Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of glycolchitosan cas 123938-86-3 in China. Welcome to wholesale bulk high quality glycolchitosan cas 123938-86-3 for sale here from our factory. Good service and reasonable price are available.
Glycolchitosan is a chitosan derivative with hydrophilic ethylene glycol branches. Enhanced membrane permeability and leakage of Glycine xHarosoy63W cells. It is water-soluble, biocompatible, and biodegradable. It can inhibit the growth of Escherichia coli, Staphylococcus aureus, and Streptococcus pneumoniae, with MIC values of 4 μ g/mL, 32 μ g/mL, and<0.5 μ g/mL, respectively.
Molecular formula C24H47N3O16, CAS 123938-86-3. Chitosan is a high molecular weight compound composed of acetylated glucosamine units naturally present in crustaceans such as shrimp, crabs, and insects. It is obtained by deacetylation or enzymatic deacetylation of chitin under strong alkaline conditions. It is also a linear structure composed of a large number of glucosamine units and a small number of acetylated glucosamine units through 1,4-glycosidic bonds, and the only natural polysaccharide biopolymer compound with alkaline characteristics. Compared with chitosan, there are more polar amino groups that can be protonated in the molecular structure of chitosan. Therefore, while maintaining the good biodegradability, biocompatibility, and environmental friendliness of chitosan, its solubility in acidic media is also significantly improved compared to chitosan, and its application performance is also significantly improved.

|
|
|

Hydroxyethyl deacetylated chitosan, also known as Glycolchitosan, is a derivative of chitosan obtained through chemical modification, which introduces hydrophilic ethylene glycol branches into its structure. This modification not only improves the water solubility of chitosan, but also retains its original biocompatibility and biodegradability, thus demonstrating broad application prospects in the field of biology.
Has significant antibacterial activity and can inhibit the growth of various bacteria. Research has shown that it has highly effective inhibitory effects on common pathogenic bacteria such as Escherichia coli, S. aureus, and S. enteritidis, with minimum inhibitory concentrations (MICs) of 4 μ g/mL, 32 μ g/mL, and<0.5 μ g/mL, respectively.


This antibacterial property makes hydroxyethyl chitosan have potential application value in fields such as biomedicine, food preservation, and agriculture. For example, in the field of biomedicine, it can be used as an ingredient in wound dressings or antibacterial coatings to effectively prevent wound infections; In terms of food preservation, it can extend the shelf life of food and reduce food spoilage.
It has good biocompatibility and degradability, making it an ideal drug carrier material. It can load effective payloads such as drug molecules, genes, or photosensitizers into its interior or surface through physical encapsulation or chemical coupling, forming stable complexes or nanoparticles.

These complexes or nanoparticles have excellent stability and targeting properties in vivo, which can significantly improve the bioavailability and therapeutic efficacy of drugs. For example, hydroxyethyl chitosan nanoparticles (NPs) can achieve efficient targeted delivery in tumor tissues based on enhanced permeability and retention (EPR) effects, reducing the side effects of drugs on normal tissues.
The nanoparticles of this substance have also shown great potential in the field of cell imaging. Due to their low cytotoxicity and high biocompatibility, these nanoparticles can safely enter the interior of cells and achieve real-time, non-invasive monitoring of cells through fluorescence labeling or other imaging techniques.

This is of great significance for studying cell biology, disease mechanisms, and drug action mechanisms. In addition, hydroxyethyl chitosan nanoparticles can also serve as tracers for drug delivery systems, helping researchers monitor the distribution and metabolism of drugs in the body.
In the field of tissue engineering, it has been widely studied due to its good biocompatibility and degradability. It can serve as a scaffold material, providing a suitable microenvironment for cell growth and differentiation.

By binding with bioactive substances such as extracellular matrix proteins and growth factors, hydroxyethyl chitosan scaffolds can simulate the structure and function of natural tissues, promoting the repair and regeneration of damaged tissues. For example, in bone tissue engineering, hydroxyethyl chitosan scaffolds can guide bone cell growth and differentiation, promote fracture healing and bone defect repair.
In recent years, with the increasing demand for natural cosmetics, it has also received widespread attention in the field of cosmetics. As a natural moisturizer, antioxidant, and antibacterial agent, hydroxyethyl chitosan can significantly improve the performance and safety of cosmetics.
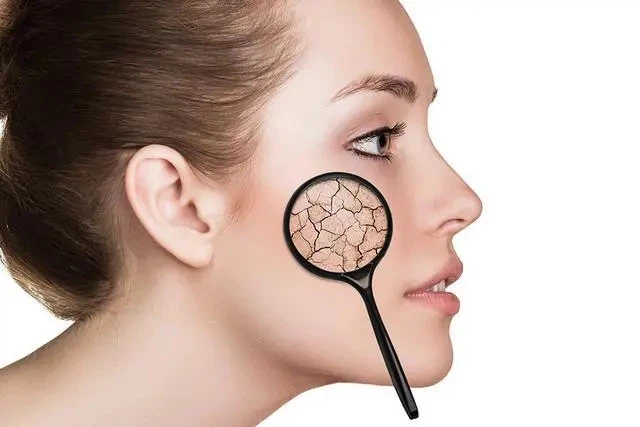

