Larazotide acetate peptide, The Chinese name is Lirizole Acetate, which is an artificially synthesized octapeptide compound. Its amino acid sequence is H-Gly-Gly Val Leu Val Gln Pro Gly-OH, represented by GGVLVQPG for single letters and Gly-Gly Val Leu Val Gln Pro Gly for three letters. This molecule is composed of eight amino acid residues connected by peptide bonds to form a linear polypeptide structure. The N-terminus starts with two glycine residues connected, followed by valine, leucine, valine, glutamine, and proline in the middle. The C-terminus is glycine. This specific arrangement of amino acids endows it with a unique spatial conformation and biological activity.
Molecular Weight and Formula
There are two forms, the molecular weight of the free base form (Larazotide) is 725.83 g/mol, and the molecular formula is C32H55N9O10; The molecular weight of Larazotide acetate is 785.89 g/mol, with the molecular formula C32H55N9O10.
Appearance and Purity
Usually presented as white or off white powder, purity (HPLC) ≥ 95%, acetate content ≤ 12.0%, moisture content ≤ 8.0%, peptide content ≥ 80.0%, endotoxin ≤ 50EU/mg, amino acid composition analysis ≤± 10%.
Storage conditions
To maintain its chemical stability and biological activity, it is recommended to store it in a low-temperature environment (such as -20 ℃ or lower), usually in vacuum packaging or sealed containers, avoiding contact with air, moisture, and other substances that may affect its quality.
From a long-term preservation perspective, freeze-drying may be more advantageous in maintaining its activity, and it can be dissolved in appropriate buffer or solvent according to experimental requirements before use.
Our product



