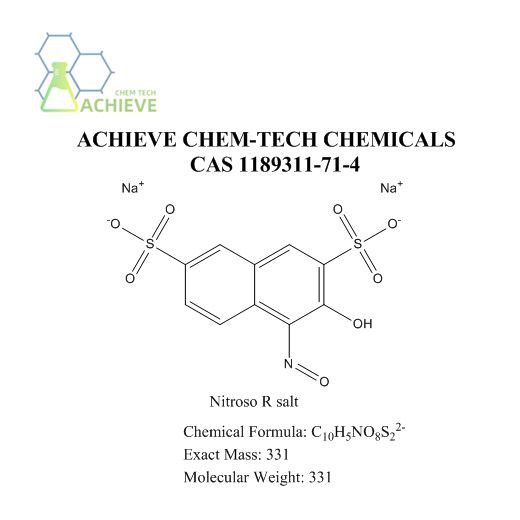
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of nitroso r salt cas 1189311-71-4 in China. Welcome to wholesale bulk high quality nitroso r salt cas 1189311-71-4 for sale here from our factory. Good service and reasonable price are available.
The appearance and color of Nitroso R salt depend on its molecular structure and environmental conditions. Molecular formula C10H7NNa2O9S2, CAS 1189311-71-4. Usually, it may take on a solid or liquid form, with colors ranging from colorless to light yellow or dark brown. These differences are mainly influenced by the "R" group in the molecule, as well as the interaction between nitroso and the "R" group. The solubility in water varies depending on the different "R" groups. Some nitroso salts have good water solubility, while others may only be slightly soluble in water or completely insoluble. In addition, it may also be soluble in organic solvents such as ethanol, ether, etc., depending on the hydrophilicity or hydrophobicity of the "R" group. Has a certain melting and boiling point. These values are influenced by intermolecular forces and molecular weight. Generally speaking, a higher melting point indicates stronger intermolecular forces, while the boiling point reflects the equilibrium of molecules between the gas and liquid states. It has multiple functions and applications and can play an important role in organic synthesis, drug research and development, agriculture, materials science, environmental science, and other fields. Due to the different specific "R" groups, their functions and properties may vary. Therefore, in practical applications, it is necessary to select suitable Nitroso R for research and application based on specific needs and conditions.

|
Chemical Formula |
C10H7NNa2O9S2 |
|
Exact Mass |
394.94 |
|
Molecular Weight |
395.26 |
|
m/z |
394.94 (100.0%), 395.94 (10.8%), 396.93 (9.0%), 396.94 (1.8%), 395.94 (1.6%) |
|
Elemental Analysis |
C, 30.39; H, 1.79; N, 3.54; Na, 11.63; O, 36.43; S, 16.22 |
We don't do anything shame on Great Chemistry! Don't trust the unreasonable price, please compare the price in the same level quality!
|
|
|

Nitroso R salt (chemical name: 1-nitroso-2-naphthol-3,6-disulfonic acid disodium salt, CAS number: 525-05-3), also known as nitroso red salt or cobalamin, is a golden yellow crystalline solid that is soluble in water and slightly soluble in methanol and ethanol. Its chemical properties are stable and have wide applications in analytical chemistry, organic synthesis, and industrial fields.
1. Trace determination of cobalt ions (Co ³ ⁺/Co ² ⁺)
It is one of the most sensitive reagents for detecting cobalt ions, and its detection principle is based on colorimetric reaction and photometry:
Color reaction: Under weakly acidic conditions (pH 4-6), it forms a stable orange red complex with cobalt ions, with a maximum absorption wavelength of 372nm.
Application of photometry: By measuring the absorbance of the complex with a spectrophotometer, the cobalt content can be quantitatively analyzed with a detection limit as low as 0.01 μ g/mL.
Standard Method: This reagent has been included in national standards such as GB/T 13747.8-1992 (Zirconium Alloy), GB/T 6730.38-2017 (Iron Ore), and GB/T 223.22-1994 (Steel and Alloy) for accurate determination of cobalt content in materials.


