Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of bromothymol blue powder cas 76-59-5 in China. Welcome to wholesale bulk high quality bromothymol blue powder cas 76-59-5 for sale here from our factory. Good service and reasonable price are available.
Bromothymol blue powder, also known as bromothymol blue, is an acid-base indicator and an adsorption indicator, with a chemical formula of c27h28o5sbr2. Light rose colored crystalline powder, easily soluble in ethanol, ether, methanol and dilute alkali hydroxide solution. Slightly soluble in benzene, toluene and xylene, slightly soluble in water, almost insoluble in petroleum ether. It is blue in alkaline solution and yellow in acidic solution. BTB in English. It is often used as a respiratory reagent of aquatic organisms in biological experiments.

|
Chemical Formula |
C27H28Br2O5S |
|
Exact Mass |
622 |
|
Molecular Weight |
624 |
|
m/z |
624 (100.0%), 622 (51.4%), 626 (48.6%), 625 (19.5%), 627 (14.2%), 623 (10.6%), 625 (9.7%), 626 (4.5%), 623 (4.4%), 626 (3.9%), 624 (2.3%), 628 (2.2%), 628 (2.0%), 624 (1.1%), 624 (1.0%), 626 (1.0%) |
|
Elemental Analysis |
C, 51.94; H, 4.52; Br, 25.59; O, 12.81; S, 5.13 |
|
|
|
In biological experiments, it is often used as a respiratory reagent for aquatic organisms. The short-term life of organisms in the water containing this reagent is not affected. The water added with 0.1% BTB solution generally appears light green. If the content of carbon dioxide in the water increases and becomes acidic, the water will change from light green to yellow.
Identifying the presence of CO2 is a bit like clarifying lime water, but the accuracy is higher than that of lime water, and there is no interference of SO2. Bromothymol blue aqueous solution can identify carbon dioxide (CO2), and the solution changes from blue to green and then to yellow.
BTB reagent will form blue solution when encountering alkaline solution, and BTB reagent will form yellow solution when encountering acidic solution.
Bromothymol blue solution is an indicator. The lower concentration of CO2 makes it turn from blue to green, and the higher concentration of CO2 makes it turn from blue to green and then yellow. An experimental group used the experimental device shown in the figure to carry out the following operations at 25 ° C for many times:
1. Open valves a and B, push the piston of syringe 1, and then close a and B
2. After a period of time, the piston of the syringe 2 is pulled to promote the gas flow in the two bottles. The discoloration of bromothymol blue solution was observed. (when valves a and B are closed, the whole device is sealed)
Bromothymol blue powder (add 1g bromothymol blue to 1L of 20% alcohol solution) is an acid-base indicator with a discoloration range of pH6.0 (yellow) to 7.6 (blue). Ordinary water is neutral, the pH is about 7, and it is almost light blue. After dissolving carbon dioxide, carbonic acid will be formed, which is a weak acid, so the pH will not drop too much and turn yellow. The transition color is green. CO2 generation can be detected: the bromothymol blue aqueous solution changes from blue to green and then yellow. Compared with the detection method of clarified lime water, it has higher accuracy and no interference of SO2.
According to the color change, it can be used to detect CO2 released by aerobic and anaerobic respiration.

