Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of acetaldehyde solution cas 75-07-0 in China. Welcome to wholesale bulk high quality acetaldehyde solution cas 75-07-0 for sale here from our factory. Good service and reasonable price are available.
Acetaldehyde solution, also known as acetaldehyde, is an organic compound, CAS 75-07-0, The chemical formula is CH3CHO. Belonging to aldehyde ketone organic compounds, it is a colorless and transparent liquid with a pungent odor, volatile and flammable properties. It is easily soluble in water and can be mixed in any proportion with organic solvents such as ethanol, ether, benzene, gasoline, toluene, etc. It is mainly used as a reducing agent, fungicide and standard solution for colorimetric determination of aldehydes. Used in industry for manufacturing acetaldehyde, acetic acid, synthetic rubber, etc.
Widely used as raw materials, disinfectants, explosives, reducing agents for organic synthesis of acetic acid, acetic anhydride, butanol, polyacetaldehyde, synthetic rubber and other products, and can also be used to prepare standard solutions for formaldehyde determination by colorimetric method. The industrial production of acetaldehyde includes methods such as direct oxidation of ethylene, oxidation of ethanol, direct hydration of acetylene, dehydrogenation of ethanol, and hydrogenation of acetic acid. Its downstream products include pyridine, crotonaldehyde, and sorbic acid.

|
Chemical Formula |
C2H4O |
|
Exact Mass |
44 |
|
Molecular Weight |
44 |
|
m/z |
44 (100.0%), 45 (2.2%) |
|
Elemental Analysis |
C, 54.53; H, 9.15; O, 36.32 |
|
|
|

1. Catalytic oxidation of acetaldehyde
![]()
2. Acetaldehyde combustion

3. Silver mirror reaction
![]()
4. Acetaldehyde and newly prepared copper hydroxide

5. Acetaldehyde reacts with hydrogen to produce ethanol
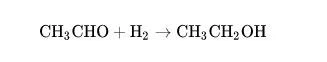
Acetaldehyde solution can be produced in many ways:
1. Ethylene direct oxidation method ethylene and oxygen are directly oxidized to synthesize crude acetaldehyde in one step through a catalyst containing palladium chloride, copper chloride, hydrochloric acid and water, and then the finished product is obtained by distillation.
2. Ethanol oxidation method acetaldehyde was prepared by air oxidative dehydrogenation of ethanol vapor at 300-480 ℃ using silver, copper or silver copper alloy mesh or particles as catalyst.
3. Acetylene direct hydration method acetylene and water are directly hydrated under the action of mercury catalyst or non mercury catalyst to obtain acetaldehyde. Due to the problem of mercury damage, it has been gradually replaced by other methods.
4. Ethanol dehydrogenation method in the presence of copper catalyst with cobalt, chromium, zinc or other compounds, ethanol is dehydrogenated to produce acetaldehyde.
5. Saturated hydrocarbon oxidation method. Consumption quota of raw materials: 610kg of 99% acetylene per ton of product produced by acetylene hydration; The ethanol oxidation method consumes 1200kg of 95% ethanol; The ethylene oxidation method (one-step method) consumes 710kg of 99% ethylene and 300m3 of oxygen (99%). Commercial industrial acetaldehyde, the purity of acetaldehyde by ethylene method is 99.7%, and the purity of acetaldehyde by ethanol method is 98%.


Acetaldehyde solution is a colorless, transparent, and volatile liquid with a pungent odor. The aldehyde group (- CHO) in its molecular structure endows it with high reactivity and can participate in various chemical reactions. It is widely used in chemical, pharmaceutical, food, daily chemical, agricultural and other fields.
Core application areas: Chemical intermediates and solvents

