Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of ninhydrin hydrate cas 485-47-2 in China. Welcome to wholesale bulk high quality ninhydrin hydrate cas 485-47-2 for sale here from our factory. Good service and reasonable price are available.
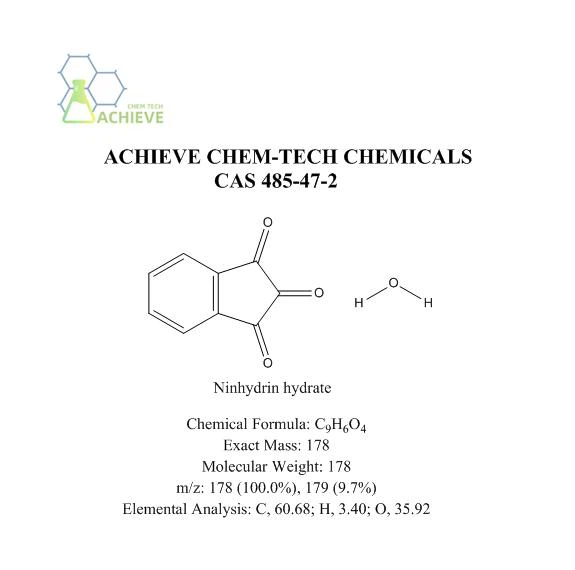
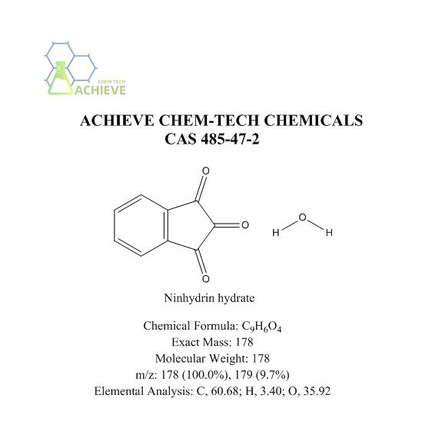
Ninhydrin hydrate is an organic compound with the molecular formula C9H6O4, CAS 485-47-2, and a molecular weight of 178.141. It is a nearly white crystalline or light yellow crystalline powder, slightly soluble in ether and chloroform, and turns red above 100 ℃. Easy to dissolve in water, soluble in ethanol, slightly soluble in ether and chloroform. Can be used as a colorimetric probe for quantitative analysis of amino acids and proteins. Can be used to monitor deprotection in solid-phase peptide synthesis (Kaiser test). The peptide chain is connected to the solid matrix through the C-terminus, and the N-terminus is used to expand the peptide chain. When the N-terminus is deprotected, the ninhydrin test shows a blue color. Amino acid residues are connected to the peptide chain while being protected at the N-terminus. Therefore, if the next amino acid residue is successfully connected to the peptide chain, the ninhydrin test will give a colorless or yellow result.

|
Chemical Formula |
C9H6O4 |
|
Exact Mass |
178 |
|
Molecular Weight |
178 |
|
m/z |
178 (100.0%), 179 (9.7%) |
|
Elemental Analysis |
C, 60.68; H, 3.40; O, 35.92 |
|
|
|

Ninhydrin hydrate, also known as dihydroindene-1,2,3-trione hydrate, with the molecular formula C9H6O4, is a white to pale yellow crystalline powder with a unique tricarbonyl structure. The highly electrophilic central carbon atom in its molecule makes it a core reagent for detecting amino containing compounds, widely used in fields such as biochemistry, forensic science, clinical diagnosis, agricultural science, and materials analysis.
The color reaction of indenone is based on its specific reaction with compounds containing alpha amino groups. Under weakly acidic conditions (pH 4-6), indanone undergoes a three-step reaction with alpha amino acids:
Condensation reaction: Dehydration condensation of α - amino group with indanone to form Schiff base;
Decarboxylation isomerization: Schiff base undergoes decarboxylation and isomerization to imine, releasing one molecule of CO ₂;
Oxidation reduction and color development: Imine further reacts with indene ketone to form a deep blue to purple Ruhemann purple complex, with a maximum absorption wavelength of 570 nm.
Special case: Proline and hydroxyproline have different reaction pathways due to the presence of pyrrolidine rings, resulting in the formation of yellow products; Aspartic acid reacts to produce a brown substance. This selectivity makes indene ketone an important tool for amino acid structure analysis.