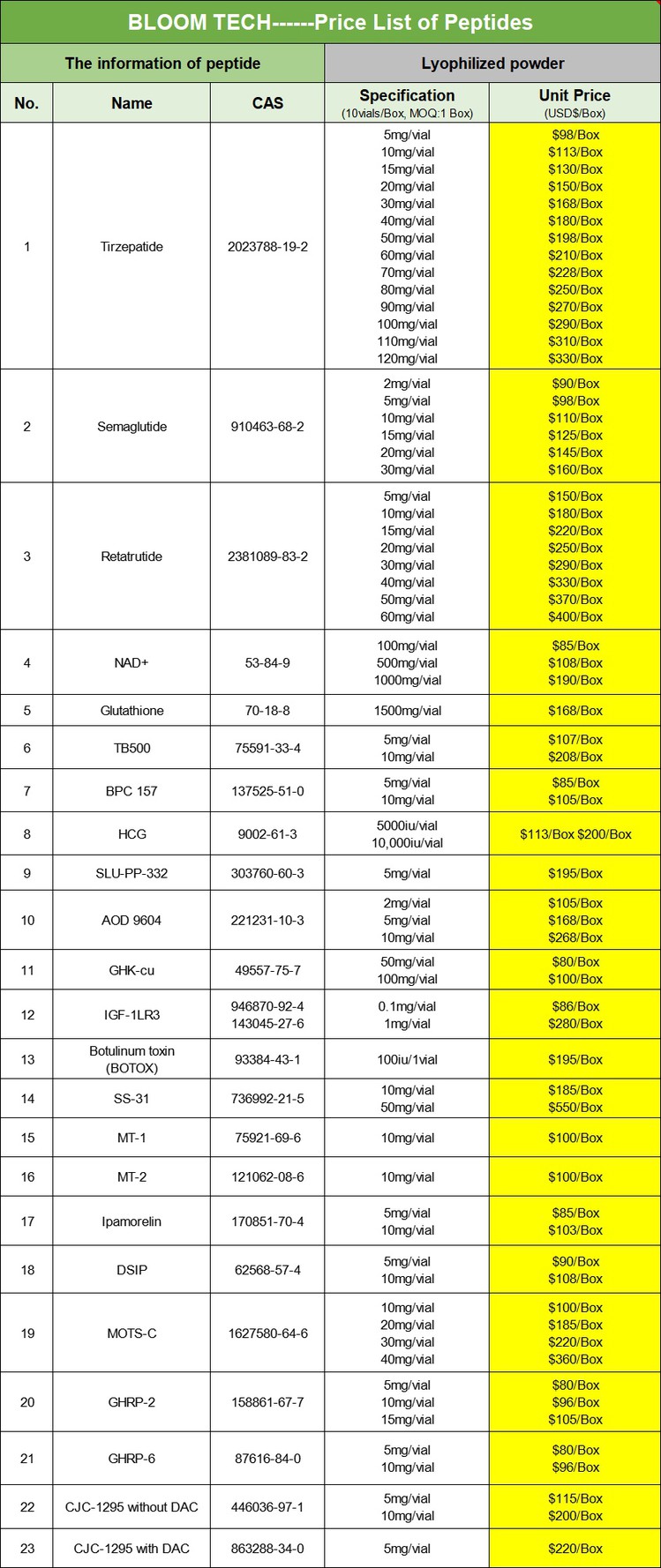



Larazotide acetate. COA
 |
||
Certificate of Analysis |
||
|
Compound name |
Larazotide acetate | |
|
CAS No. |
881851-50-9 | |
|
Grade |
Pharmaceutical grade | |
|
Quantity |
Customized | |
|
Packaging standard |
Customized | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
|
Lot No. |
20250109001 |
|
|
MFG |
Jan 12th 2025 |
|
|
EXP |
Jan 8th 2029 |
|
|
Structure |
|
|
| TEST STANDARD | GB/T24768-2009 Industry. Stnndard | |
|
Item |
Enterprise standard |
Analysis result |
|
Appearance |
White or almost white powder |
Conformed |
|
Water content |
≤4.5% |
0.30% |
| Loss on drying |
≤1.0% |
0.15% |
|
Heavy Metals |
Pb≤0.5ppm |
N.D. |
|
As≤0.5ppm |
N.D. | |
|
Hg≤0.5ppm |
N.D. | |
|
Cd≤0.5ppm |
N.D. | |
|
Purity (HPLC) |
≥99.0% |
99.5% |
|
Single impurity |
<0.8% |
0.48% |
|
Residue on ignition |
<0.20% |
0.064% |
|
Total microbial count |
≤750cfu/g |
80 |
|
E. Coli |
≤2MPN/g |
N.D. |
|
Salmonella |
N.D. | N.D. |
|
Ethanol (by GC) |
≤5000ppm |
400ppm |
|
Storage |
Store in a sealed, dark and dry place at-20 degrees |
|

|
||

Formulation development and administration strategy
Formulation Form
It is usually stored in freeze-dried powder form and can be dissolved in appropriate buffer or solvent according to experimental needs during use. The development of its formulation needs to consider its stability and bioavailability to ensure optimal efficacy in vivo.
Administration route and dosage
In animal experiments, the administration routes of Larazotid acetate include oral and intraperitoneal injection. For research on celiac disease, the usual oral dose is 5 mg/kg/day; For colitis models, the oral dose is 5 mg/kg/day; For the cardiotoxicity protection model, the intraperitoneal injection dose is 250 μ g, twice a week. Dose optimization studies have shown that the efficacy of Larazotid acetate is dose-dependent, but no additional benefits have been observed beyond a certain dose, and there may be an increased risk of adverse reactions.

Larazotid acetate is an orally active zonulin antagonist with broad application prospects in the medical field. Its unique molecular structure and mechanism of action make it valuable in the treatment of celiac disease, the study of viral infectious diseases and other related fields.

