Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of eptifibatide cas 188627-80-7 in China. Welcome to wholesale bulk high quality eptifibatide cas 188627-80-7 for sale here from our factory. Good service and reasonable price are available.
Eptifibatide is an antiplatelet compound that belongs to the category of glycoprotein IIb/IIIa inhibitors. It is a small molecule compound with relatively low molecular weight and relatively small molecular volume. It is a white or almost white powder or crystalline solid. Molecular formula C35H49N11O9S2, CAS 188627-80-7. Exhibiting different electrical conductivity under different conditions. After dissolution in water, its conductivity may increase.
It is also used in patients with ST segment elevation myocardial infarction (STEMI). It can be used in combination with thrombolytic drugs (such as recombinant tissue plasminogen activator) to enhance thrombolysis and restore coronary blood flow. It can be used for the treatment of patients with non ST segment elevation myocardial infarction (NSTEMI) and unstable angina (UA). It reduces or prevents myocardial ischemia by blocking platelet aggregation and reducing the formation of blood clots.
|
Customized Bottle Caps And Corks:
|
|
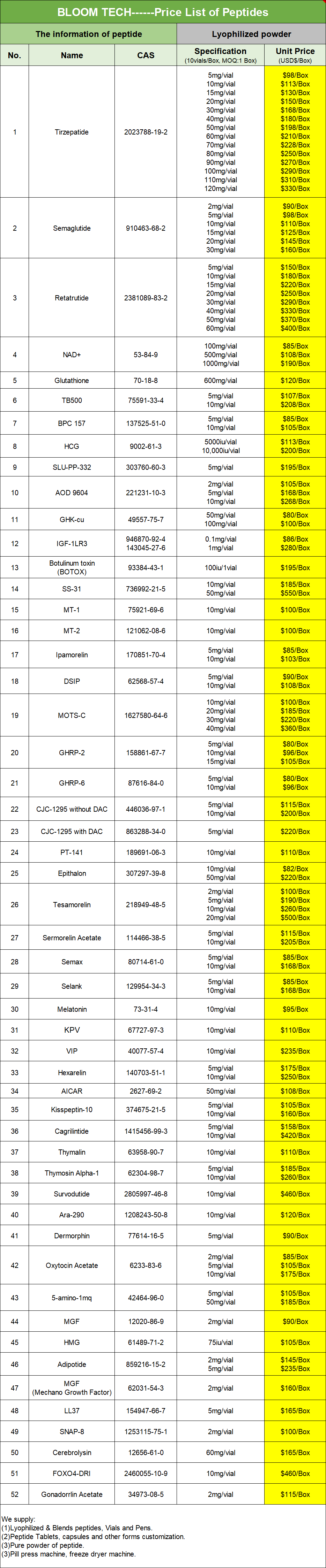
|
Chemical Formula |
C35H49N11O9S2 |
|
Exact Mass |
831 |
|
Molecular Weight |
832 |
|
m/z |
831 (100.0%), 832 (37.9%), 833 (9.0%), 833 (7.0%), 832 (4.1%), 834 (3.4%), 833 (1.8%), 832 (1.6%), 833 (1.1%) |
|
Elemental Analysis |
C, 50.53; H, 5.94; N, 18.52; O, 17.31; S, 7.71 |

Eptifibatide is an IIb/IIIa receptor antagonist that specifically binds to IIb/IIIa receptors on the surface of platelets, blocking the binding of fibrinogen to these receptors.
Inhibition of platelet aggregation:
Platelet aggregation is one of the key steps in the formation of blood clots. IIb/IIIa receptors are key molecules in platelet aggregation, and their activation can mediate the formation of transverse bridging connections between platelets, thereby promoting platelet aggregation and thrombosis. By competitively binding to IIb/IIIa receptors, it prevents platelet aggregation and thrombus formation.
Pharmacokinetics:
The half-life in the body is about 2.5 hours, mainly through renal metabolism and excretion. Due to its short-term effects, it is usually administered intravenously rather than orally.


Reversibility:
The antiplatelet effect is reversible. Once discontinued, the drug will be metabolized and excreted, and platelet function will gradually return to normal. This reversibility makes Eptiibati very useful in situations where short-term antiplatelet therapy is needed, such as in patients with myocardial infarction who undergo coronary intervention (PCI).
Antithrombotic effect:
Antithrombotic effect makes it useful in various clinical situations. It is widely used in patients with acute coronary syndrome (ACS), myocardial infarction (MI), and patients undergoing coronary intervention (PCI) to reduce the risk of thrombosis and cardiovascular events.