Acetic acid and acetate production
It is the main industrial raw material for acetic acid (CH3 COOH), which converts acetaldehyde into acetic acid through oxidation reactions (such as the Wacker method), and further synthesizes derivatives such as vinyl acetate and acetic anhydride. Acetic acid is used as an acidic regulator in textile printing and dyeing to improve the dyeing effect of fabrics; As an acidity regulator in the food industry, it is widely used in the production of seasonings and canned foods. Vinyl acetate is the core raw material for manufacturing cellulose acetate (such as film substrates and plastics), and acetaldehyde plays a key bridging role in this process.
Synthesis of Pentaerythritol
Acetaldehyde condenses with three molecules of formaldehyde under alkaline conditions to form pentaerythritol (C (CH ₂ OH) ₄), an important polyol used in the manufacture of resins, coatings, lubricants, and explosives (such as pentaerythritol tetranitrate). The derivatives of pentaerythritol are irreplaceable in fields such as aerospace and electronic packaging. Acetaldehyde, as the starting point of the reaction, directly determines the direction of the industrial chain extension.


Preparation of Pyridine and Its Derivatives
It reacts with ammonia under the action of a catalyst to produce pyridine, which can further be used to synthesize vitamin B3 (niacin), anti tuberculosis drug isoniazid, etc. Pyridine compounds occupy a core position in the pharmaceutical, pesticide, and dye industries. For example, pyridine reacts with chloroacetic acid to synthesize herbicide 2,4-D, and the participation of acetaldehyde makes the synthesis of these high value-added products possible.
Solvent and cleaning agent
It can dissolve resins, oils, and various organic substances, and is commonly used for metal cleaning, electronic component degreasing, and precision instrument decontamination. In semiconductor manufacturing, acetaldehyde is used to clean the residual photoresist on the surface of silicon wafers. Its low surface tension characteristics can penetrate micrometer level gaps, ensuring cleanliness meets nanometer level standards. Although some applications have been replaced due to toxicity issues, it is still difficult to completely replace them in high-precision cleaning scenarios.

Special Purpose: Pharmaceutical and Food Industry

Synthesis of pharmaceutical intermediates
Antibiotics and antiviral drugs: They participate in the synthesis of side chain structures of antibiotics such as penicillin and cephalosporins, and introduce specific functional groups through aldol condensation reactions to enhance drug activity. For example, after condensation and hydrolysis with cyanide ions and ammonia, the substance can be used to synthesize alanine, which can be further used to synthesize the antiepileptic drug gabapentin.
Vitamin production: Acetaldehyde is an important raw material for the synthesis of vitamin B ₁ (thiamine), which condenses with cyanoacetic acid to form a thiazole ring and ultimately forms vitamin B ₁ molecules. In addition, acetaldehyde is also used as an intermediate for the synthesis of vitamin A, β - ionone, and its reaction selectivity directly affects the yield of vitamin A.
Anesthetics and sleeping pills: They are chlorinated to form trichloroacetaldehyde, and their hydrate (chloral hydrate) has been widely used as a sedative hypnotic drug. Although it has been replaced by safer alternatives due to side effects, it still has applications in the field of veterinary medicine.


Food additives and essence
Seasoning agent: Trace amounts of this substance naturally exist in coffee, bread, and ripe fruits, giving them a special aroma. In industry, acetaldehyde is used to prepare fruit essence such as oranges, oranges and apples, and wine essence such as wine and rum. The concentration of acetaldehyde in the final flavored food is about 3.9~270 mg/kg.
Preservatives: can inhibit microbial growth and extend the shelf life of food. For example, in cheese production, acetaldehyde is used to prevent mold contamination while enhancing the flavor of the cheese.
Deinonylation process: It was once used for coffee deintercalation by extracting caffeine and distilling to recover the solvent. Although supercritical CO2 extraction technology has gradually become popular, its process is still retained in some regions.
Additives for daily chemical products
Shampoo and shower gel: As lubricants, they can improve the texture of products, making hair soft and skin smooth. Its low volatility ensures stability during product storage, and it also participates in the slow release system of essence to extend the fragrance retention time.
Cosmetics: Acetaldehyde is used to synthesize certain antioxidants, prevent oil oxidation and rancidity in formulations, and extend product shelf life. In addition, its derivatives (such as glyoxylic acid) are used as whitening ingredients in skincare products, reducing melanin production by inhibiting tyrosinase activity.


