4-methylcatechol, molecular formula C7H8O2, CAS 452-86-8, At room temperature and pressure, it is a white to white crystalline powder with certain solubility in water. It is a phenolic derivative with significant acidity, which can be prepared from natural 4-methylguaiacol through oxidation reaction. In organic synthesis, this substance can be applied to the preparation of catechol derivatives through the acidity and nucleophilicity of phenolic hydroxyl groups. In addition, the compound can also be used as an industrial production raw material for the production of antibacterial agents, antioxidants, and high-efficiency polymerization inhibitors. It plays an important role in organic synthesis, and its phenolic hydroxyl group's acidity and nucleophilicity make it one of the important raw materials for preparing catechol derivatives, which can be used for the synthesis of various phenolic bioactive molecules. In addition to its application in organic synthesis, it is also widely used as a raw material for industrial production. It can be used to produce chemicals such as antibacterial agents, antioxidants, and high-efficiency polymerization inhibitors. These compounds have a wide range of applications in the pharmaceutical, chemical, and other industrial fields, and can be used to manufacture products such as pharmaceuticals, skincare products, rubber, and plastics.

|
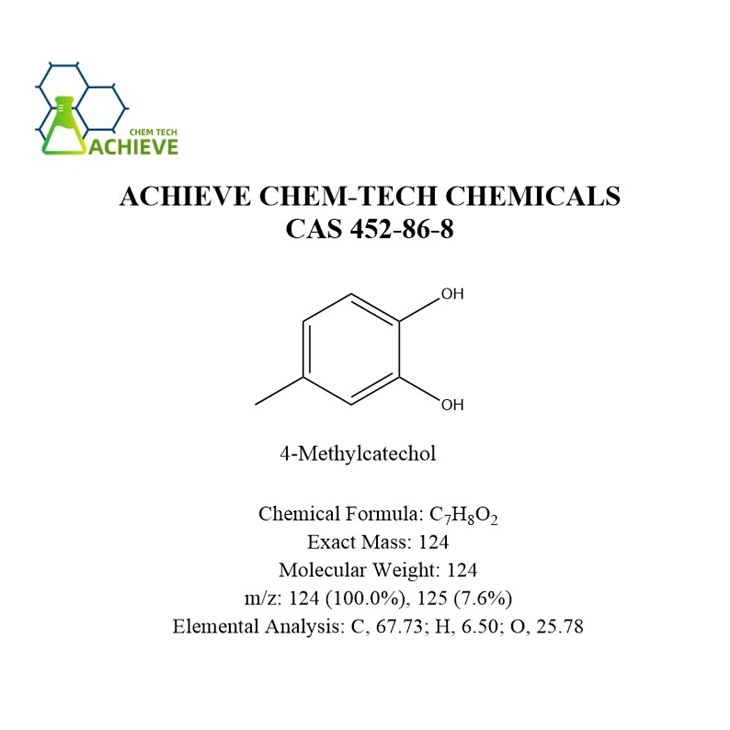
Chemical Formula |
C7H8O2 |
|
Exact Mass |
124 |
|
Molecular Weight |
124 |
|
m/z |
124 (100.0%), 125 (7.6%) |
|
Elemental Analysis |
C, 67.73; H, 6.50; O, 25.78 |
|
|
|

4-methylcatechol plays an important role in organic synthesis, and the acidity and nucleophilicity of its phenolic hydroxyl group make it one of the important raw materials for preparing catechol derivatives, which can be used for the synthesis of various phenolic bioactive molecules. In addition to its application in organic synthesis, it is also widely used as a raw material for industrial production. It can be used to produce chemicals such as antibacterial agents, antioxidants, and high-efficiency polymerization inhibitors.
Pharmaceutical Chemistry and Biopharmaceuticals

Cardiovascular drug development
The catechol structure of 4-methylcatechl is the core pharmacophore of adrenergic compounds such as adrenaline and norepinephrine. Methyl substitution can enhance molecular hydrophobicity and improve pharmacokinetic properties.
Example analysis:
Compound A (Dobutamine analog)
Synthesis pathway: 4-Methylcatecho is introduced into nitro group through nitration reaction, and reduced to obtain 3,4-dihydroxyphenylethylamine skeleton, which is then condensed with chloroacetyl chloride to form the target molecule.
Pharmacological effects: Selective activation of β - adrenergic receptors enhances myocardial contractility, increases cardiac index (CI) by 35% without significantly increasing heart rate.
Clinical advantages: Methyl substitution prolongs plasma half-life (t ²=3.2 h vs dobutamine 2.5 h) and reduces dosing frequency.
Compound B (alpha/beta receptor dual agonist)
Structural characteristics: catechol A is environmentally friendly and retained, while the B ring introduces a methyl substituted amino side chain.
Pharmacological data: In the rat ex vivo right ventricular experiment, the positive inotropic effect EC ₅₀=0.8 nM reduced peripheral vascular resistance by 40%.
Development stage: Currently in phase II clinical trials for the treatment of refractory heart failure.