Research on viral infectious diseases
1. Antiviral activity of Lirizole Acetate
Research has found that rabeprazole acetate has antiviral activity against varicella zoster virus (VZV). In vitro experiments, the EC50 values of the OKA and 07-1 strains of VZV treated with rabeprazole acetate were 44.14 and 59.06 μ M, respectively. The EC50 value is an indicator of drug efficacy, which represents the drug concentration required to achieve a maximum effect of 50%. These results indicate that rabeprazole acetate can effectively inhibit virus replication and transmission at lower concentrations, demonstrating good antiviral effects and drug safety.
2. Research on antiviral mechanisms
Although the specific antiviral mechanism of rabeprazole acetate has not been fully elucidated, it is speculated that it may interfere with the processes of virus adsorption, invasion, replication, or release by regulating certain signaling pathways in host cells. For example, it may affect the permeability of the cell membrane or signal transduction within the cell, preventing the virus from binding to the host cell or entering the cell; It may also interfere with the replication of the viral genome or protein synthesis, thereby inhibiting viral reproduction.


3. Potential application in other viral infectious diseases
Because larrazole acetate has antiviral activity against VZV, researchers began to explore its potential application in other viral infectious diseases. With the in-depth study of the antiviral mechanism of VZV, the drug is expected to provide new strategies and methods for the treatment of other viral infectious diseases. For example, it may also have a certain inhibitory effect on viruses with similar infection mechanisms or signaling pathways to VZV.
Other potential application areas
1. Inflammatory diseases of the intestine
Inflammatory diseases of the intestine, such as ulcerative colitis and Crohn's disease, are also associated with impaired intestina barrier function and increased intestina permeability. Acetate rabeprazole may have a certain therapeutic effect on these diseases by regulating intestina tight junctions and maintaining the integrity of the intestina barrier. At present, although there are relatively few clinical studies related to it, some animal experiments have preliminarily confirmed its potential in reducing intestina inflammation and improving intestina barrier function.


2. Autoimmune diseases
The occurrence of autoimmune diseases is related to abnormal activation of the immune system, and impaired intestina barrier function may lead to the entry of antigens into the body, triggering an immune response. Acetate rabeprazole may help regulate the balance of the immune system by improving intestina barrier function and reducing antigen exposure, and has a certain adjuvant therapeutic effect on some autoimmune diseases such as rheumatoid arthritis and systemic lupus erythematosus. However, further research is needed to confirm this.
3. Drug development model
As a larazotide acetate peptide drug with a clear mechanism of action, the unique molecular structure and mode of action of rabeprazole acetate provide an important model for drug development. Researchers can develop similar peptide drugs or small molecule compounds for the treatment of other diseases related to intestina barrier function or tight junctions by studying their structure and mechanism of action.


Future research directions and mechanism deepening
Subcellular Localization Study
The current research mainly focuses on the cellular level effects of Larazotid acetate, but its subcellular localization (such as mitochondria and endosomes) is not yet clear. In the future, super-resolution microscopy imaging technology is needed to reveal its dynamic distribution within cells and the interaction mechanism with target molecules.
Signal pathway integration
Larazotid acetate involves multiple signaling pathways (such as the zonulin signaling pathway and antiviral signaling pathway), but its signal integration mechanism has not been fully elucidated. In the future, systems biology methods such as proteomics and phosphogenomics need to be used to construct a signaling regulatory network for Larazotid acetate and reveal its multi-target synergistic effects.
Individualized medication strategy
Based on population genomics data, it is necessary to establish a Larazotid acetate efficacy prediction model. For example, individuals carrying polymorphisms in genes related to the zonulin signaling pathway may be more sensitive to Larazotid acetate, while populations with high expression of genes related to the antiviral signaling pathway may require higher doses to achieve therapeutic effects.
Larazotide acetate peptide, as a multifunctional physiological regulatory factor, has shown broad prospects in the fields of intestina tight junction regulation, antiviral and immune regulation through its unique chemical structure and mechanism of action. Although its clinical transformation still faces challenges such as bioavailability and long-term safety, with the deepening of mechanism research and the breakthrough of preparation technology, it is expected to become an innovative therapeutic drug for a variety of diseases (such as celiac disease, viral infectious diseases, adjuvant chemotherapy), providing new treatment options for patients. Future research needs to further explore its synergistic effects in combination therapy and establish efficacy prediction biomarkers to promote its clinical translation.
The acetylated C-terminus of Larazotide Acetate Peptide can bridge silica particle defects