(1) Sp2 hybridization
The structures of aldehydes and ketones both contain carbon oxygen double bonds (- C=O, carbonyl). The carbon atom forms three sp2 hybridized orbitals with the oxygen atom and two other atoms, forming three sigma bonds located in the same plane with a bond angle of approximately 120 °. The remaining p orbital of the carbonyl carbon that does not participate in hybridization overlaps with one p orbital of the oxygen atom from the side to form a π bond, while the two p orbitals of the oxygen atom have two pairs of lone pair electrons.
Taking formaldehyde, which has the simplest structure, as an example, the carbon oxygen double bond and carbon hydrogen single bond lengths are 120.3 pm and 110 pm, respectively.

Due to the greater electronegativity of oxygen atoms compared to carbon atoms, the electron cloud in the carbon oxygen double bond tends to be biased towards oxygen atoms, resulting in a higher electron cloud density around them, while the electron cloud density of carbon atoms is lower. Therefore, carbonyl groups have polarity, and Acetaldehyde solution is a polar molecule, which also explains why acetaldehyde is easily soluble in polar solvents (similar solubility).
(2) Alpha hydrogen atom
① Weakly acidic
The alpha hydrogen atoms of aldehydes and ketones are very active for two main reasons: firstly, the electron withdrawing induction effect of carbonyl groups; The second is the hyperconjugation effect of alpha carbon hydrogen bonds on carbonyl groups.

Taking 2-methylcyclohexanone as an example, isotope exchange experiments have shown that the alpha hydrogen atom next to the carbonyl group has high activity and can be replaced by deuterium atoms under the action of deuterated sodium oxide (heavy sodium hydroxide, NaOD) and heavy water (D2O).

Although the alpha H activity of different carbonyl compounds varies, aldehydes have stronger acidity compared to alkanes, alkynes, and ketones of the same series. On the one hand, the steric hindrance of alkyl groups is greater than that of hydrogen atoms, and on the other hand, the hyperconjugation effect between alkyl groups and carbonyl groups reduces the positive charge of carbonyl carbons.

Note: p represents negative logarithm, and the smaller the pKa, the stronger the acidity.
② Tautomerism
Generally, most aldehydes and ketones have tautomers. Taking acetaldehyde as an example, there are tautomers between the ketone and enol forms. Due to the instability of the enol form structure, the ketone form structure of acetaldehyde accounts for almost 100%, with an equilibrium constant of about 6.0 × 10-5.

Note: The reason for the instability of the enol structure is that the presence of carbon carbon double bonds increases the density of the π electron cloud of the carbon atom. However, due to the strong electronegativity of oxygen, the electron cloud tends to approach the oxygen atom. This contradictory result leads to the instability of the enol structure.
③ Aldol condensation
Under the action of dilute alkaline solution, acetaldehyde molecules can undergo aldol condensation reaction at low temperature, where α - hydrogen atoms attack carbonyl oxygen atoms, and other functional groups combine with carbonyl carbon atoms to form β - hydroxybutyraldehyde, which doubles the number of carbon atoms.

(3) Nucleophilic addition
The positive charge of the carbon atom in the carbonyl structure is easily attacked by nucleophiles, and can undergo π - bond cleavage addition reactions in acidic and alkaline environments.

① Hydrocyanic acid
Hydrocyanic acid is a typical nucleophile that reacts with Acetaldehyde solution to produce 2-hydroxypropionitrile (α - hydroxynitrile). The reaction rate will be greatly accelerated under alkaline conditions because HCN, as a weak acid, is prone to generate cyanide negative ions (CN -) under alkaline conditions, thereby increasing the concentration of reactants; On the contrary, if carried out under acidic conditions, hydrogen ions undergo protonation with carbonyl groups, enhancing the electrophilicity of carbonyl carbons, which is not conducive to the progress of the reaction and slows down the reaction rate.