Its excellent moisturizing performance can help the skin lock in moisture, prevent dryness and the formation of fine lines; Its antioxidant properties can neutralize the damage of free radicals to the skin and delay aging; Its antibacterial properties can reduce the irritation and infection risk of cosmetics on the skin. Therefore, hydroxyethyl chitosan has broad application prospects in skincare, cosmetics, and oral care products.
In addition to the aforementioned applications, it can also serve as a substrate or additive for biomedical materials. For example, when preparing medical devices such as biomedical membranes, catheters, and artificial skin, adding an appropriate amount of hydroxyethyl chitosan can improve the biocompatibility and antibacterial properties of the materials.


When preparing biosensors, the specific recognition ability of hydroxyethyl chitosan can be utilized to achieve sensitive detection of biomolecules. In addition, hydroxyethyl chitosan can also be combined with other biomaterials such as collagen and hyaluronic acid to further enhance the performance and application range of the material.
In the field of environmental protection, it also has certain potential for application. Due to its excellent biodegradability and environmental friendliness, it can be used as a green material for wastewater treatment, soil remediation, and other applications.

For example, in wastewater treatment, hydroxyethyl chitosan can serve as an efficient adsorbent or flocculant to remove harmful substances such as heavy metal ions and organic pollutants from wastewater; In soil remediation, it can promote the activity and diversity of soil microorganisms, accelerate the degradation and transformation of harmful substances in the soil.

Preparation of Glycolchitosan (hydroxyethyl deacetylated chitosan):

Add an alkaline aqueous solution with a mass percentage concentration of 20-50% to chitosan in a mass ratio of 2.0-10:1.0. The alkaline aqueous solution is composed of a mixed base of sodium hydroxide, lithium hydroxide, and/or potassium hydroxide.
After thorough stirring and mixing, alkalize at -20~50 ℃ for 10~48 hours; Disperse the alkalized chitosan in isopropanol in a mass ratio of 1.0:10-100, and then stir at a constant temperature of 10-90 ℃ for 5-150 minutes;
Add a solution of bromoethanol, epoxyethane, and/or chloroethanol in isopropanol in a ratio of 0.1-20 of the amount of bromoethanol, epoxyethane, and/or chloroethanol to the amount of chitosan glucosamine units in the alkalized chitosan isopropanol system. After addition, react at a temperature of 10-100 ℃ for 2-48 hours;
Centrifuge or filter the reaction material, dissolve or disperse the obtained solid in water, and obtain an aqueous solution or slurry containing hydroxyethyl chitosan (hydroxyethyl deacetylated chitosan);
Evaporate and concentrate an aqueous solution or slurry containing hydroxyethyl chitosan (hydroxyethyl deacetylated chitosan) under negative pressure, then transfer the concentrated solution to a dialysis bag with a molecular weight cut-off of 500-50000, and dialyze in distilled or deionized water for 12-72 hours to remove inorganic salts and excess alkali from the concentrated solution;
After secondary concentration of the concentrated solution after dialysis, slowly add it to anhydrous ethanol and/or acetone in a volume ratio of 1.0:1.0-50 under stirring to precipitate hydroxyethyl chitosan (hydroxyethyl deacetylated chitosan);
Finally, filter and freeze dry the obtained solid material to obtain hydroxyethyl chitosan (hydroxyethyl deacetylated chitosan) with an average grafting degree of 5-300% glucosamine units.

