Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of fmoc-glu(obzl)-oh cas 123639-61-2 in China. Welcome to wholesale bulk high quality fmoc-glu(obzl)-oh cas 123639-61-2 for sale here from our factory. Good service and reasonable price are available.
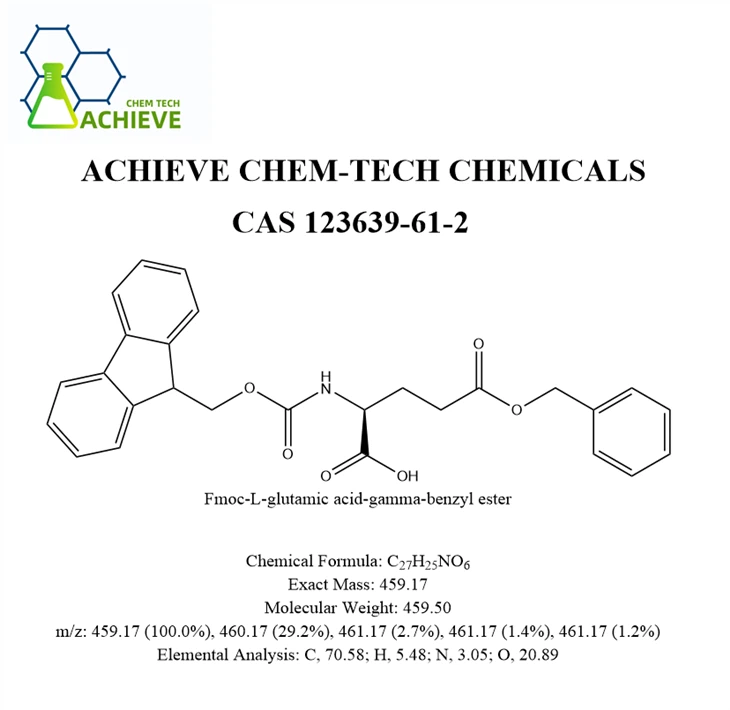
Fmoc-Glu(OBzl)-OH is a crucial dual-protected glutamic acid derivative in solid-phase peptide synthesis. Its structure design ingeniously achieves precise control over the side chain and main chain functional groups of glutamic acid. This molecule employs fluorene methoxycarbonyl (Fmoc) as the protection group for the α-amino group, which can be efficiently removed through mild piperidine treatment, ensuring the successive extension of the synthesis process. Meanwhile, the side chain carboxyl group is permanently protected by benzyl ester (OBzl). This strategy effectively prevents intramolecular cyclization that may form pyroglutamic acid or unnecessary side chain cross-linking during peptide chain assembly. This dual protection feature makes it a key building block for constructing complex peptide segments containing glutamic acid residues, especially indispensable in applications where the side chain carboxyl group needs to be retained for subsequent functionalization (such as preparing antigen peptides, dendrimeric molecules, or metal chelating structures). After the final peptide chain synthesis is completed, global deprotection under strong acid conditions (such as HF or TFMSA) can simultaneously remove the OBzl group, releasing the active glutamic acid side chain, providing a molecular basis for the development of diagnostic probes, vaccine immunogens, and protein mimics.

 |
 |
|
Chemical Formula |
C7H3ClN2O2 |
|
Exact Mass |
182 |
|
Molecular Weight |
183 |
|
m/z |
182 (100.0%), 184 (32.0%), 183 (7.6%), 185 (2.4%) |
|
Elemental Analysis |
C, 46.05; H, 1.66; Cl, 19.42; N, 15.34; O, 17.53 |


Peptide Synthesis
Solid-Phase Peptide Synthesis (SPPS):
- Serves as an important intermediate in solid-phase peptide synthesis, a widely used technique for the synthesis of peptides and proteins.
- The Fmoc group, being a protecting group for the amino group, facilitates efficient coupling reactions between amino acids during the synthesis process.
- After the desired sequence is assembled, the Fmoc group can be removed under mild conditions, revealing the free amino group for further reactions or purification steps.
Pharmaceutical Industry
- The pharmaceutical industry heavily relies on the synthesis of bioactive peptides and proteins for the development of novel drugs and therapeutics.
- As a key intermediate, plays a crucial role in the synthesis of such compounds, enabling the production of compounds with specific biological activities.


Research Applications
- In biochemical and chemical biology research, it serves as a valuable tool for studying protein-protein interactions, enzyme mechanisms, and other biological processes.
- Its use in the synthesis of labeled or modified peptides allows researchers to probe these processes with greater specificity and sensitivity.
Protein Engineering
- Protein engineering, which involves the rational design and modification of proteins to enhance their properties or confer new functions, often requires the synthesis of specific peptides and protein fragments.
- With its unique structural features, can be incorporated into such designs to achieve the desired properties.