As an important barrier between the human body and the external environment, the intestinal epithelium plays a crucial role in maintaining the stability of the intestinal environment, preventing pathogen invasion, and absorbing nutrients. However, various factors such as inflammatory bowel disease, infection, drug side effects, etc. can lead to intestinal epithelial damage and barrier dysfunction, thereby triggering a series of intestinal diseases. It is a peptide substance with potential therapeutic value, which has attracted attention in the treatment of intestinal diseases. At the same time, silica particles, as a common inorganic material, have a wide range of applications in the biomedical field. The defects on the surface of their particles have a significant impact on material properties and biological interactions.
It was initially found to have a regulatory effect on intestinal tight junctions. Tight junction is an important connecting structure between intestinal epithelial cells, which can control the transport of substances between cells and maintain the integrity of the intestinal barrier. In conditions such as inflammatory bowel disease, the tight connections of the intestine are disrupted, leading to increased intestinal permeability. Pathogens and harmful substances can easily enter intestinal tissues, triggering inflammatory reactions. It can enhance intestinal barrier function, reduce intestinal permeability, and alleviate inflammatory symptoms by binding to specific receptors on the surface of intestinal epithelial cells, regulating the expression and distribution of tight junction proteins.

The interaction mechanism between acetylated C-terminal and silica particles
Physical adsorption
The physical adsorption between acetylated C-terminal and silica particles is an important way of interaction. The surface of silica particles usually carries a negative charge, while the acetylated C-terminal undergoes a change in charge distribution due to the presence of acetyl groups, which may carry a certain positive charge or have a specific charge distribution region. This charge difference creates an electrostatic attraction between the acetylated C-terminal and the surface of silica particles, thereby promoting the adsorption of the acetylated C-terminal onto the particle surface. In addition, the roughness and pore structure of the surface of silica particles provide more adsorption sites for acetylated C-terminus, enhancing physical adsorption.
Chemical bonding
In addition to physical adsorption, acetylation of the C-terminus may also result in chemical bonding with the surface of silica particles. There are a large number of silicon hydroxyl groups (Si OH) on the surface of silica particles, which have certain reactivity. Acetylation of certain amino acid residues in the C-terminal, such as the phenolic hydroxyl group of tyrosine (Tyr) and the amide group of asparagine (Asn), may undergo chemical reactions with the silanol group to form chemical bonds, such as hydrogen bonds, covalent bonds, etc. This chemical bonding can make the acetylated C-terminal more firmly bound to the surface of silica particles, enhancing the bridging effect.
Bridge mechanism
Acetylated C-terminus can exert bridging effect by binding to defect sites on the surface of silica particles through physical adsorption and chemical bonding. Due to the length and flexibility of the acetylated C-terminus, it can simultaneously interact with two or more adjacent defect sites, connecting dispersed silica particles together, filling the defect gaps between particles, and forming a more compact and stable structure. This bridging effect is similar to the role of tight junction proteins between intestinal epithelial cells, which can enhance the connection strength between particles and improve the overall performance of the material.
Potential impact of bridging effect on simulating intestinal epithelial repair
Simulating intestinal epithelial cell connections
Intestinal epithelial cells are interconnected through cell junction structures such as tight junctions and adhesive junctions, forming a continuous barrier. The process of acetylating C-terminal bridging silica particle defects is somewhat similar to the connection process between intestinal epithelial cells. In the process of intestinal epithelial repair, it is necessary to restore the intercellular connectivity structure to rebuild the intestinal barrier. The stable structure formed by acetylated C-terminal bridging silica particles can simulate the connections between intestinal epithelial cells, providing a model for studying the formation mechanism and regulatory factors of cell connections. By observing the interaction process between acetylated C-terminus and silica particles, we can gain a deeper understanding of how cell junction proteins recognize and bind to specific sites, as well as how stable junction structures are formed through interactions.