Glycolchitosan is a high molecular weight compound composed of acetylated glucosamine units naturally present in crustaceans such as shrimp, crabs, and insects. Chitin is obtained by deacetylation or enzymatic deacetylation under strong alkaline conditions. It is also a linear structure composed of a large number of glucosamine units and a small number of acetylated glucosamine units through 1,4-glycosidic bonds, and is the only natural polysaccharide biopolymer compound with alkaline characteristics.
Compared with chitosan, there are more polar amino groups that can be protonated in the molecular structure of chitosan. Therefore, while maintaining the good biodegradability, biocompatibility, and environmental friendliness of chitosan, its solubility in acidic media is also significantly improved compared to chitosan, and its application performance is also significantly improved.
However, due to the molecular weight of chitosan being tens of thousands or even hundreds of thousands, and the presence of a large number of hydroxyl and amino groups in its structure, there are strong hydrogen bonding interactions between its molecular chains.
This hydrogen bonding often gives it good crystallinity, so its solubility in pure water and alkaline media is still not very good, which affects and limits its further application. Considering the shortcomings of chitosan in terms of water solubility, researchers have proposed methods such as grafting, catalytic hydrolysis, and chemical copolymerization to modify chitosan to improve its water solubility.
For example, carboxymethyl chitosan can be obtained by introducing carboxymethyl groups that can dissociate in water into the molecular structure of chitosan, hydroxyethyl chitosan (hydroxyethyl deacetylated chitosan) can be obtained by introducing polar hydroxyethyl groups that can destroy its crystallinity, 2-hydroxypropyl trimethylammonium chloride with good hydrophilicity can be introduced to obtain 2-hydroxypropyl trimethylammonium chloride chitosan, and chitosan can be oxidized and degraded into oligosaccharides with molecular weight below tens of thousands and crystallinity destroyed by hydrogen peroxide and other methods.
Due to the introduction of hydrophilic groups and the disruption of hydrogen bonding structures between chitosan chains or the decrease in molecular weight, these chitosan derivatives often exhibit good water solubility.

As a natural basic polysaccharide, chitosan exhibits biocompatibility, biodegradability, and antibacterial activity. However, it is only soluble in dilute acids and insoluble at physiological pH, which severely limits its application in biomedicine.Starting in the 1980s, the scientific community focused on hydrophilic modification of chitosan to improve solubility by introducing hydroxyl groups, polyethylene glycol, and other moieties. As a key water-soluble derivative, Glycolchitosan became a major research focus.
In the early 1990s, research teams from Japan and South Korea achieved the first breakthrough: using chitin as the starting material, hydroxyethylation with ethylene oxide was performed to introduce glycol side chains, followed by deacetylation with concentrated alkali to yield glycol chitosan for the first time.By adjusting the amount of ethylene oxide and reaction temperature, the degree of glycol substitution could be precisely controlled. The product was water-soluble over the entire pH range of 1–14, resolving the solubility limitation of chitosan.In 1992, its structure as hydroxyethylated chitosan was confirmed by ¹H NMR and FTIR spectroscopy, verifying that glycol groups were grafted onto both hydroxyl and amino sites of chitosan.
After 1995, its biosafety and application potential were systematically evaluated: it showed low cytotoxicity, enzymatic degradability, and retained the cationic amino groups, making it suitable for drug delivery systems and tissue engineering materials.From 2000 to 2005, researchers at Queen's University (Canada) optimized the synthetic process to improve the uniformity of substitution, promoting its application in gene delivery and anticancer drug carriers. It has since become a benchmark among water-soluble chitosan derivatives.
Architectural design & planning cepteur sint occaecat cupidatat proident, taken possession of my entire soul, like these sweet mornings of spring which I enjoy with my whole...Architectural design & planning cepteur sint occaecat cupidatat proident, taken possession od my entire soul, like these sweet mornings of spring which I enjoy with my whole Lorem ipsum dolor sit ament, consectetur adipisicing elit,sed do eiusmod tempor incididunt labore et dolore magna aliqua. it enim ad minim veniam.
FAQ
What is glycol chitosan?
+
-
Glycol chitosan is a derivative of chitosan that offers the advantages of biocompatibility and biodegradability, and glycol groups can enhance water solubility; therefore, it has become a potential candidate for drug delivery in many polymer carriers.
What is the drug chitosan used for?
+
-
It's used as medicine and in drug manufacturing. Chitosan is a fibrous substance that might reduce how much fat and cholesterol the body absorbs from foods. It also helps blood clot when applied to wounds.
Hot Tags: glycolchitosan cas 123938-86-3, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, Dithizone Reagent, 3 methyl 4 piperidone, CAS 1000869 26 0, CAS 1911641 83 2, 3 9H carbazol 9 yl 5 4 4 5 5 tetramethyl 1 3 2 dioxaborol an 2 yl benzonitrile, 4 amino 2 6 dimethylbenzonitrile












