Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of thioacetamide solution cas 62-55-5 in China. Welcome to wholesale bulk high quality thioacetamide solution cas 62-55-5 for sale here from our factory. Good service and reasonable price are available.
Thioacetamide, or TAA for short, is an organic compound with chemical formula C2H5NS, which is a white crystalline powder. Very slightly soluble in benzene and ether to form thioacetamide solution. It is mainly used in the production of catalysts, stabilizers, polymerization inhibitors, electroplating additives, photographic drugs, pesticides, dyeing aids and mineral processing agents, and also used as curing agents, cross-linking agents, rubber aids and pharmaceutical raw materials for polymers.

|
Chemical Formula |
C2H5NS |
|
Exact Mass |
75 |
|
Molecular Weight |
75 |
|
m/z |
75 (100.0%), 77 (4.5%), 76 (2.2%) |
|
Elemental Analysis |
C, 31.97; H, 6.71; N, 18.64; S, 42.67 |



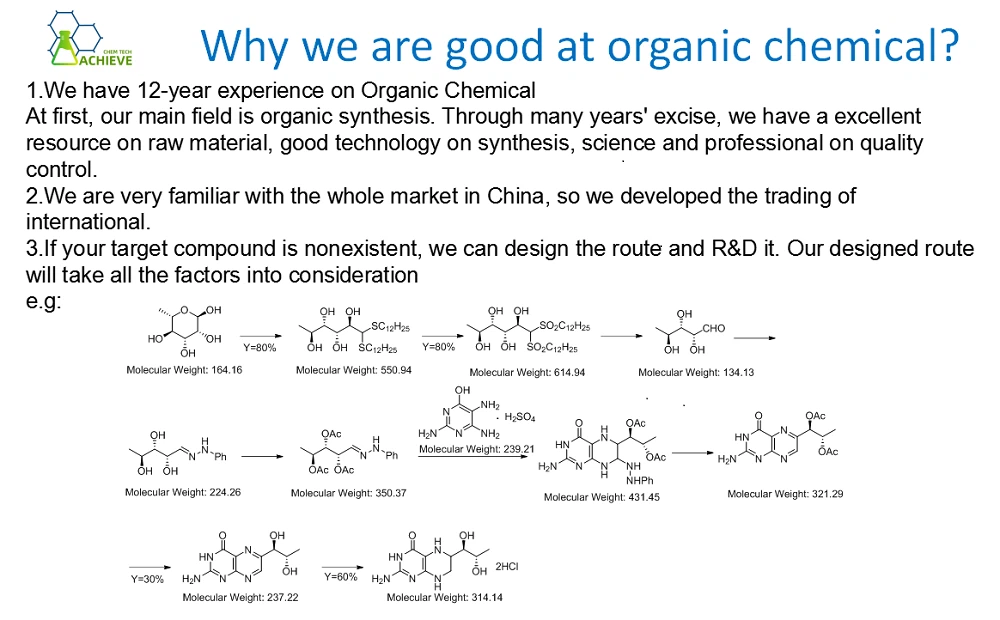
01. Application Cases in Pharmaceutical Field
In pharmaceutical manufacturing, thioacetamide, as an important pharmaceutical raw material, is involved in the synthesis of a wide range of drugs. For example, it can be used as a precursor for the synthesis of certain thio compounds with specific pharmacological activities, which play a key role in the development of antitumor, antibacterial or antiviral drugs. Especially in the development of anticancer drugs, the introduction of thioacetamide enhances the targeting and bioavailability of the drug, thus improving therapeutic efficacy and reducing side effects.
02. Application examples in the rubber industry
In the rubber industry, thioacetamide is widely used as a vulcanizing and cross-linking agent in the vulcanization of natural and synthetic rubber. By adjusting the dosage and vulcanization conditions of thioacetamide, the hardness, tensile strength, abrasion resistance and aging resistance of rubber products can be precisely controlled. For example, in the production of automobile tires, the use of thioacetamide can significantly improve the durability and driving safety of tires.