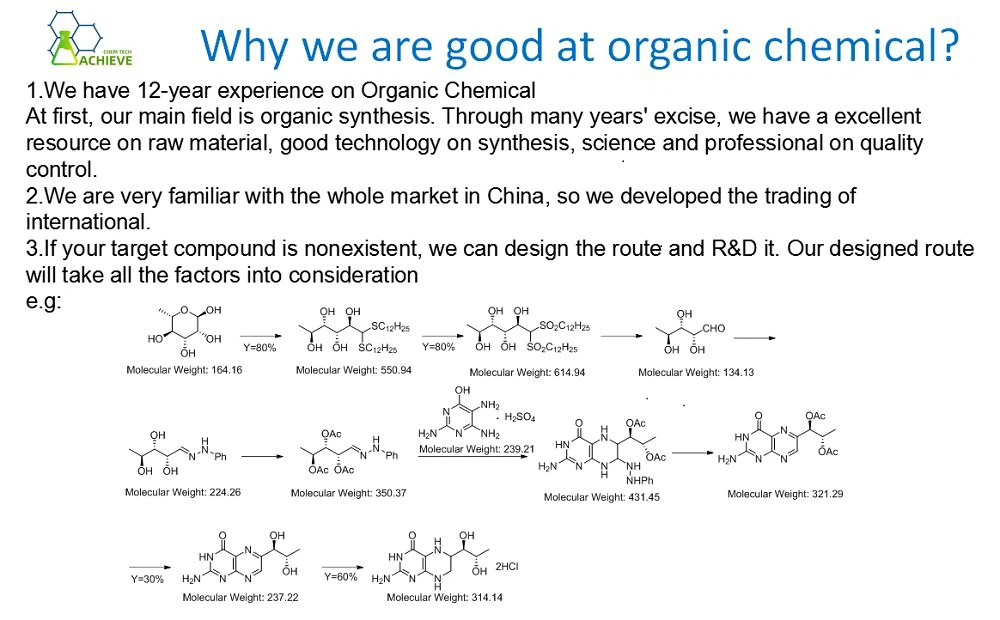
Synthesis method of bromothymol blue:
1. Under stirring, bromine is introduced into the suspension of thymol blue in glacial acetic acid, and the reaction is carried out at 50 ℃. Preparation of thymol blue: thymol is condensed with o-sulfobenzoic anhydride to form thymol blue.
2. Add the excess bromine glacial acetic acid solution slowly to the aqueous acetic acid solution of thymol blue under stirring, react at 40 ~ 50 ℃ for 4h, cool to below 20 ℃, filter and dry to obtain colorless bromine thymol blue.
After the seeds absorb water, the breathing intensity of living seeds increases rapidly. The released CO2 dissolves in water and becomes H2CO3, which further dissociates into H + and HCO3 -, thus increasing the acidity around the seed embryo. And the change of acidity can discolor the pH indicator BTB. The discoloration range of BTB is pH 6.0 ~ 7.6, and the acidity is yellow and the alkalinity is blue. Therefore, the carbon dioxide released by respiration can be tested to judge the dead or alive seeds.
Bromothymol blue is an acid-base indicator with a discoloration range of pH 6.0 (yellow) to 7.6 (blue).
Ordinary water is neutral, the pH is about 7, and it is almost light green.
After dissolving carbon dioxide, carbonic acid will be formed, which is a weak acid, so the pH will not drop too much and turn yellow.
The transition color is green or the generation of CO2 is detected: the clear lime water is turbid, and the bromothymol blue aqueous solution is changed from blue to green and then yellow.

Bromothymol blue powder (BTB), also known as Bromothymol Blue or Bromothymol Blue, is a common acid-base indicator with multiple uses.
(1) Determine the acidity and alkalinity of the solution
Color change range and characteristics: It is a pH indicator with a color change range of pH 6.0 (yellow) to 7.6 (blue). It appears yellow in acidic solutions, blue in alkaline solutions, and has precise color conversion near neutrality. Its pKa is about 7.1, the chemical formula is C27H28Br2O5S, and the acid-base equilibrium reaction formula is C27H28Br2O5S ⇌ H ⁺+C27H27Br2O5 ⁻.
Specific application scenarios
Chemical analysis: used as a standard indicator for titration analysis to help determine the acid-base neutralization point of a solution, accurately determine the reaction endpoint in chemical experiments, and improve the accuracy of experimental results.
Water quality testing: used to detect the acidity and alkalinity of water and evaluate the water quality condition. By observing the color change of bromothymol blue, one can quickly understand the acid-base environment of water bodies, providing important basis for water quality monitoring and protection.


Environmental monitoring: used to monitor the acidity and alkalinity of soil and water, and understand the changes in environmental acidity and alkalinity. In environmental science research, it helps to assess the health status of soil and water bodies, providing data support for environmental protection and governance.
(2) Biochemistry Experiment
Monitoring pH changes in biological reactions
Enzyme activity assay: Bromothymol blue has been used together with phenol red to monitor fungal asparaginase activity. When phenol red turns pink and bromothymol blue turns blue, it indicates an increase in pH and enzyme activity. However, a recent study suggests that due to the bright yellow ring form in the enzyme activity region, methyl red is more useful in determining activity.
Cell culture: Maintaining a suitable pH environment is crucial for cell growth and metabolism during the cell culture process. Bromothymol blue can be used as an acid-base indicator to monitor the pH changes of the culture medium in real time, helping researchers adjust the culture conditions in a timely manner and ensure the normal growth of cells.
Observing photosynthesis and respiration
Photosynthesis observation: Bromothymol blue powder can be used to observe photosynthesis. During photosynthesis, plants absorb carbon dioxide and release oxygen, causing a change in the acidity or alkalinity of the solution. By observing the color change of bromothymol blue, one can indirectly understand the intensity and efficiency of photosynthesis.
Respiratory indicator: In respiratory experiments, adding CO ₂ will cause the bromothymol blue solution to turn yellow. For example, by exhaling into its neutral solution through a tube, the color of the solution changes from green to yellow due to the absorption of CO ₂ from respiration into the solution to form carbonic acid. Therefore, it is commonly used in science courses to demonstrate that the more muscles are used, the greater the output of CO ₂.