Fmoc-L-glutamic acid-gamma-benzyl ester (CAS No. 123639-61-2) does have a wide range of applications in the field of biomedicine, especially in peptide synthesis and drug development. The following are specific examples to illustrate its application in drug molecules and diagnostic reagents:
Use as drug molecules for the treatment of diseases
Protective groups and chemical modification: It is mainly used in protein synthesis to protect the amino group of L-glutamic acid through its specific chemical properties, which facilitates subsequent chemical modification. This protecting group is critical in peptide synthesis, allowing researchers to precisely control and tune the structure and function of peptides.
DRUG DEVELOPMENT: Peptides synthesized via Fmoc-L-glutamate-gamma-benzyl ester have specific biological activities and can be used to treat a wide range of diseases. For example, some peptide drugs are able to target specific receptors or enzymes, thereby inhibiting or promoting certain biochemical processes in organisms for the purpose of treating diseases. Although examples of peptide drugs specifically synthesized using it for the treatment of diseases may not be widely disclosed due to the stage of research or patent protection, significant progress has been made in the application of peptide drugs in the fields of oncology, neurological diseases, and infectious diseases.
Use as diagnostic reagents for disease diagnosis
Immunochemical analysis: with the wide application of monoclonal antibody preparation technology, immunoturbidimetric assay, enzyme immunoassay, fluorescence immunoassay, luminescence immunoassay, and other automated detection techniques are developing rapidly. peptides synthesized by it can be used as the key components of these immunoassay techniques for the diagnosis of diseases. For example, peptides can be used as antigens that bind to specific antibodies to form antigen-antibody complexes, thereby detecting the presence of specific pathogens or biomarkers in a sample.
Biochemical separation and analysis: Peptides can also be used as probes or markers for biochemical separation and analysis. By utilizing the interaction of peptides with specific biomolecules (e.g., affinity, enzyme activity, etc.), the separation and purification of complex biological samples can be achieved. In addition, peptides can be used as signaling molecules in biosensing and imaging technologies to provide powerful support for early diagnosis of diseases.
In summary, the peptides synthesized from it have a wide range of applications in the field of biomedicine, not only as drug molecules for the treatment of diseases, but also as diagnostic reagents for the diagnosis of diseases. These applications not only improve the therapeutic effect of diseases, but also provide new means and methods for early diagnosis and prevention of diseases.