03. Electroplating Industry Application Cases
In the electroplating industry, thioacetamide is used as an electroplating additive to improve the electrochemical performance of the plating solution and the quality of the plated layer. By adjusting the concentration of thioacetamide and plating conditions, the thickness, uniformity and gloss of the plated layer can be controlled. Especially in copper, nickel and zinc plating processes, the introduction of thioacetamide can significantly improve the adhesion and corrosion resistance of the plated layer. This is of great significance for improving the reliability and prolonging the service life of electronic products.
04. Application cases in the field of environmental protection
In the field of environmental protection, thioacetamide is also used to treat wastewater containing heavy metal ions. Since thioacetamide can form stable complexes with heavy metal ions, it can effectively remove them from wastewater. This method has the advantages of easy operation, high treatment efficiency, and low cost. Especially in the treatment of industrial wastewater containing cadmium, lead, mercury and other toxic heavy metal ions, the application of thioacetamide can significantly reduce the heavy metal content of the wastewater to meet the environmental emission standards.


05. Application cases in analytical chemistry
In analytical chemistry, thioacetamide, as an important analytical reagent, is used to decompose copper complexes of amino acids and to check elements such as bismuth. By utilizing the reaction characteristics of thioacetamide with copper complexes of amino acids, the content of amino acids in a sample can be accurately determined.
Meanwhile, thioacetamide can also be used as an additive for the asymmetric reduction of β-ketoacetate by immobilized yeast, which is used to improve the selectivity and yield of the asymmetric reduction reaction.
In summary
In summary, as a multifunctional chemical, thioacetamide plays an important role in many fields such as medicine, rubber, electroplating, environmental protection and analytical chemistry. With the continuous progress of science and technology and the continuous expansion of application fields, the application prospect of thioacetamide will be even broader. However, when using thioacetamide, it is also necessary to pay attention to its hepatotoxicity and carcinogenicity to ensure that it is produced and applied under safe conditions.


A preparation method of thioacetamide solution is disclosed, including the following steps:
Step 1:
Prepare HS by reacting sulfuric acid with NaHS. The chemical reaction formula is as follows: 2NaHS+HSO (dilute)=2HS+NaSO.
Step 2:
Prepare crude thioethylamine with HS and acetonitrile (CHCN) obtained in step 1 under the catalysis of organic amine catalyst, and the chemical reaction formula is as follows: CHCN+HS=CHCSNH;
Step 3:
Distill the crude thioethylamine obtained in Step 2 to remove the residual CHCN and catalyst, dissolve the filtrate in anhydrous alcohol, crystallize and recrystallize, and then vacuum dry to obtain the finished thioethylamine.
The method has high product yield, simple production process, is suitable for industrial large-scale production, and the purity of the product obtained is high, which meets the use requirements of biomedical intermediates.
Chemical properties of thioacetamide solution:

After boiling for a long time, CH3CONH2 is further hydrolyzed:
![]()
![]()
The hydrolysis rate of thioacetamide increases with the increase of temperature, and the reaction is generally carried out in boiling water bath. The hydrolysis rate in alkaline solution is faster than that in acidic solution.
It can reduce the escape of toxic H2S gas and reduce the degree of air pollution in the laboratory.
The precipitation of relatively pure metal sulfide is obtained by uniform precipitation, and the co-precipitation is less, which is convenient for separation.
As (V) is reduced to As (III) by thioacetamide at a fast rate.
The tin sulfide precipitate obtained by precipitation of Sn (II) with thioacetamide is soluble in NaOH or CH3CSNH2+NaOH. The reaction is as follows:
![]()
Co2+and Ni2+react with CH3CSNH2 in NH4+- NH3 solution, and precipitate into β- CoS and β- NiS.
When Cu2+reacts with thioacetamide in a solution with pH<1, it is first reduced to Cu+, and then forms a complex with thioacetamide. When heated, the complex decomposes to form Cu2S precipitation. When the acidity of the solution decreases, Cu2+and CH3CSNH2 form a mixture of Cu2S and CuS.
When Hg2+reacts with thioacetamide in HCl solution, it often produces a series of intermediate products and precipitates of different colors.
Oxidative substances should be removed in advance to prevent some thioacetamide from being oxidized to SO42 -, so that the fourth group of cations can precipitate at this time.
The amount of thioacetamide should be appropriately excessive to ensure complete precipitation of thioacetamide.
Precipitation shall be carried out by heating in boiling water bath, and heating at this temperature for an appropriate long time to promote the hydrolysis of thioacetamide to ensure complete sulfide precipitation.
After the precipitation of the third group of cations, there is still a considerable amount of thioacetamide in the solution. In order to avoid oxidation and premature precipitation of the fourth group of cations, the fourth group of cations should be immediately analyzed.