Anti tumor drug design
The redox properties of catechol make it an ideal trigger group for prodrug design, and methyl substitution can regulate the activation rate of prodrugs.
Example analysis:
Compound C (nitrogen mustard prodrug)
Mechanism of action: 4-methylcatecho is linked to nitrogen mustard through a thioether bond. Under the action of NADPH/CYP450 reductase, which is highly expressed in tumor cells, the thioether bond is broken to release active nitrogen mustard.
In vitro activity: For multidrug resistant MCF-7/ADR breast cancer cells, IC ∨ ₀=1.2 μ M, significantly better than unmethylated control (IC ∨ ₀=8.9 μ M).
In vivo experiment: In the tumor bearing mouse model, the tumor growth inhibition rate (TGI) reached 72%, and the cardiac toxicity was lower than that of doxorubicin.
Compound D (Topoisomerase inhibitor)
Structural design: 4-methylcatechol and camptothecin are linked by ester bonds, utilizing the acidic microenvironment sensitivity of catechol to achieve targeted release.
Advantages: The release rate in tumor tissue (pH 6.5) is 5 times faster than in normal tissue (pH 7.4), reducing systemic toxicity.


Treatment of neurodegenerative diseases
Simulate dopamine metabolism pathway and design compounds with antioxidant and neuroprotective effects.
Example analysis:
Compound E (GSH conjugate)
Synthesis strategy: 4-methylcatecho is linked to glutathione through disulfide bonds, utilizing the coordination ability of catechol to promote blood-brain barrier penetration.
Pharmacological evaluation: In the MPTP induced Parkinson's disease mouse model, the survival rate of dopaminergic neurons increased by 45%, and the recovery rate of motor coordination ability reached 60%.
Mechanism: Compounds are enriched in astrocytes, promoting the directed transport of GSH to neurons and inhibiting oxidative stress.
Research on Anti infective Drugs
The metal chelating ability of catechol can disrupt the metabolic homeostasis of pathogenic microorganisms.
Example analysis:
Compound F (antifungal agent)
Target: Chelate Fe ³ ⁺ in fungal cell walls, disrupt membrane integrity, and cause content leakage.
In vitro activity: MIC=0.5 μ g/mL against Candida albicans, still effective against fluconazole resistant strains.
In vivo experiment: In the mouse model of systemic candidiasis, the survival rate increased from 10% in the control group to 65%.

Advanced functional materials field

Biomimetic adhesive materials
The strong coordination ability of catechol groups (forming five membered ring chelates with metal oxide surfaces) makes them outstanding in underwater adhesive materials.
Example analysis:
Polymer G (underwater adhesive)
Synthesis method: 4-Methylcatecho is copolymerized with acrylamide, and the molecular weight distribution is controlled by RAFT polymerization (Đ=1.12).
Performance parameters: The underwater adhesion strength to aluminum alloy reaches 1.8 MPa (better than the 0.8 MPa of commercial epoxy resin), and the salt spray resistance exceeds 500 hours.
Application case: Used for ship anti fouling coating, with an efficiency of 92% in preventing barnacles from adhering.
Hydrogel H (tissue adhesive)
Preparation process: 4-methylcatecho modified hyaluronic acid is cross-linked with Fe ³+to form self-healing water gel.
Medical application: Used for skin wound closure, with a burst strength of 25 kPa and biocompatibility tested according to ISO 10993 standard.


Conductive Polymer Composite Materials
As a dopant, it improves the processability and environmental stability of conductive polymers.
Example analysis:
Composite Material I (Flexible Electrode)
Material composition: PEDOT: PSS blended with 4-methylcatechl, prepared by solution coating method.
Electrical performance: The conductivity remains at 120 S/cm, and the tensile strength is increased to 45 MPa (pure PEDOT: PSS is 18 MPa).
Device application: Used for flexible supercapacitors, with an energy density of 8.7 Wh/kg and a capacity retention rate of 82% after 1000 cycles.
Thermal sensitive material J (temperature responsive conductive adhesive)
Design principle: 4-Methylcatechl is copolymerized with poly (N-isopropylacrylamide) (PNIPAm), utilizing the redox responsiveness of catechol.
Performance characteristics: At 32 ℃, the conductivity suddenly changes (from 0.1 S/cm to 15 S/cm), and the response speed is less than 1 s.


