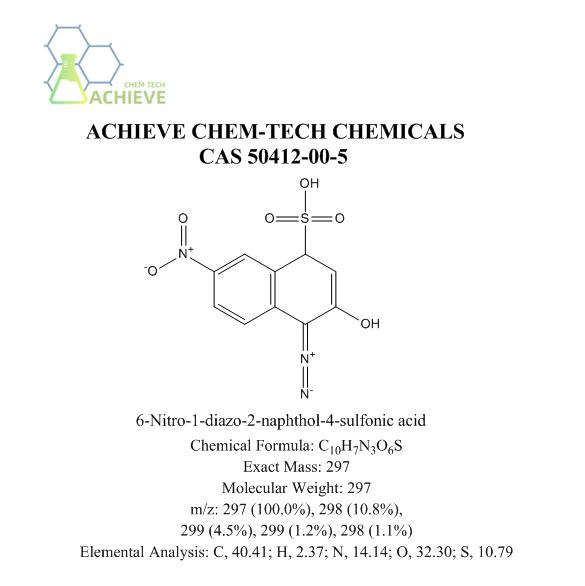
Chemical Formula |
C10H7N3O6S |
|
Exact Mass |
297 |
|
Molecular Weight |
297 |
|
m/z |
297 (100.0%), 298 (10.8%), 299 (4.5%), 299 (1.2%), 298 (1.1%) |
|
Elemental Analysis |
C, 40.41; H, 2.37; N, 14.14; O, 32.30; S, 10.79 |
Content: ≥ 70%, Main content (HPLC): ≥ 95%, Unacceptable: ≤ 0.1%, Free acid: ≤ 2%, Iron ion: ≤ 30mmp
|
|
|
6-Nitro-1-Diazo-2-Naphthol-4-Sulfonic Acid (NDAS) is an organic compound with special structure and properties.
The chemical formula of NDAS is C10H5N3O7S, with a relative molecular weight of 311.23 g/mol. It consists of a benzene ring and a naphthalene ring, with nitro (NO2) and diazo (N=N) functional groups attached to the naphthalene ring, as well as sulfonic acid (SO3H) functional groups on the benzene ring.
NDAS has special optical properties, which can absorb ultraviolet and visible light. Under ultraviolet light irradiation, it can take place photocatalysis reaction and produce living intermediates with certain photosensitivity.


6-Nitro-1-diazo-2-naphthol-4-sulfonic acid (abbreviated as NDAS) is an important organic synthesis intermediate with a special structure and diversified functions. Its molecular structure simultaneously contains multiple reactive functional groups such as diazo group (-N=N-), nitro group (-NO₂), phenolic hydroxyl group (-OH) and sulfonic acid group (-SO₃H). The synergistic effect of these functional groups endows NDAS with excellent reaction activity and diverse functional properties, making it irreplaceable in various fields including dye synthesis, photosensitive material preparation, and chemical analysis and detection, while also laying a foundation for its expansion into emerging fields. The core application directions are elaborated in detail below.
Application in Dye Synthesis
NDAS is an indispensable core intermediate in the synthesis of naphthalene-based dyes. Relying on its unique functional group structure, it can precisely regulate key indicators such as chromogenic properties and color fastness of dyes through a series of organic reactions including substitution and coupling, thereby synthesizing a variety of naphthalene-based dyes with both practical value and aesthetic effect.
From the perspective of reaction mechanism, the diazo group in the NDAS molecule is the core active site for realizing coupling reactions, while the nitro group regulates the electron cloud density of the molecule through electronic effects, further enhancing the selectivity and efficiency of coupling reactions. In the actual synthesis process, NDAS can undergo coupling reactions with aromatic amines and phenolic compounds of different structures to form aromatic chromophores with conjugated systems.

Such conjugated systems can effectively absorb specific wavelengths of light in the visible range, thus presenting bright and diverse colors, covering red, orange, yellow, blue and other color systems. The synthesized naphthalene-based dyes not only have bright and full colors, but also possess excellent dyeing properties, including good hydrophilicity, high affinity for fiber materials, and outstanding fastness to sunlight and washing.
In specific application scenarios, such dyes are widely used in the dyeing of natural fibers such as cotton, linen, silk and wool, as well as synthetic fibers such as polyester and nylon, and can also be used for coloring products such as paper, leather and printing ink. For example, in cotton fabric dyeing, the acidic naphthalene-based dyes synthesized with NDAS as the intermediate can combine with the hydroxyl groups on the surface of cotton fibers through ionic bonds to achieve uniform dyeing, and the dyed fabrics have a soft hand feel and long-lasting colorfastness.

In ink production, the coupling products based on NDAS, with good tinting strength and dispersibility, can be used to prepare high-gloss and high-covering power printing inks to meet the needs of packaging printing, book and periodical printing and other fields. In addition, by modifying the molecular structure of NDAS, dyes with special functions such as fluorescent dyes and thermosensitive dyes can be synthesized, further expanding its application scope in the field of special dyeing.
Application in Photosensitive Materials
NDAS and its derivatives have important applications in the preparation of photosensitive materials and the research and development of photosensitizers. Their unique structure makes them highly responsive to ultraviolet light, enabling effective regulation of the photochemical behavior of materials and providing core material support for key photochemical processes such as photolithography and photopolymerization.
In the modification of basic photosensitive materials, adding NDAS to polymer materials such as epoxy resin and acrylic resin can significantly improve the sensitivity and response speed of the materials to ultraviolet light. This is because the diazo group in the NDAS molecule undergoes decomposition reaction under ultraviolet irradiation, generating nitrogen gas and reactive free radicals.