HCN + NaOH → NaCN + H2O
CH3CHO + HCN → CH3-CH(OH)-CN

In addition, 2-hydroxypropionitrile can be hydrolyzed under acidic conditions to produce 2-hydroxypropionic acid (commonly known as "lactic acid"). Therefore, the nucleophilic addition reaction of hydrogen cyanide can be used to synthesize hydroxy acids with an additional carbon atom.
CH3-CH(OH)-CN + 2H2O + H+ → CH3-CH(OH)-COOH + NH4+
② Sodium bisulfite
Acetaldehyde and excess saturated sodium bisulfite solution can undergo a nucleophilic reaction to form sodium bisulfite adducts without the need for a catalyst.
CH3CHO + NaHSO3 → CH3-CH(OH)-SO3Na

The sodium bisulfite adduct (sodium alpha hydroxysulfonate) is easily soluble in water but difficult to dissolve in organic solvents, so it diffuses from the organic phase to the aqueous phase to form crystals. Therefore, this reaction can be used to separate aldehydes from organic compounds that are insoluble in water.
Note: Sodium alpha hydroxysulfonate reacts with sodium cyanide, and the sulfonic acid group can be replaced by a cyanide group to form alpha hydroxynitrile (nitrile alcohol), thereby avoiding the production of highly toxic and volatile hydrogen cyanide.
CH3-CH(OH)-SO3Na + NaCN → CH3-CH(OH)-CN + Na2SO3
③ Format reagent
Acetaldehyde can react with Grignard reagent (commonly known as "Grignard reagent", abbreviated as "RMgX") in the presence of anhydrous ether, first generating magnesium substituted compounds (intermediate products), and then hydrolyzing under acidic conditions to directly generate alcohols. This reaction is also one of the ways to synthesize alcohols through nucleophilic addition reactions, similar to organic lithium reagents.

Taking the reaction of cyclohexane as a hydrocarbon based reagent with acetaldehyde as an example.
CH3CHO + C6H11-MgX → H11C6-CH(OH)-CH3

Note: Format reagent was synthesized by French scientist Francois Auguste Victor Grignard (1871-1935) in 1901. It is an organic magnesium reagent formed by reacting organic halogen (chlorine, bromine, iodine) compounds (halogenated alkanes, active halogenated aromatic hydrocarbons) with magnesium metal in dry anhydrous ether.
④ Alcohol
Alcohols also have affinity, and under the catalysis of acids such as p-toluenesulfonic acid and hydrogen chloride, they can undergo nucleophilic addition reactions with acetaldehyde to form unstable hemiacetals, which can then be removed from one molecule of water to form acetals.

The specific reaction mechanism is as follows: firstly, the carbonyl and hydrogen ions undergo protonation to form oxonium ions, which increases the electrophilicity of the carbonyl carbon atom; Secondly, during addition reactions with alcohols, protons are lost, resulting in the formation of unstable hemiacetals; Subsequently, it combines with H+to form oxonium ions for dehydration; Finally, it reacts with alcohol to form a more stable aldehyde, and the overall result is that one molecule of aldehyde ketone can react with two molecules of alcohol to form one molecule of aldehyde.

Taking methanol as an example, it can react with acetaldehyde to produce dimethoxyethane (aldehyde).
CH3CHO + 2CH3OH → (H3CO)2-CH-CH3 + H2O
⑤ Water
In acidic environments, water can undergo nucleophilic addition reactions with acetaldehyde to produce dihydroxyethane (diol).

CH3CHO + H2O → (HO)2-CH-CH3
Note: The molecular structure of two hydroxyl groups connected by the same carbon atom lacks thermodynamic stability and tends to revert back to aldehydes and ketones upon dehydration, indicating that the addition reaction between water and carbonyl is a reversible reaction with equilibrium biased towards the reactant side.
⑥ Ammonia and its derivatives
All aldehydes and ketones can undergo nucleophilic addition reactions with ammonia and its derivatives (such as hydroxylamine, hydrazine, phenylhydrazine, semicarbazide, etc.), producing stable products such as oxime, hydrazone, phenylhydrazone, and urea. However, the products obtained from the reaction with ammonia are unstable.