(3) Teaching Experiment
Basic Chemistry Teaching: In basic chemistry teaching, bromothymol blue is often used to demonstrate acid-base reactions. By allowing students to observe the color changes of bromothymol blue in different acid-base solutions, it helps them intuitively understand the principles and processes of acid-base reactions, and enhances their learning interest and experimental skills.
Biochemistry teaching: In biochemistry teaching, bromothymol blue is used to help students understand pH changes in biological reactions. For example, when explaining cell respiration and photosynthesis, students were asked to observe the color change of bromothymol blue through experiments, deepening their understanding of the mechanism of acid-base balance regulation in living organisms.
(1) Obstetric testing
Detecting premature rupture of membranes: The pH of amniotic fluid is usually>7.2, so bromothymol blue will turn blue when it comes into contact with the leaked liquid from the amniotic membrane. Due to the acidic pH of the vagina, blue indicates the presence of amniotic fluid. But in the presence of other alkaline substances (such as blood or semen) or bacterial vaginosis, this test may show false positives.
Clinical significance: Premature rupture of membranes is one of the common complications in obstetrics, which may lead to adverse consequences such as premature birth and infection. The bromothymol blue detection method provides obstetricians with a simple and rapid diagnostic method, which helps to take timely treatment measures and ensure maternal and infant health.
(2) Assist in diagnosing diseases
Gastroscopy assistance: During gastroscopy, bromothymol blue can be used to observe the acid-base changes in the gastric mucosa and assist in the diagnosis of gastric diseases. The acid-base environment of the gastric mucosa is closely related to the occurrence and development of gastric diseases. By observing the color changes of bromothymol blue, doctors can understand the acid-base status of the gastric mucosa and provide reference for the diagnosis and treatment of diseases.


Efficacy evaluation: For patients receiving antacid therapy, the treatment effect can be evaluated by observing the color change of bromothymol blue. If the color change of bromothymol blue after treatment indicates an improvement in the gastric acid-base environment, it indicates that the treatment is effective; On the contrary, it may be necessary to adjust the treatment plan.
(3) Auxiliary testing items
Urine routine and other tests: Bromothymol blue can react with potassium bromide and ammonium bromide, resulting in more accurate results for urine routine, stool routine, liver function, blood routine, and other tests. During these checks, bromothymol blue indicator can help detect certain components in the sample, improving the sensitivity and accuracy of the detection.
Precautions for use: When conducting relevant examinations, it is necessary to follow the guidance of a doctor and avoid self operation to avoid affecting the accuracy of the examination results. At the same time, attention should be paid to the use and dosage of bromothymol blue, and relevant operating procedures should be followed.
(1) Components of bacterial culture medium
Count of Bacillus cereus: Bromothymol blue powder can be used to count Bacillus cereus in agar gel culture medium. Bacillus cereus is a common foodborne pathogen. By adding bromothymol blue to the culture medium, Bacillus cereus can be distinguished and counted based on the color changes around the colony.
Other bacterial detection substrates: Bromothymol blue can also be used as a bacterial growth component and detection substrate, such as cysteine/lactose/electrolyte deficient medium (CLED/BROLACIN), polymyxin pyruvate yolk mannitol Bromothymol blue medium (PEMBA), and MaConkey agar medium. These culture media utilize the acid-base indicator properties of bromothymol blue to help identify and isolate different types of bacteria.


(2) BTB paper method
Determination of whole blood cholinesterase activity: The BTB paper method can be used for emergency determination of whole blood cholinesterase activity and for the diagnosis of organophosphate poisoning. Organophosphorus pesticide poisoning can lead to a decrease in whole blood cholinesterase activity. The BTB paper method can quickly and easily detect whole blood cholinesterase activity, providing a basis for early diagnosis and treatment of organophosphate poisoning.
Operation method and advantages: The BTB paper method is easy to operate, does not require complex instruments and equipment, and is suitable for use in primary medical institutions and on-site first aid. This method has the advantages of speed, sensitivity, and accuracy, which can provide timely diagnostic information for doctors and improve the success rate of patient treatment.
Hot Tags: bromothymol blue powder cas 76-59-5, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, CHICAGO SKY BLUE 6B, Consumable, Iridium III chloride, 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID, Cerium sulfate powder