These reactive free radicals can initiate cross-linking or degradation reactions of polymer materials, thereby realizing photoinduced modification of the materials. For example, adding an appropriate amount of NDAS to UV-curable coatings can accelerate the curing speed of the coatings under ultraviolet irradiation, shorten the curing time, and at the same time improve the hardness, wear resistance and corrosion resistance of the cured coating, optimizing the construction performance and application effect of the coating.
In the research and development of special photosensitizers, by structurally modifying NDAS to synthesize derivatives with specific structures and functions, special photosensitizers suitable for different photochemical processes can be prepared. In the field of photolithography, photosensitizers based on NDAS are widely used in the preparation of photoresists, which are core materials in high-end fields such as integrated circuit manufacturing and micro-nano processing.

When ultraviolet light irradiates the surface of the photoresist, the NDAS-based photosensitizers in it undergo photodecomposition reaction, changing the solubility of the photoresist. Then through processes such as development and etching, the pattern on the photomask can be accurately transferred to the substrate material, realizing the precise preparation of micro-nano structures. In the field of photopolymerization, NDAS derivatives, as photosensitizers, can initiate polymerization reactions of monomers under ultraviolet irradiation, which are used in fields such as 3D printing and printing plate making, and can improve the efficiency of polymerization reactions and the performance stability of products. In addition, such photosensitizers can also be applied to emerging fields such as photodynamic therapy and photocatalytic degradation, showing broad application prospects.
Application in Chemical Analysis
Relying on its unique chemical reaction characteristics, NDAS can be used as an analytical reagent, indicator, chromogenic reagent and selective coordination reagent for metal ions in the field of chemical analysis and detection, providing precise and efficient solutions for material composition detection, content determination and metal ion separation and enrichment.
In the detection of organic compounds, NDAS exhibits a strong vic-diketone diazo test reaction with para-aminocompounds and nitroso compounds. This specific reaction makes it an exclusive reagent for the detection of such compounds. When NDAS comes into contact with para-aminocompounds or nitroso compounds, a specific chromogenic reaction occurs to generate products with characteristic colors.

By observing the color change of the products and combining with analytical methods such as spectrophotometry, qualitative detection and quantitative determination of such compounds can be realized.This method has the advantages of high sensitivity, strong selectivity and simple operation, and is widely used in environmental monitoring, food testing, pharmaceutical analysis and other fields. For example, in the detection of trace nitroso compounds in environmental water samples, the chromogenic reaction of NDAS can be used to quickly and accurately determine the content of nitroso compounds in water samples, providing reliable data for environmental quality assessment. In pharmaceutical analysis, it can be used to detect para-amino impurities produced during drug synthesis to ensure the quality and safety of drugs.
In the analysis of metal ions, NDAS can serve as a selective coordination reagent for metal ions. Functional groups such as phenolic hydroxyl groups and sulfonic acid groups in its molecule can form stable complexes with various metal ions. The complexes formed by different metal ions and NDAS have different characteristic colors and stability. Using this property, separation, enrichment and determination of metal ions can be achieved.
For example, in water quality monitoring, NDAS can form characteristic complexes with copper ions, iron ions, zinc ions and other ions in water, and the content of each ion can be determined separately by spectrophotometry. In the metallurgical industry, the selective coordination effect of NDAS with specific metal ions can be used to realize the separation and enrichment of target metal ions and impurity ions, improving the purity of metal products.
In addition, NDAS can also be used as an indicator in titration analysis, indicating the titration endpoint through color changes, improving the accuracy and convenience of titration analysis, and is widely used in routine analysis experiments such as acid-base titration and complexometric titration.

We are the supplier of 6-Nitro-1-Diazo-2-Naphthol-4-Sulfonic Acid.
Remark: BLOOM TECH(Since 2008), ACHIEVE CHEM-TECH is the subsidiary of us.


The synthesis methods of NDAS mainly include nitration reaction, diazotization reaction of naphthylcyclodiphenol, and sulfonation reaction. First, under appropriate conditions, 2-Naphthol is nitrated with nitric acid to obtain 2-naphthol-6-nitro, and then 2-naphthol-6-diazo group is obtained through phenol Diazonium compound tautomerism. Finally, 2-Naphthol 6-diazo group is converted into product by sulfonation Acylation reaction.
The following is a detailed description of various synthesis methods for NDAS.
First, 2-Naphthol is reacted with nitric acid to produce 2-naphthol-6-nitro. The reaction is generally carried out at low temperature, and can be realized by dropping concentrated nitric acid into the mixture of 2-Naphthol. After the reaction, 2-Naphthol -6-nitro was obtained.
2-naphthol-6-nitro is reacted with nitrite such as Sodium nitrite or Copper(II) nitrate to obtain 2-naphthol-6-diazo group by diazotization reaction. The reaction is often carried out under acidic conditions, and nitrite can be gradually added to the reaction system at low temperature. After the reaction is completed, 2-Naphthol -6-diazo group is obtained.
The 2-Naphthol 6-diazo group is reacted with sulfonation reagent (such as sulfonyl chloride), and the sulfonation reaction is carried out to obtain product. This reaction is often carried out under alkaline conditions and can be achieved by adding sulfonation reagents dropwise to the reaction system. After the reaction is completed, a 6-Nitro-1-Diazo-2-Naphthol-4-Sulfonic Acid is obtained.
In summary, the synthesis methods of NDAS include nitration reaction, diazotization reaction, and sulfonation reaction. Through these reactions in turn, 2-Naphthol can be converted into it. This synthesis method can efficiently prepare NDAS and can be used for large-scale production.
Hot Tags: 6-nitro-1-diazo-2-naphthol-4-sulfonic acid cas 50412-00-5, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, BENZENE D6, 2 6 Pyridinedicarboxylic acid, 2 5 Dihydroxybenzaldehyde, squaric acid treatment, N N N Trimethylethylenediamine, 3 Nitrobenzonitrile