Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of 3-aminophenylboronic acid pinacol ester cas 210907-84-9 in China. Welcome to wholesale bulk high quality 3-aminophenylboronic acid pinacol ester cas 210907-84-9 for sale here from our factory. Good service and reasonable price are available.
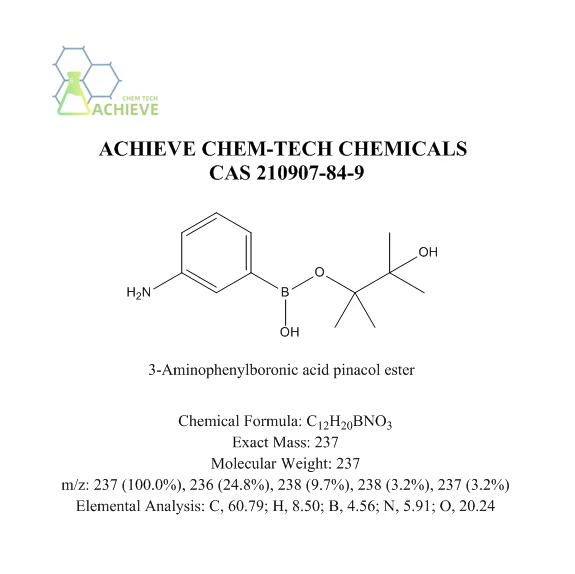
3-Aminophenylboronic acid pinacol ester is an organic compound with the molecular formula C12H18BNO2, CAS 210907-84-9, and a relative molecular weight of 225.09 g/mol. It is a solid state, usually in the form of white or similar white granular crystals. It may also be a white or similar white powder. It has a certain solubility in common organic solvents, such as dichloromethane, chloroform, ethanol, methanol, etc. It can dissolve in most polar solvents, but its solubility is lower in non-polar solvents. Relatively stable under conventional storage conditions, but contact with oxygen, moisture in the air, and strong oxidants should be avoided.

The crystal structure can be analyzed by methods such as X-ray diffraction. By analyzing the crystal structure, the molecular arrangement and spatial geometric configuration of the compound can be understood. Under high temperature conditions, decomposition or loss of crystalline water may occur. The thermal decomposition temperature and thermal stability depend on factors such as experimental conditions and purity. Commonly used as a reagent, ligand, or intermediate in chemical synthesis. It can participate in various organic synthesis reactions, such as boric acid coupling reaction, Suzuki Miyaura cross coupling reaction, etc. These reactions have wide applications in the preparation of drugs, pesticides, and functional molecules.
|
C.F |
C12H9Br |
|
E.M |
223 |
|
M.W |
233 |
|
m/z |
232 (100.0%), 234 (97.3%), 233 (13.0%), 235 (12.6%) |
|
E.A |
C, 61.83; H, 3.89; Br, 34.28 |
|
|
|
There are multiple positions in the product molecule that can form hydrogen bonds. The following are some possible positions for hydrogen bond formation:
The amino group (NH2) in the product molecule has hydrogen atoms that can form hydrogen bonds with appropriate receptor or donor molecules. Hydrogen atoms on amino groups can form hydrogen bonds with molecules with electronegative atoms such as oxygen, nitrogen, or fluorine. The formation of this hydrogen bond can be achieved through the interaction between free hydrogen in the amino group and atoms with higher electron density.
The hydroxyl group (OH) on the pinacol group can also form hydrogen bonds. The hydrogen on this hydroxyl group can form hydrogen bonds with atoms with lone pair electrons such as oxygen or sulfur.
The hydrogen atom (H) in the ester group can also participate in the formation of hydrogen bonds. For example, it can form hydrogen bonds with oxygen or nitrogen atoms in ligands or other molecules.

Some common uses of 3-Aminophenylboronic acid pinacol ester in drug research and development:
1. Drug molecular design:
3-aminophenylboronic acid piperone ester, as an intermediate in organic synthesis, is often used in drug research to design and synthesize new drug molecules. By introducing different functional groups into its structure, its biological activity, solubility, and stability can be changed, thereby optimizing the performance of drugs.
2. Drug screening: The use of a compound library composed of 3-aminophenylborate piperone ester and its derivatives for high-throughput screening (HTS) is an important step in drug development. By evaluating the interactions between these compounds and specific targets, candidate compounds with potential pharmacological effects can be screened.


3. Pharmacodynamic evaluation: 3-Aminophenylboronic acid piperone ester can be used in drug research to evaluate the efficacy and side effects of drugs. By studying its binding ability and selectivity with specific targets, the mechanism of action and potential clinical applications of drug molecules in vivo can be evaluated.
4. Drug delivery system: 3-aminophenylborate piperone ester can combine with carrier materials to form a drug delivery system, which is used to enhance the solubility, stability, and targeting of drugs. By controlling the release rate and action location of drugs in the body, the effectiveness of drug treatment can be improved and side effects can be reduced.
5. Tumor treatment: 3-aminophenylborate piperone ester can be used for the design and synthesis of anti-tumor drugs. For example, it can be introduced into small molecule compounds to inhibit the growth and spread of cancer cells. In addition, it can also be combined with targeted ligands to achieve selective targeted therapy of tumor tissue.
6. Anti-inflammatory drugs: 3-aminophenylborate piperone ester has good anti-inflammatory activity and can be used to develop and optimize anti-inflammatory drugs. By regulating the inflammatory signaling pathway and immune response, the symptoms of inflammatory diseases and the production of inflammatory mediators can be alleviated.