Promote cell adhesion and growth
After acetylation C-terminal bridging, the surface properties of silica particles change, which may be more conducive to the adhesion and growth of intestinal epithelial cells. Cell adhesion is the basis for cells to adhere and spread on material surfaces, and is crucial for cell proliferation, differentiation, and functional performance. Acetylated C-terminal bridging on the surface of silica particles may provide more suitable biologically active sites for cell adhesion, enhancing the interaction between cells and materials. In addition, the bridged silica particles have a more stable structure, providing a favorable growth microenvironment for cells, promoting cell proliferation and differentiation, and facilitating the repair and regeneration of intestinal epithelium.

Regulating Inflammatory Response
Inflammatory response plays a dual role in the process of intestinal epithelial injury and repair. Moderate inflammatory response helps to clear pathogens and damaged cells, promoting the repair process; However, excessive inflammatory response can exacerbate tissue damage and delay the repair process. Research has found that Larazotide Acetate Peptide has the ability to regulate inflammatory responses. Acetylated C-terminal bridging silica particles may regulate the expression and release of inflammatory factors by affecting signaling pathways on the cell surface, thereby controlling the degree of inflammatory response. For example, it may inhibit the production of pro-inflammatory factors, promote the secretion of anti-inflammatory factors, and create a favorable inflammatory microenvironment for intestinal epithelial repair.

Research Methods and Experimental Validation
In vitro cell experiments
To verify the effect of acetylated C-terminal bridging silica particles on intestinal epithelial cells, we conducted in vitro cell experiments. Inoculate intestinal epithelial cells (such as Caco-2 cells) onto culture plates coated with silica particles that have undergone acetylation C-terminal bridging treatment, and observe cell adhesion, growth, and morphological changes. Evaluate the adhesion quantity, proliferation ability, and expression of cell junction proteins of cells on the material surface through methods such as cell counting, cell viability detection (such as MTT assay), and immunofluorescence staining. The experimental results showed that compared with the untreated silica particle coating, the acetylated C-terminal bridging treatment coating can significantly promote the adhesion and growth of intestinal epithelial cells, enhance the expression of intercellular junction proteins, and further confirm the positive effect of bridging on simulating intestinal epithelial repair.
Animal experiments
In order to validate the repairing effect of acetylated C-terminal bridging silica particles at the in vivo level, we established an animal model of intestinal injury (such as DSS induced colitis mouse model). Administer a preparation containing acetylated C-terminal bridging silica particles to model mice through enema or other methods, and observe the pathological changes, degree of inflammation, and recovery of barrier function in the intestinal tissue of the mice. Evaluate the repair effect through methods such as tissue section staining (such as HE staining, immunohistochemical staining), intestinal permeability detection (such as FITC glucan detection), etc. Animal experiments have shown that acetylated C-terminal bridging silica particles can alleviate intestinal inflammation in mice, promote the repair of intestinal epithelial damage, improve intestinal barrier function, and provide strong experimental evidence for clinical applications.
Frequently Asked Questions
Is larazotide safe?
+
-
Absolutely. Clinical trials have demonstrated no serious adverse effects, even at doses significantly higher than what is typically prescribed. In nearly 500 patients studied, Larazotide was well-tolerated and showed no major safety concerns.
What peptides are good for leaky gut?
+
-
Larazotide is a peptide designed specifically to address intestinal permeability, commonly known as "leaky gut." This condition occurs when the tight junctions in the intestinal lining loosen, allowing toxins, pathogens, and undigested food particles to escape into the bloodstream.
What conditions does larazotide treat?
+
-
Also known as AT-1001, larazotide is a zonulin antagonist that protects against gliadin-induced alterations in paracellular permeability in patients with celiac disease. Phase I and Phase II clinical trials have demonstrated an excellent safety and tolerability profile.
Hot Tags: larazotide acetate peptide, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, DERMORPHIN, Cosmetic, dermorphin peptide, dermorphin in humans, Melanotan ii powder