Optical functional materials
The UV absorption characteristics of catechol (λ _max=280 nm) make it have potential applications in photoresist and photosensitive coatings.
Example analysis:
Photoresist K (microelectronic processing)
Formula composition: Epoxy resin/photoacid generator system containing 4-methylcatechol.
Lithography performance: Under 365 nm illumination, the curing depth reaches 45 μ m, and the resolution is better than that of commercial SU-8 photoresist (line width<2 μ m).
Application: Used for MEMS device manufacturing, with interlayer alignment accuracy of ± 0.1 μ m.
Photochromic coating L (intelligent packaging)
Color changing mechanism: 4-methylcatecho is blended with spiropyran and undergoes cis trans isomerization after UV irradiation, resulting in a color change from colorless to blue.
Commercialization prospects: Used for drug shelf life indication labels, with a color change threshold error of less than 5%.

Agricultural Chemicals and Agricultural Technology

Fungicide Enhancer
Methyl substitution enhances the permeability of compounds in the plant cuticle and improves the efficacy of fungicides.
Example analysis:
Enhancer M (Azoxystrobin Companion)
Compound ratio: 4-methylcatecho and azoxystrobin are compounded in a ratio of 1:5.
Field experiment: The control effect of cucumber downy mildew increased from 65% to 88%, reducing pesticide use by 30%.
Mechanism of action: Enhancers disrupt the integrity of pathogenic bacterial cell membranes and promote the delivery of azoxystrobin to the target site.
Herbicide safety agent
Selective protection of crops from herbicide damage and improvement of crop safety.
Example analysis:
Safety agent N (corn specific)
Chemical structure: derivative of 4-methylcatecho sulfonate.
Effect: When applied together with nicotinuron, the survival rate of corn seedlings increased from 30% to 85%, and the weed control effect remained at 90%.
Biochemical mechanism: Inducing crop GST enzyme activity and accelerating herbicide metabolism into non-toxic products.


Plant growth regulators
Simulate auxin activity to promote root development and stress resistance.
Example analysis:
Regulator O (anti stress growth promoter)
Synthesis method: 4-Methylcatecho is linked to naphthylacetic acid through ester bonds.
Biological activity: The lateral root density in Arabidopsis increased by 2.1 times, and the survival rate under drought stress increased by 40%.
Commercial product: Registered as a fertilizer additive, recommended dosage is 20-30 mg/L.
Pesticide slow-release agents
Utilizing the coordination effect between catechol and metal ions to achieve controlled release.
Example analysis:
Extended release agent P (avermectin carrier)
Preparation process: 4-Methylcatecho modified mesoporous silica with a drug loading of 32%.
Release characteristics: Release half-life of 28 days in pH 5.5 soil (simulating rhizosphere environment), reducing leaching losses.
Effectiveness evaluation: Extend the duration of nematode control by 40 days.

Organic synthesis and fine chemicals

Total synthesis of natural products
As a key intermediate involved in the construction of complex molecules.
Example analysis:
Compound Q (anti-tumor natural product analogue)
Synthesis route: 4-Methylcatecho is subjected to Diels Alder reaction to construct a hydrogenated naphthalene skeleton, which is then subjected to Baeyer Villiger oxidation and reduction rearrangement to obtain the target molecule.
Key steps: The oxidation state control of catechol requires low temperature (-78 ℃) and strict anhydrous conditions, with a yield of 42%.
Biological activity: for MDA-MB-231 breast cancer cell IC ≮ ₀=0.3 μ M, the mechanism of action is tubulin inhibitor.
Synthesis of essence and fragrance
Methyl substitution brings a unique vanilla aroma with low threshold and long-lasting fragrance.
Example analysis:
Spice R (Cream Aroma Type)
Synthesis pathway: 4-methylcatechol is acetylated and selectively reduced to obtain 4-methylguaiacol.
Application: It is used in dairy essence formula, and 0.5-2 ppm can give rich cream aroma.
Market data: The global annual demand is about 800 tons, mainly used for baked goods and ice cream.


Dye intermediates
The coordination ability of catechol is used for the synthesis of metal complex dyes.
Example analysis:
Dye S (red dye)
Synthesis method: 4-methylcatecho is complexed with Cr ³ ⁺ under acidic conditions to generate a deep red dye.
Dyeing performance: The color fastness to dyeing wool fabric reaches 4-5 levels, and the sun fastness reaches 6 levels.
Environmental friendliness: Does not contain banned aromatic amines and meets Oeko Tex Standard 100 standards.
Liquid Crystal Materials
As a side chain liquid crystal element, it endows the material with temperature responsiveness.
Example analysis:
Liquid crystal compound T
Molecular design: 4-methylcatecho is connected to the cyanobiphenyl liquid crystal nucleus through flexible spacer groups.
Phase transition behavior: Clearing point=125 ℃, liquid crystal phase temperature range Δ T=42 ℃.
Application: Used for thermochromic displays with a response speed of<10 ms.