Safety:
Although Epifiibatid can effectively prevent thrombosis, it also increases the risk of bleeding. Therefore, during the use of Epifiibatid, medical staff need to closely monitor the patient's coagulation function, pay attention to any possible bleeding, and take corresponding measures. In addition, it may not be applicable in certain specific situations, such as active bleeding, uncontrolled hypertension, severe coagulation dysfunction, etc.
Drug interactions:
May interact with other drugs, especially those that affect platelet function and coagulation processes. When using Epifiibatid, medical staff need to pay attention to other medications that the patient is using and adjust the dosage or monitor as needed.


The laboratory synthesis method of Eptifibatide usually involves multiple steps, including the selection of raw materials, control of reaction conditions, and purification. Provide a possible laboratory synthesis method for Epitibatide:
Firstly, select appropriate raw materials such as Trt Mpa OH, H-Lys (Fmoc) OMe, and other basic amino acids and reagents required for dipeptide synthesis. Ensure that the purchased raw materials meet the chemical purity standards to ensure smooth synthesis.
Trt Mpa OH and H-Lys (Fmoc) OMe are used as starting materials to synthesize dipeptides through liquid-phase synthesis. This step usually involves reactions such as the setting of protective groups, deprotection, and the formation of peptide bonds. In the synthesis process, it is necessary to strictly control the reaction conditions, such as temperature, pH value, raw material ratio, etc., to ensure the smooth progress of the reaction and the stability of the product.
The synthesized dipeptide is condensed with the H-Gly Asp (OtBu) - Trp (Boc) - Pro Cys (P1) - NH2 fragment. This step typically involves reactions such as removing Fmoc protective groups, forming peptide bonds, and setting up side chain protective groups. The condensation reaction needs to be carried out under specific conditions to ensure the correctness of the product structure and the improvement of yield.
The synthesized dipeptide is linked to the product obtained by fragment condensation to form the full protective peptide Trt Mpa Har (Boc) 2-Gly Asp (OtBu) - Trp (Boc) - Pro Cys (P1) - NH2. The reaction conditions and operation in this step are similar to the condensation of dipeptides and fragments, and strict control of the reaction conditions is required to ensure the stability and correctness of the product.
In fully protected peptides, the protective groups on each side chain need to be removed and undergo an oxidation reaction to obtain the final crude Eptiibatide. This step usually involves using acidic or alkaline solutions for deprotection, and using oxidants such as H2O2 for oxidation reactions. In the process of deprotection and oxidation, it is necessary to control the reaction conditions to avoid product loss and quality degradation caused by excessive or incomplete oxidation.
Purify the crude Ettifabatide product to remove impurities and by-products. The purification method can be selected according to the actual situation, such as gel filtration, reverse phase high performance liquid chromatography, etc. Through purification, high-purity Eptifiatide can be obtained, laying the foundation for subsequent biological activity determination and drug development.
Conduct biological activity testing on purified Eptiibatide to evaluate its expected biological effects. The activity determination method can be selected according to actual needs, such as the determination of platelet glycoprotein GP IIb/IIIa receptor antagonistic activity. By conducting activity assays, the pharmacological properties of Epifiibatide can be preliminarily evaluated, providing a basis for subsequent drug development and clinical research.

Pharmacodynamics is the scientific study of the absorption, distribution, metabolism, and excretion (ADME) processes of drugs in living organisms and their temporal variations. It is crucial for understanding drug mechanisms, optimizing dosing regimens, predicting drug interactions, and evaluating drug safety. Etriptide is a platelet glycoprotein IIb/IIIa receptor antagonist widely used in acute coronary syndrome (ACS) and percutaneous coronary intervention (PCI) to reduce the risk of thrombosis and cardiovascular events. Its unique pharmacokinetic properties play a crucial role in ensuring its effectiveness and safety in clinical practice.