7. Immunomodulators: In the field of immunotherapy, 3-aminophenylboronic acid piperone ester can be used as an immune modulator. By regulating the activity and balance of the immune system, it can enhance the immune response, suppress autoimmune diseases, and regulate organ transplant rejection.
8. Anti infective drugs: 3-aminophenylborate piperone ester and its derivatives also have potential applications in the research of anti infective drugs. By interfering with the metabolic pathways of microorganisms or targeting specific infection related proteins, new antibacterial or antiviral drugs can be developed.

3-Aminophenylboronic acid pinacol ester, CAS number 210907-84-9, is an important organic compound that contains amino and boronic acid pinal ester groups in its molecular structure. This unique structure endows it with various excellent chemical properties, making it widely used in multiple fields.
1. As an important organic intermediate
Which alcohol ester plays a key intermediate role in organic synthesis. The boronic acid pinal ester group in its molecule has high reactivity and can participate in various types of chemical reactions, such as Suzuki coupling reactions. In Suzuki coupling reaction, it can react with halogenated aromatic hydrocarbons to generate aromatic compounds. This reaction has the advantages of mild reaction conditions, high yield, and good selectivity, and is widely used in fields such as drug synthesis, natural product synthesis, and preparation of functional materials.


2. Participate in other types of organic reactions
In addition to Suzuki coupling reactions, it can also participate in various other organic reactions. For example, it can undergo condensation reactions with carbonyl compounds such as aldehydes and ketones to generate corresponding imines or enamines. These imine or enamine compounds play an important intermediate role in organic synthesis and can be further converted into other useful organic molecules.
In addition, it can also undergo substitution reactions with some electrophilic reagents to introduce different functional groups, thereby achieving modification and alteration of the molecular structure. This reaction flexibility makes 3-amino-phenylboronic acid pinal ester a very useful reagent in organic synthesis.
1. As a biomaterial
Can be used as biomaterials for life science related research. The amino and boronic acid ester groups in its molecule can interact with specific groups in biomolecules, enabling labeling, detection, and separation of biomolecules.
For example, in the preparation of biosensors, they can be combined with biomolecules (such as proteins, nucleic acids, etc.) to form biosensors with specific recognition functions. This biosensor can be used to detect specific substances in biological samples, such as glucose, protein markers, etc. It has the advantages of high sensitivity, good selectivity, and easy operation.


2. Application in drug development
In the process of drug development, it also has important application value. It can serve as a precursor or intermediate for drug molecules, participating in the synthesis and structural modification of drugs. By modifying the molecular structure of drugs, the pharmacokinetic properties of drugs can be improved, the biological activity and selectivity of drugs can be enhanced, and more effective drugs can be developed.
For example, in the development of some anti-tumor drugs, the chemical reactions they participate in can introduce specific functional groups, enhance the binding ability of drugs with tumor cell targets, and improve the anti-tumor activity of drugs. Meanwhile, by modifying the structure of drug molecules, the toxic side effects of drugs can be reduced and the therapeutic index of drugs can be improved.
1. Fixed phase modification in chromatographic analysis
In chromatographic analysis techniques, it can be used to modify the stationary phase of chromatographic columns. Taking liquid chromatography as an example, bonding the compound to the surface of the stationary phase through appropriate chemical methods can endow the stationary phase with unique separation properties.


Due to the specific recognition and binding ability of boronic acid groups to compounds containing cis diol structures (such as sugars, nucleotides, etc.), modified stationary phases can achieve specific separation of these substances. In sugar analysis, traditional chromatographic methods are often difficult to effectively separate sugar molecules with similar structures. However, the modified stationary phase can achieve efficient separation and analysis of complex sugar mixtures based on the differences in the interaction between cis diols and boronic acid groups in sugar molecules, providing strong technical support for sugar biology research.
2. As an analytical reagent used for detecting specific substances
3-Aminophenylboronic acid pinacol ester can also be used as an analytical reagent to detect substances with specific functional groups. For example, when detecting compounds containing aldehyde groups, condensation reactions can occur with aldehyde groups to generate products with specific spectral properties such as fluorescence or absorption spectrum changes.


By monitoring the spectral changes of the solution before and after the reaction, such as the enhancement of fluorescence intensity or the shift of absorption peaks, quantitative detection of aldehyde compounds can be achieved. This method has the advantages of high sensitivity and good selectivity, and can be used for detecting aldehyde pollutants in air or water in environmental monitoring. It can also be used for the analysis of aldehyde metabolites in biological samples, providing important information for the research and diagnosis of related diseases.
Hot Tags: 3-aminophenylboronic acid pinacol ester cas 210907-84-9, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, Cerium sulfate powder, Consumable, 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID, CHICAGO SKY BLUE 6B, Iridium III chloride