Taking 2,4-dinitrophenylhydrazine as an example, the chemical equation for the reaction with acetaldehyde and dehydration to generate 2,4-dinitrophenylhydrazone is shown in the figure.

Note: Oxime, hydrazone, and urea are generally stable crystals with a fixed melting point. Hydrolysis in acidic environments can restore the carbonyl structure. Therefore, these nucleophilic reactions can be used to identify and purify aldehydes and ketones.
Products of certain amine derivatives reacting with carbonyl groups

(4) Oxidation reaction
① Color reaction
The aldehyde group of Acetaldehyde solution molecules can be oxidized to - COO - by Fehling reagent and Tollens reagent, producing brick red precipitate (Cu2O) and silver mirror (elemental Ag), respectively. The principle of identifying aldose (reducing sugar) lies in this, and the reaction that occurs with Tollens reagent (requiring heating) is also known as the "silver mirror reaction" [3].
CH3CHO + 2Ag(NH3)2OH → 2Ag↓+ 3NH3↑+ 2H2O + CH3COONH4
CH3CHO + 2Cu(OH)2 → Cu2O↓+ 2H2O + CH3COOH
Note: Fehling reagent and Tollens reagent are both reagents that can identify reducing substances. The former is generally composed of sodium hydroxide (NaOH) and copper sulfate (CuSO4) solution, invented by German chemist Herman von Fehling (1812-1885) in 1849; The latter can only be prepared in situ, and its main component is an ammonia solution of silver nitrate, namely Ag (NH3) OH, also known as "silver ammonia solution", invented by German chemist Bernhard Tollens (1841-1918) in the 19th century.
② Strong oxidizing agent
Due to the reducibility of aldehyde groups, they can be oxidized to acetic acid by inorganic strong oxidant potassium permanganate. Under acidic conditions, potassium permanganate is reduced to divalent manganese ions, resulting in fading of the deep purple solution; Under alkaline conditions, it is reduced to IV valent manganese dioxide, and the phenomenon is that the deep purple solution fades, producing a brownish black precipitate. The ion equation is as follows.
5CH3CHO + 2MnO4- + 6H+ →2Mn2+ + 5CH3COOH +3H2O
3CH3CHO + 2MnO4- + H2O →2MnO2↓+ 3CH3COOH + 2OH-
Note: Potassium permanganate has stronger oxidizing properties in acidic environments and is reduced to lower valence compounds. Similar strong oxidants include potassium dichromate (K2Cr2O7), chromic acid (H2CrO4), hydrogen peroxide (H2O2), etc.
③ Catalytic oxidation
Under the catalysis and heating conditions of copper metal, acetaldehyde can be oxidized by oxygen to acetic acid. Firstly, copper reacts with oxygen under heating conditions to form copper oxide, which then acts as an oxidant and reacts with acetaldehyde to reduce itself to elemental copper (catalyst) [2] [20-28].
2Cu + O2 → 2CuO
CH3CHO + CuO → Cu + CH3COOH
2CH3CHO + O2 → 2CH3COOH
④ Oxygen (combustion)
Acetaldehyde, as an organic compound, can be burned in oxygen to produce carbon dioxide and water (completely oxidized).
2CH3CHO + 5O2 → 4CO2 + 4H2O
(5) Reduction reaction
Acetaldehyde contains unsaturated carbon oxygen double bonds (- C=O), which can be reduced to hydroxymethyl (- CH2OH) by reducing agents.
① Catalytic hydrogenation
Acetaldehyde can be reduced to ethanol by hydrogen gas under the action of metal catalysts such as nickel and palladium.
CH3CHO + H2 → CH3CH2OH
② Metal hydride
Acetaldehyde can be reduced to ethanol by metal hydrides (strong reducing agents) such as lithium aluminum hydride, sodium borohydride, etc. under anhydrous ether conditions.
CH3CHO + LiAlH4 +2H2O → CH3CH2OH + LiAlO2 + 3H2↑
CH3CHO + NaBH4 + 3H2O → CH3CH2OH + NaBO3 + 4H2↑
③ Clemmensen restoration
Under the action of HCl and zinc mercury (Zn Hg), the aldehyde group of acetaldehyde can be reduced to methyl, that is, acetaldehyde is reduced to ethane under strong acidic conditions. This reaction is suitable for the reduction of carbonyl compounds that are sensitive to alkali.