Thioacetamide (CAS No. 62-55-5) does have some side effects and risks, which include the following:
Toxic effects:
Thioacetamide has direct hepatotoxic effects. After ingestion, it can be metabolized to TAA thioredoxin by cytochrome P450 mixed-function oxidase in hepatocytes, which interferes with intracellular RNA transfer in the nucleus, affects protein synthesis and enzyme activity, and increases DNA synthesis and mitosis in the nucleus of hepatocytes, thus promoting the development of liver cirrhosis.
Carcinogenicity:
Thioacetamide has been tested and shown to be potentially carcinogenic in experimental animals. Therefore, prolonged or heavy exposure to thioacetamide may increase the risk of cancer.
Skin and Eye Irritation:
Thioacetamide can cause severe skin and eye irritation. Skin contact may cause sensitization, dryness, inflammation, and even non-allergic contact dermatitis. Eye contact may cause severe eye irritation and damage.
Inhalation Hazards:
Thioacetamide is harmful if swallowed, and inhalation of its vapors or dust may also cause adverse health effects, such as inflammation of the mucous membranes and respiratory system, and adverse effects on the kidneys, liver, and central nervous system. Symptoms may include headache, nausea, dizziness, fatigue, weakness, sluggishness, and in extreme cases even unconsciousness.
Thioacetamide is harmful to aquatic organisms and has long lasting effects. If released into the environment, it may cause damage to aquatic ecosystems.
The use of thioacetamide may result in injury to the operator from the chemical if it is not handled properly or the necessary protective measures are not taken. In addition, in case of leakage of thioacetamide, it may cause pollution to the surrounding environment.
To minimize the side effects and risks of thioacetamide, strict precautions should be taken when using it. For example, wear appropriate personal protective equipment (e.g. protective gloves, protective clothing, eye protection and breathing masks) to avoid direct contact with the chemical; wash your body and clothing thoroughly before and after operation; ensure that the workplace is well ventilated to avoid inhalation of vapors or dust; and follow the correct operating procedures and safety guidelines.
In summary, thioacetamide has a variety of side effects and risks, including health hazards, environmental risks and risks of use. Therefore, it should be used with extreme caution and necessary precautions should be taken to ensure safety.
Frequently Asked Questions
Here are five frequently asked questions (FAQs) about Thioacetamide, along with their answers:
What is Thioacetamide?
+
-
Thioacetamide (TAA) is an organosulfur compound with the molecular formula C₂H₅NS. It appears as colorless crystals with a slight mercaptan odor. Thioacetamide is soluble in water and serves as a source of sulfide ions in chemical synthesis. It is a prototypical thioamide and has various industrial applications, although its use is now primarily limited to laboratory settings.
What are the primary uses of Thioacetamide?
+
-
Thioacetamide is currently used mainly as a replacement for hydrogen sulfide in qualitative analyses due to its safer handling properties. It is also employed as a reactant in the synthesis of metal salt nanoparticles. Historically, it had applications in industries such as leather, textile, and paper as an organic solvent, as well as in the vulcanization of rubber and stabilization of motor fuel, but these uses have largely been discontinued.
Is Thioacetamide hazardous to health?
+
-
Yes, Thioacetamide is considered a hazardous compound. It is classified as a carcinogen by various health and safety organizations. Exposure to Thioacetamide can occur through inhalation or dermal contact, and it has been linked to liver cancer and other tumors in experimental animals. Therefore, it should be handled with appropriate safety precautions, including the use of personal protective equipment and adherence to safety data sheet guidelines.
How is Thioacetamide synthesized?
+
-
Thioacetamide can be synthesized through the reaction of hydrogen sulfide with acetonitrile in the presence of a catalyst. Improved processes involve using polymer-supported amine catalysts to enhance yield and efficiency. The reaction is typically carried out at elevated temperatures, and the product is purified through various methods, such as crystallization or chromatography, depending on the desired purity level.
What safety measures should be taken when handling Thioacetamide?
+
-
When handling Thioacetamide, it is essential to follow all safety precautions outlined in the safety data sheet. This includes wearing appropriate personal protective equipment, such as gloves and eye protection, to prevent skin and eye contact. Work should be conducted in a well-ventilated area to minimize inhalation exposure. After handling, thorough washing of the face, hands, and any exposed skin is recommended. Additionally, Thioacetamide should be stored in a cool, dry place away from incompatible substances, and all safety guidelines should be reviewed and understood before use.
Hot Tags: thioacetamide solution cas 62-55-5, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 3 methyl 4 piperidone, Methylamine hydrochloride powder, Additive, N N N Trimethylethylenediamine, DIMETHYLPHOSPHINE OXIDE, 2 Chloro 4 pyridinecarboxylic acid