1. Absorption
Etriptide is mainly administered through intravenous injection, so its absorption process does not involve the traditional gastrointestinal absorption barrier. After intravenous injection, the drug enters the bloodstream directly, avoiding the first pass effect and allowing the blood drug concentration to quickly reach the therapeutic level. Within just 5 minutes after intravenous injection, the peak blood concentration of eptibatide can be reached, and this rapid onset characteristic is particularly critical for emergency situations that require rapid inhibition of platelet aggregation.
2. Distribution
After entering the bloodstream, drugs will quickly distribute to various tissues throughout the body. The distribution volume of eptipatide is approximately 185ml/kg, which reflects the widespread distribution of the drug in the body, but does not significantly enter cells or tightly bind with other tissues. Its distribution half-life is only 5 minutes, indicating that the drug's distribution process in the body is very rapid and can reach the effective concentration of each target organ in a short period of time. It is worth noting that the total protein binding rate of eptibatide is 25%, which means that most drugs exist in free form in the blood, which facilitates rapid binding and action of drugs with targets such as glycoprotein IIb/IIIa receptors on platelets.


3. Metabolism
The metabolism of eptifibatid in the body is relatively simple, mainly through deamination reaction to generate deamination eptifibatid and polar metabolites. These metabolites do not have biological activity, so they will not interfere with the pharmacological effects of the original drug. The main site of drug metabolism is the liver, but the metabolic rate of eptifibatid is not high, which helps maintain its long-lasting efficacy. In addition, due to the inactivity of metabolites, there is no need to worry about their potential toxicity or drug interactions.
4. Excretion
The excretion pathways of eptibatide are diverse, mainly including renal excretion, respiratory excretion, and fecal excretion. Among them, renal excretion is the main excretion pathway, accounting for about 71.4% of the total amount of drugs. This high renal clearance rate (3.79L/h) ensures that the drug does not accumulate excessively in the body, helping to maintain the stability of blood drug concentration. In contrast, the amount of drugs excreted through respiration and feces is extremely small, less than 1% and 1.5% respectively, indicating that these two excretion pathways have limited effects on the clearance process of eptipatide.


5. Eliminate half-life
The elimination half-life is the time required for a drug to be eliminated by half in the body, and is an important indicator for evaluating the duration of drug action. The elimination half-life of eptipatide is 1.13-2.5 hours, indicating that the drug has a faster clearance rate in the body. This characteristic enables eptibatide to achieve therapeutic effects in a short period of time and be quickly cleared from the body after completing its pharmacological effects, reducing the potential burden on the body.
5. Clearance of hemodialysis
For patients with impaired kidney function, hemodialysis is an effective medication clearance method. Etriptide can be cleared through hemodialysis, which provides a safe guarantee for patients with renal insufficiency to use Etriptide. During hemodialysis, drugs are filtered out of the body through the dialysis membrane, thereby reducing blood drug concentration and avoiding excessive accumulation of drugs in the body.

As an efficient platelet glycoprotein IIb/IIIa receptor antagonist, the unique pharmacokinetic properties of eptifibatide provide strong support for its widespread clinical application. Future research can further explore the pharmacokinetic differences of eptipatide in different patient populations, as well as its interaction mechanisms with other drugs, in order to optimize dosing regimens, improve treatment efficacy, and reduce the risk of adverse reactions. Meanwhile, with the continuous advancement of drug development technology, the research and development of new platelet inhibitors will also provide more choices for the prevention and treatment of cardiovascular diseases.
Frequently Asked Questions
Is eptifibatide an anticoagulant?
+
-
Eptifibatide is an anti-coagulant that selectively and reversibly blocks the platelet glycoprotein IIb/IIIa receptor.
What are the 5 antiplatelet drugs?
+
-
Antiplatelet medications divide into oral and parenteral agents, and oral agents subdivide further based on the mechanism of action. Aspirin was the first antiplatelet medication and is a cyclooxygenase inhibitor. Other oral antiplatelet include clopidogrel, ticagrelor, and prasugrel, cilostazol, and dipyridamole.
Is eptifibatide a peptide?
+
-
Eptifibatide is a disintegrin type of peptide that mimics a portion of barbourin, a toxic peptide found in the venom of the Southeastern pygmy rattlesnake (Sistrurus miliarius barbouri) .
Hot Tags: eptifibatide cas 188627-80-7, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, Melanotan ii powder, Cosmetic, dermorphin in humans, dermorphin peptide, DERMORPHIN