Hg does not participate in the Clemmensen reduction reaction, but acts as a catalyst. After forming a mercury amalgam (Zn Hg) alloy with zinc, the activity of zinc is increased due to the formation of an electric couple in the alloy, thereby promoting the reaction.
Zinc amalgam can be prepared by reacting zinc powder/zinc particles with mercury salt (HgCl2) in dilute hydrochloric acid solution. Elemental zinc can reduce divalent mercury ions to elemental mercury, and then mercury forms a mercury amalgam on the surface of zinc, and the reduction reaction occurs on the activated surface of zinc.
Note: The Clemmensen reduction reaction was discovered by Danish chemist Erik Christian Clemmensen (1876-1941) in 1913.
④ Kishner Wolff Huang Minglong restoration
Aldehydes and ketones can react with anhydrous hydrazine to form hydrazones (C=NNHR), which can then be decomposed into nitrogen gas by heating with anhydrous ethanol and sodium ethoxide in a high-pressure vessel to 180-200 ℃. Carbonyl groups are reduced to methylene groups under alkaline conditions, and this reaction is called the Wolff Kishner reduction reaction.

Huang Minglong (1898-1979), a famous organic chemist in China and an academician of the CAS Member, improved the reaction. A higher yield could be achieved by replacing anhydrous hydrazine with an aqueous solution of hydrazine. That is, aldehyde or ketone, sodium hydroxide, aqueous solution of hydrazine, and high boiling solvent (diethylene glycol, diethylenegly clo, HOCH2CH2OCH2CH2OH) were co heated to form hydrazone, and then excess hydrazine and water were evaporated. After reaching the decomposition temperature of hydrazone, the reaction was refluxed to the completion of the reaction. This reaction is called Wolff Kishner Huang Minglong reaction.
Taking acetaldehyde as an example, it can be reduced to ethane through the Wolff Kishner and Wolff Kishner Huang Minglong reactions, which is suitable for the reduction of acid sensitive carbonyl compounds.
(6) Hydrogen bond
Acetaldehyde can form hydrogen bonds in water, which is another reason why acetaldehyde (lower aldehydes) is easily soluble in water.

FAQ
What is the smell of acetaldehyde?
HOW DOES ACETALDEHYDE SMELL OR TASTE? Typical sensory descriptors associated with acetaldehyde include green (Granny Smith) apples, pumpkin flesh/seed, unripe avocado, and latex paint. Acetaldehyde is somewhat unique in this regard; that the "character" of its aroma can change as its concentration changes.
What is the formula for acetaldehyde?
Acetaldehyde - C2H4O
Acetaldehyde, ethanal, is a colourless, water-soluble, burnable fluid that has the chemical formula C2H4O and the structural formula CH3CHO. Acetaldehyde has a low boiling point at 21 °C.
What drinks are high in acetaldehyde?
In addition, acetaldehyde is contained in beverages such as tea and soft drinks (0.2-0.6 ppm), beer (0.6-24 ppm), wine (0.7-290 ppm) and spirits (0.5-104 ppm) 2).
Hot Tags: acetaldehyde solution cas 75-07-0, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID, Iridium III chloride, Consumable, CHICAGO SKY BLUE 6B, Cerium sulfate powder









