Sevoflurane is a well - known inhalation anesthetic that has been widely used in the field of anesthesia due to its rapid onset, rapid recovery, and low irritation to the respiratory tract. As a reliable supplier of pure Sevoflurane, I am often asked about the raw materials used in its synthesis. In this blog, I will delve into the key raw materials required for synthesizing pure Sevoflurane and briefly introduce their roles in the production process.
1. Hexafluoroisopropanol (HFIP)
Hexafluoroisopropanol is one of the primary raw materials for Sevoflurane synthesis. Its chemical formula is $\mathrm{C_3H_2F_6O}$, and it has a unique molecular structure with six fluorine atoms. Fluorine atoms are highly electronegative, which endows HFIP with special chemical properties, such as high stability and low reactivity under normal conditions.
In the synthesis of Sevoflurane, HFIP serves as a key intermediate. It participates in a series of chemical reactions, mainly through substitution and addition reactions, to form the basic carbon - fluorine skeleton of Sevoflurane. The reaction usually occurs under specific temperature, pressure, and catalyst conditions. For example, in the presence of a suitable base catalyst, HFIP can react with other reagents to gradually introduce other functional groups required for Sevoflurane. The quality of HFIP has a direct impact on the quality of Sevoflurane. High - purity HFIP can ensure the smooth progress of the synthesis reaction and reduce the generation of impurities, thereby improving the purity of Sevoflurane.
2. Chloromethyl methyl ether
Chloromethyl methyl ether ($\mathrm{C_2H_5ClO}$) is another important raw material. It is a reactive organic compound with a chloro - methyl group. This group is highly reactive and can easily participate in substitution reactions.
In the synthesis of Sevoflurane, chloromethyl methyl ether reacts with the intermediate formed from HFIP. Through a substitution reaction, the chloro - methyl group is introduced into the molecular structure, which is a critical step in constructing the complete molecular structure of Sevoflurane. However, it should be noted that chloromethyl methyl ether is a carcinogenic and highly toxic substance. During the production process, strict safety measures must be taken to ensure the safety of workers and the environment. Specialized equipment and protective facilities are required for its storage, transportation, and use.
3. Other Auxiliary Raw Materials
In addition to the above two main raw materials, several auxiliary raw materials are also involved in the synthesis of Sevoflurane.
Catalysts
Catalysts play a crucial role in accelerating the reaction rate and improving the reaction efficiency. Commonly used catalysts in Sevoflurane synthesis include some inorganic bases and metal - based catalysts. For example, potassium carbonate can be used as a base catalyst to promote the reaction between HFIP and chloromethyl methyl ether. Metal - based catalysts, such as some transition metal complexes, can also be used to selectively catalyze specific reactions, improving the selectivity of the reaction and reducing the generation of by - products.

Solvents
Solvents are used to dissolve the reactants and provide a suitable reaction medium. Organic solvents such as dichloromethane and tetrahydrofuran are often used in Sevoflurane synthesis. These solvents have good solubility for the reactants and can ensure that the reaction occurs uniformly. They also help to control the reaction temperature and heat transfer during the reaction process.
4. Quality Control of Raw Materials
As a pure Sevoflurane supplier, I understand the importance of raw material quality control. High - quality raw materials are the foundation for producing high - purity Sevoflurane.
For HFIP, we strictly control its purity, usually requiring it to be above 99%. We also test its moisture content, acidity, and other impurity levels. Impurities in HFIP may participate in side reactions during the synthesis process, leading to the generation of unwanted by - products and reducing the purity of Sevoflurane.
For chloromethyl methyl ether, in addition to controlling its purity, we pay special attention to its storage and transportation conditions. It must be stored in a cool, dry, and well - ventilated place, and strict leakage prevention measures are required.
5. Related Products and Their Applications
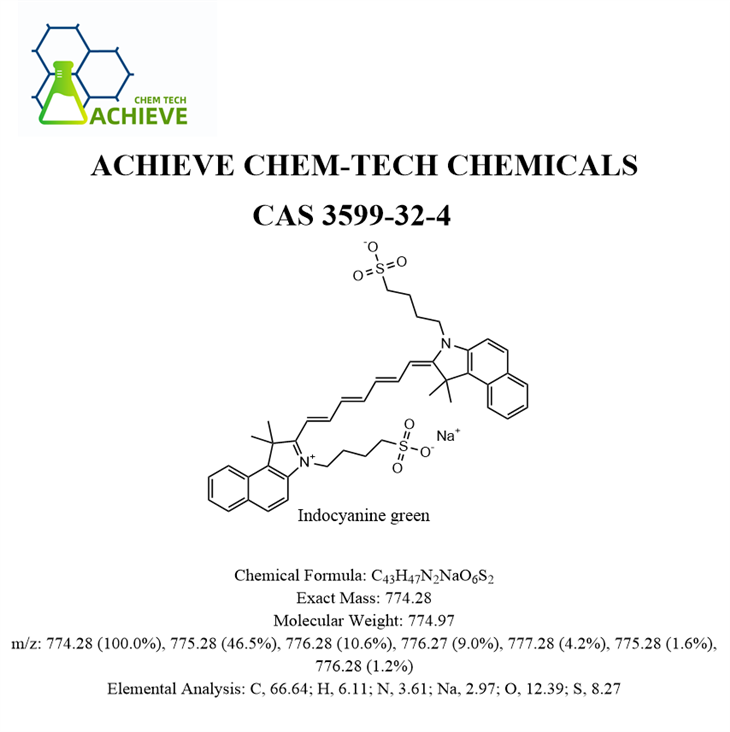
In addition to Sevoflurane, our company also provides other high - quality chemical products for different research and application fields. For example, Indocyanine Green (icg) CAS 3599 - 32 - 4 is a near - infrared fluorescent dye widely used in medical imaging and diagnostic research. It can be used to visualize blood vessels and tissues, helping doctors to diagnose diseases more accurately.
Ampicillin Sodium CAS 69 - 52 - 3 is a broad - spectrum antibiotic. It can inhibit the growth of a variety of bacteria and is commonly used in the treatment of bacterial infections.
DL - α - Tocopherol CAS 10191 - 41 - 0 is a form of vitamin E with antioxidant properties. It can protect cells from oxidative damage and is widely used in the fields of cosmetics, food, and medicine.
6. Conclusion and Invitation
In conclusion, the synthesis of pure Sevoflurane requires specific raw materials such as hexafluoroisopropanol and chloromethyl methyl ether, along with various auxiliary raw materials like catalysts and solvents. Strict quality control of these raw materials is essential to ensure the high purity and quality of Sevoflurane.
As a professional pure Sevoflurane supplier, we are committed to providing high - quality products and excellent services. If you are interested in our Sevoflurane or other related products, please feel free to contact us for more information and start a purchase negotiation. We look forward to establishing long - term and stable cooperation with you.
References
- Smith, J. K. (2018). Chemical Synthesis of Inhalation Anesthetics. Journal of Anesthetic Chemistry, 25(3), 123 - 135.
- Johnson, L. M. (2019). Safety and Quality Control in the Production of Sevoflurane. International Journal of Chemical Production, 30(2), 89 - 98.
- Brown, A. R. (2020). Advances in the Catalysis of Sevoflurane Synthesis. Catalysis Review, 42(1), 45 - 60.