2. Multi metal ion detection and masking
Determination of Iron (Fe ² ⁺), Ruthenium (Ru ³ ⁺), and Potassium (K ⁺): It can be used as a color reagent to achieve selective detection by adjusting the pH value of the solution. For example, under pH 2-3 conditions, it forms a yellow complex with iron ions, and the absorbance is linearly related to the iron concentration.
The masking effect in nickel (Ni ² ⁺) determination: During nickel ion determination, it can form more stable complexes with cobalt ions, thereby eliminating the interference of cobalt on nickel determination and improving analytical accuracy.
Other metal ion detection: This reagent can also be used for color detection of silver (Ag ⁺), barium (Ba ² ⁺), calcium (Ca ² ⁺), lead (Pb ² ⁺) ions, and selective reactions can be achieved by adjusting reaction conditions.
3. Coordination titration indicator
Nickel (Ni ² ⁺) and copper (Cu ² ⁺) titration: In EDTA coordination titration, they can be used as metal indicators. For example, in an ammonia buffer solution with pH 9-10, it forms a red complex with nickel ions. When titrated to the endpoint, EDTA captures the nickel ions from the complex, and the solution changes from red to yellow.
Sensitivity and selectivity: This indicator shows a significant change in the titration endpoint of nickel ions and is not affected by common coexisting ions such as zinc and magnesium, making it suitable for complex sample analysis.

Organic synthesis field: Key intermediates and catalysts

1. Synthesis of dye intermediates
Can serve as a key intermediate for the synthesis of azo dyes. For example, coupling with aniline diazonium salts can generate azo dyes with bright colors, which are widely used in the textile, leather, and paper industries.
Reaction mechanism: Its nitroso group (- NO) undergoes coupling reaction with the diazonium salt coupling group (- N ₂⁺), generating azo bonds (- N=N -) and forming the dye molecular skeleton.
Product advantages: Dyes synthesized through nitroso-R salts have high color fastness and strong light resistance, meeting the needs of high-end textiles.
2. Synthesis of pharmaceutical intermediates
In drug synthesis, it can be used to prepare compounds with biological activity. For example, it can react with amino compounds to generate nitrosamine derivatives with anti-tumor or antibacterial activity.
Reaction conditions: Under low temperature (0-5 ℃) and acidic conditions, nitroso R salt undergoes nitrosation reaction with amino compounds to produce the target product.
Application case: In the synthesis of an anti-tumor drug intermediate, nitroso-R salt was used as a nitrosating reagent, significantly improving the selectivity and yield of the reaction.

Industrial field: Functional additives and environmental applications

1. Gasoline anti glue agent
In gasoline, it can be used as an anti gelling agent to prevent unsaturated hydrocarbons from oxidizing and forming tree like deposits, thereby maintaining engine cleanliness.
Mechanism of action: The nitroso (- NO) of nitroso-R salt undergoes an addition reaction with the double bond of unsaturated hydrocarbons, generating stable nitrogen oxides and blocking the oxidative chain reaction.
Application effect: Adding 0.01% -0.05% nitroso-R salt can improve the storage stability of gasoline by more than 30% and reduce the problem of engine fuel injector blockage.
2. Metal preservatives
Its sulfonic acid group can form chelates with metal ions, forming a protective film on the metal surface to prevent corrosion.
Application scenario: In the surface treatment of metals such as steel and copper, nitroso-R salt solution can be used as a pre-treatment agent for anti-corrosion coatings to improve the adhesion and corrosion resistance of the coating.
Technical advantages: Compared with traditional chromate preservatives, it is non-toxic and environmentally friendly, and meets the requirements of the RoHS directive.
3. Wastewater treatment agents
In the treatment of electroplating wastewater, it can be used as a precipitant for heavy metal ions such as nickel and copper.
Reaction principle: Under alkaline conditions, it forms insoluble chelates with heavy metal ions and achieves wastewater purification through precipitation separation.
Treatment effect: The removal rate of nickel ions can reach over 95%, and the treated wastewater can meet the national discharge standards.