Analysis, Detection, and Sensors

Metal ion detection
The fluorescence quenching properties of catechol are used for selective detection of heavy metal ions.
Example analysis:
Probe U (Fe ³ ⁺ detection)
Design principle: Quantum dots modified with 4-methylcatecho, with Fe ³ ⁺ quenching fluorescence through electron transfer.
Performance parameters: detection limit of 0.5 nM, selectivity coefficient (Fe ³ ⁺/other metals)>100.
Practical application: Used for online monitoring of drinking water, with a detection frequency of once every 5 minutes.
Biomolecular Sensors
Utilize the redox activity of catechol to detect enzymatic reaction products.
Example analysis:
Sensor V (glucose detection)
Preparation process: Electropolymerization of 4-methylcatecho on the surface of Prussian blue modified electrode.
Detection performance: Linear range of 0.1-10 mM, sensitivity of 12.5 μ A · mM ⁻¹· cm ⁻ ², response time<3 s.
Clinical application: used for sweat glucose monitoring of diabetes patients, with correlation coefficient R ²=0.992.


Environmental pollutant detection
The coordination ability of catechol is used for detecting pollutants such as organophosphorus pesticides.
Example analysis:
Reagent kit W (for oxygen phosphorus detection)
Reaction principle: 4-methylcatecho forms a purple complex with Fe ³ ⁺, which competes with oxygen phosphorus through phosphatase hydrolysis to produce phenolic coordination compounds.
Detection limit: 0.1 μ g/L, suitable for rapid screening of organic phosphorus residues in vegetable samples.

The structure of 4-methylcatechol contains two phenolic hydroxyl units, which exhibit strong acidity and can undergo acid-base neutralization reactions with common alkaline substances. Under alkaline conditions, this compound can undergo nucleophilic substitution reactions with common alkyl halide compounds to obtain corresponding ether derivatives. Due to the adjacent positions of the two hydroxyl groups on the benzene ring, it can be used to synthesize cyclic aromatic ether compounds.

Dissolve 4-methylcatechl (5.0 g, 40.28 mmol) in N at 0 ° C in a dry reaction flask, N-dimethylformamide (DMF) was added to the reaction mixture, followed by the addition of imidazole (13.7 g, 201.39 mmol) and t-butyldimethylchlorosilane (TBSCl) (18.2 g, 120.83 mmol) under nitrogen. Restore the obtained reaction mixture to room temperature and stir the reaction overnight at room temperature. After complete reaction, add ice water to the reaction mixture and extract it three times with dichloromethane (3 × 150 mL). Combine all organic phases and wash with saturated NaCl solution. Separate the organic layer and dry it on anhydrous sodium sulfate. Filter to remove the desiccant and concentrate the resulting filtrate under vacuum to obtain the target product molecule.
4-Methylcatechol is a remarkable compound with diverse applications in industry, medicine, and environmental science. Its unique chemical properties, including redox activity and metal chelation, underpin its utility in photography, hair dyes, and pollution control. Meanwhile, its biological activities-such as antioxidant, anti-inflammatory, and antimicrobial effects-highlight its potential in therapeutic development.
As research progresses, sustainable synthesis methods and novel applications (e.g., nanotechnology, green chemistry) will further expand the horizons of 4-MC. However, safety considerations must remain paramount to ensure responsible handling and minimize environmental impact.
By continuing to explore the multifaceted roles of 4-methylcatechol, scientists and industries can unlock new opportunities for innovation while addressing global challenges in health and sustainability.
Frequently Asked Questions
What is the structure of 4-Methylcatechol?
+
-
4-Methylcatechol is an organic compound with the formula CH 3C 6H 3(OH) 2 A white solid, it is one of the isomers of methylbenzenediol. Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
What is the solubility of 4-Methylcatechol?
+
-
4-Methylcatechol is soluble in organic solvents such as ethanol, DMSO, and dimethyl formamide. The solubility of 4-methylcatechol in these solvents is approximately 3, 20, and 25 mg/ml, respectively.
What is another name for 4-methylphenol?
+
-
P-Cresol
p-Cresol. para-Cresol, also 4-methylphenol, is an organic compound with the formula CH3C6H4(OH). It is a colourless solid that is widely used intermediate in the production of other chemicals. It is a derivative of phenol and is an isomer of o-cresol and m-cresol.
How to remove impurities from coal?
+
-
The majority of coal cleaning processes use upward currents or pulses of a fluid such as water to fluidize a bed of crushed coal and impurities. The lighter coal particles rise and are removed from the top of the bed. The heavier impurities are removed from the bottom.
What chemical is used to clean coal?
+
-
MCHM is a colorless foam used to clean coal, removing impurities and separating it from other geological products.
Hot Tags: 4-methylcatechol cas 452-86-8, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, tetravisc, CAS 2189692 44 0, 11 bromo benzo b naphtho 1 2 d furan, CAS 890042 13 4, tetramisole hydrochloride powder, CAS 1247053 55 9