Fmoc-L-glutamic acid-gamma-benzyl ester, as an important raw material for peptide synthesis, also shows great potential in the field of new material development. The following are examples of its application in the preparation of biocompatible materials and smart materials:
Biocompatible materials
Peptide coatings:
Peptides synthesized from it can be coated on the surface of medical implants, such as pacemakers and artificial joints, to enhance their biocompatibility.
These peptide coatings can reduce the inflammatory response between the implant and the host tissue and promote stable fixation of the implant and tissue healing.
Tissue Engineering:
In tissue engineering, peptides can be used as cell culture substrates or inducing factors to promote cell growth, differentiation and tissue formation.
Peptides synthesized from it can be used to construct tissue engineering scaffolds with specific functions, such as skin, bone, cartilage, etc., due to their specific structures and biological activities.
Smart Materials
Stimulus-responsive materials:
Fmoc-L-glutamic acid-gamma-benzyl ester synthesized peptides are responsive to specific stimuli (e.g., temperature, pH, light, etc.).
This responsiveness can be used to prepare smart materials such as self-repairing materials and shape memory materials. These materials are capable of undergoing specific physical or chemical changes when subjected to external stimuli to fulfill specific application requirements.
Molecular Recognition and Sensing:
Peptides can also be used as components for molecular recognition and sensing to detect specific molecules or biomarkers in organisms.
Peptides synthesized from it can be modified and adapted to achieve highly selective and sensitive detection of specific molecules. This detection capability can be used to prepare biosensors or bioimaging probes, which can provide powerful support for early diagnosis and treatment of diseases.
In summary, Fmoc-L-glutamic acid-gamma-benzyl ester has a wide range of applications in the field of new material development. By utilizing its advantages in the preparation of biocompatible materials and smart materials, it can provide new solutions and technical support in the fields of medical treatment, biotechnology and environmental protection. Meanwhile, with the continuous development and improvement of peptide synthesis technology, it is believed that more new peptide materials based on it will be developed and applied in real life in the future.
Adverse reactions
Fmoc-Glu(OBzl)-OH (fluorenylmethoxycarbonyl-L-glutamic acid - γ - benzyl ester) is a derivative of glutamic acid, with the chemical formula C ₂ ₇ H ₂ ₅ NO ₆, molecular weight 459.49 g/mol, CAS number 123639-61-2. Its structure is formed by the esterification of L-glutamic acid with benzyl group via gamma carboxyl group, and the introduction of Fmoc (fluorenylmethoxycarbonyl) protecting group, resulting in a white to off white solid powder. The solubility of this compound in DMSO is as high as 200 mg/mL (435.27 mM), but its solubility in water has not been clearly reported. The storage conditions should be strictly controlled at -20 ℃ (powder state can be stored for 3 years) or 4 ℃ (2 years), and storage in solvents should be -80 ℃ (6 months) or -20 ℃ (1 month) to avoid decomposition or loss of activity.
Classification and mechanism of adverse reactions
Acute toxic reaction
Contact with Fmoc-Glu(OBzl)-OH powder may cause local irritation reactions, manifested as skin erythema, itching, or mucosal congestion. The mechanism may be related to non-specific inflammatory reactions triggered by direct contact with compounds, especially when the skin barrier is damaged, the penetration of compounds is enhanced, activating mast cells to release inflammatory mediators such as histamine. Inhaling powders or aerosols may cause coughing, difficulty breathing, or laryngeal edema. Animal experiments have shown that high concentration exposure can cause damage to alveolar epithelial cells, manifested as increased exudate in the alveolar cavity and infiltration of inflammatory cells. Oral ingestion may cause nausea, vomiting, or diarrhea, especially when the compound is not completely dissolved, and its granular form may mechanically damage the gastrointestinal mucosa. In addition, the residue of DMSO solvent may exacerbate gastrointestinal irritation, as DMSO itself has a permeation enhancing effect, which may increase the direct damage of compounds to intestinal mucosa.
Chronic toxic reactions
Long term exposure may lead to elevated liver enzymes (ALT, AST), and in severe cases, jaundice or liver cell necrosis may occur. Compounds are metabolized in the liver through the cytochrome P450 enzyme system, and high-dose exposure may exceed the liver's metabolic capacity, leading to the accumulation of intermediate metabolites. The reactive oxygen species (ROS) generated during the metabolic process may attack the lipids in the liver cell membrane, triggering a chain reaction of lipid peroxidation and damaging the integrity of the cell membrane. Compounds or their metabolites may bind to liver cell proteins as haptens, forming complete antigens and triggering autoimmune reactions. Animal experiments have shown that long-term high-dose exposure may lead to vacuolar degeneration of renal tubular epithelial cells, manifested as increased urinary protein or elevated blood creatinine. The mechanism may be related to the direct toxic effects of compounds or their metabolites on renal tubular epithelial cells when they are actively secreted and excreted through the renal tubules. Very few case reports suggest that long-term exposure may cause headaches, dizziness, or peripheral neuropathy, manifested as limb numbness or sensory abnormalities. Low molecular weight compounds may cross the blood-brain barrier and interfere with neurotransmitter synthesis or release. Compounds may inhibit the activity of neuronal mitochondrial complexes, leading to a decrease in ATP production and triggering neuronal apoptosis.
Allergic Reaction
A very small number of individuals may develop IgE mediated allergic reactions to Fmoc Glu (OBzl) - OH or its metabolites, manifested as rash, urticaria, or anaphylactic shock. The mechanism is that the compound acts as an antigen to activate B cells, which differentiate into plasma cells and secrete specific IgE antibodies. When exposed again, it triggers degranulation of mast cells and releases inflammatory mediators such as histamine. Delayed type allergic reactions may manifest as contact dermatitis, typically occurring 24-72 hours after exposure, through a T-cell mediated cellular immune response that activates macrophages to release cytokines and trigger local inflammation.
Adverse reactions in special populations
Animal experiments have shown that high-dose exposure may lead to delayed embryonic development or fetal malformations, and the mechanism may be related to compounds interfering with embryonic protein synthesis through the placental barrier. After use by lactating women, compounds may be secreted through breast milk and have potential effects on infants. Children's liver and kidney function are not fully developed, and their metabolism and excretion abilities are weak, which may make them more prone to accumulation poisoning, manifested as growth retardation or precocious puberty (when exposed for a long time). The decline of liver and kidney function in elderly people and the decrease in drug metabolism enzyme activity may increase the risk of adverse reactions, especially hepatotoxicity and nephrotoxicity.
Hot Tags: fmoc-glu(obzl)-oh cas 123639-61-2, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, CAS 4786 80 5, 5 chloro 2 3 difluoroterephthalonitrile, 2 10 phenylanthracen 9 yl benzo d naphtho 2 3 b furan, 1 bromodibenzo b d furan, CAS 2173555 52 5, CAS 1548470 92 3