NITROSO R SALT MONOHYDRATE has become an indispensable key reagent in analytical chemistry, organic synthesis, and industrial fields due to its high sensitivity detection, multifunctional synthesis, and industrial application value. In the future, with breakthroughs in green chemistry and materials science, their application scope and efficiency will be further enhanced, providing strong support for technological innovation and industrial development.
Technical parameters and safety specifications
1. Physical and chemical properties
Solubility: Dissolve 2.5g in 100g of water when cold, 10g when hot; Slightly soluble in methanol and ethanol.
Melting point:>300 ℃.
Maximum absorption wavelength: 372nm (cobalt complex).
2. Quality control indicators
Purity: ≥ 98% (AR grade).
Dry weight loss: ≤ 1.0%.
Cobalt sensitivity test: Qualified.
3. Safety and Storage
Hazard: Belongs to acute toxicity category 4 substance (GHS classification), harmful by swallowing, skin contact, and inhalation.
Protective measures: Wear protective gloves, goggles, and gas masks during operation.
Storage conditions: Sealed and stored in a cool and dry place, away from light and heat.

The diazotization reaction is an important method for preparing Nitroso R Salt, especially suitable for compounds containing aromatic amine groups. This reaction process involves converting aromatic amines into corresponding diazonium salts, which can then further react with various nucleophilic reagents to generate the final nitroso compounds.
1. Basic Principles
The diazotization reaction is a reaction between aromatic amines and nitrite under low temperature and acidic conditions, producing diazonium salts. Diazo salt is an intermediate product that is very active and can further react with various nucleophilic reagents.
2. Chemical equation
(1) Diazotization reaction
Ar-NH₂ + HNO₂ → Ar-N₂⁺X⁻ + H₂O
Among them, Ar represents the aromatic ring, and X ⁻ represents anions (such as Cl ⁻, Br ⁻, etc.).
(2) Reactions with nucleophilic reagents (using phenols as an example)
Ar-N₂⁺X⁻ + PhOH → Ar-NO + PhX
Among them, Ph represents phenyl, and PhX is the phenolic salt generated by the reaction of phenol and diazonium salt.
3. Detailed steps
Firstly, suitable aromatic amines need to be prepared as starting materials. The structure of aromatic amines determines the properties of the resulting Nitroso R.
Dissolve sodium nitrite (NaNO2) in an appropriate amount of water to obtain an aqueous solution of sodium nitrite. Subsequently, slowly add dilute sulfuric acid or hydrochloric acid to convert sodium nitrite into nitrite (HNO2). This step requires strict control of the pH value, usually between 2-4.
Dissolve the aromatic amine in dilute acid and cool to 0-5 ℃ (in an ice bath). Then, slowly add nitrous acid solution while stirring. This step is an exothermic reaction, so it is necessary to control the droplet acceleration to prevent local overheating. After dripping, continue stirring for a period of time to ensure complete reaction.
After the reaction is completed, cool the reaction mixture to room temperature and extract diazonium salts with appropriate solvents (such as ether, acetone, etc.). After extraction, the organic phase is separated from the aqueous phase to obtain an organic phase containing diazonium salts.
React the diazonium salt obtained in the previous step with appropriate nucleophilic reagents (such as phenols, alcohols, thiols, etc.) under appropriate conditions to generate the final Nitroso R Salt. The reaction conditions for this step (such as temperature, pH value, etc.) need to be adjusted according to specific nucleophilic reagents.
Hot Tags: nitroso r salt cas 1189311-71-4, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, Phosphoric acid reagent, 4 5 Chloro 2 pyridylazo 1 3 phenylenediamine, Sodium Diethyldithiocarbamate, Indicator Reagent, diphenylcarbazide reagent, Mercury Reagent