Applications in Biochemistry and Clinical Diagnosis

1. Quantitative analysis of amino acids and proteins
Indene ketone is the core color reagent for amino acid analyzers. After chromatographic separation, indenone reacts with amino acids to form a colored complex, which is quantified by colorimetry at a wavelength of 570 nm using spectrophotometry. For example:
Food testing: Determine the lysine content in milk and evaluate the nutritional value of protein;
Clinical diagnosis: detection of abnormal amino acid metabolism in urine (such as phenylketonuria);
Drug development: Analyze the purity of peptide drugs to ensure batch consistency.
Technical advantages: High sensitivity (detection limit in the Dana molar range), wide linear range (0.1-100 μ mol/L), suitable for complex matrix analysis.
2. Solid phase peptide synthesis (SPPS) monitoring
In solid-phase peptide synthesis, indenone is monitored in real-time for deprotection steps through Kaiser testingDeprotection detection: When the N-terminal protecting group (such as Fmoc) of the peptide chain is removed, the reaction between the free amino group and indenone appears blue, indicating completion of the reaction;
Coupling efficiency evaluation: If the next amino acid is successfully coupled, the N-terminal amino group is protected, and the indanone test shows colorless or yellow, the synthesis conditions can be optimized accordingly.


Case: During the synthesis of angiotensin II (Asp Arg Val Tyr Ile His Pro Phe), Kaiser testing confirmed that the coupling efficiency of each step was greater than 99%, and the final product purity reached 98.5%.
3. Analysis of protein hydrolysis products
Indene ketone can detect free amino acids in protein hydrolysate and assist in the study of enzymatic hydrolysis mechanisms. For example:
Enzymatic hydrolysis of casein by gastric protease: Quantify the release of phenylalanine and leucine using the ninhydrin colorimetric method to evaluate enzyme activity;
Microbial fermentation monitoring: Analyze the amino acid profile in yeast fermentation broth and optimize the fermentation process.
1. Development of latent fingerprints
Indene ketone is the "gold standard" reagent for forensic fingerprint detection. The sweat in fingerprints contains amino acids (such as lysine and serine), which react with ninhydrin to form a purple complex. The imaging effect is better than traditional iodine fumigation.
Operation process:
Spray 0.2% ninhydrin ethanol solution onto the surface of the specimen;
Heating at 60 ℃ for 10 minutes promotes the reaction;
The purple fingerprint lines are clearly visible and can be stored for several months.
Case: In a burglary case in 2023, the police successfully solved the case by extracting the suspect's fingerprints from the door handle using ninhydrin imaging technology.


2. Identification of Schistosoma Egg Activity
Schistosoma eggs contain succinate dehydrogenase, which has high activity and can catalyze the reduction of indenone to produce purple products; Dead eggs have no enzymatic activity and appear yellow in color. This method is used to evaluate the therapeutic effect of schistosomiasis:
Before treatment: the color rendering rate of live eggs is greater than 80%;
After treatment, if the color rendering rate of live eggs is less than 10%, it indicates that the drug is effective.
1. Research on the Utilization of Soil Microbial Carbon Sources
Indene ketone can track the absorption of labeled substrates by microorganisms. For example:
Add ¹ ³ C labeled urea to the soil;
Extract microbial proteins and release carboxyl carbon with ninhydrin;
Quantify the proportion of urea carbon entering microbial biomass by measuring the ¹ ³ C/¹ ² C ratio using an isotope mass spectrometer.
Research significance: To reveal the carbon fixation mechanism of ammonia oxidizing bacteria (such as Nitrosomonas) and optimize nitrogen fertilizer application strategies.


2. Analysis of plant amino acid metabolism
Indene ketone combined with paper chromatography (PC) can quickly analyze the amino acid composition in plant tissues. For example:
Extracting free amino acids from rice leaves;
Spot on filter paper, using n-butanol acetic acid water (4:1:1) as the developing agent;
Spray ninhydrin solution, heat for color development, calculate Rf value, and identify the type of amino acid.
Application scenario: Screening stress resistant varieties (such as salt tolerant rice) and studying the role of amino acids in stress response.
1. Thin layer chromatography (TLC) color reagents
Indene ketone is a universal color reagent for TLC detection of amine compounds
Detection range: primary amines, secondary amines, amino acids, amino acid esters;
Operation points: After spraying the color developer, heat it at 105 ℃ for 5 minutes, and the purple spots will be clearly visible;
Sensitivity: Can detect nanogram level amine substances.
Case: In drug synthesis, the purity of intermediates is monitored using the ninhydrin TLC method to ensure reaction selectivity.
2. Determination of amino content on the surface of polymer materials
Indene ketone can quantitatively analyze the free amino groups on the surface of polymers such as chitosan and polylysine
Immerse the material in a solution of indene ketone, heat and react, and measure the absorbance;
Calculate the amino content through the standard curve and evaluate the material modification effect.
Research significance: To guide the functional design of biomedical materials, such as tissue engineering scaffolds.


A synthesis method of ninhydrin hydrate, the reaction route is as follows:
Step S1 specifically includes the following operations: dissolve the dihydroindene-1,3-diketone in ethanol, add sulfuric acid solution, drop sodium nitrite solution, control the reaction temperature at 15-20 ℃, test the reaction end point with potassium iodide starch test paper, and obtain fine powder 2-nitroso-1H-indene-1,3 (2H) - dione. The mass fraction of the sulfuric acid solution is 50-60%, and the mass fraction of the sodium nitrite solution is 25-30%. The reaction end point tested with potassium iodide starch test paper is that the potassium iodide starch test paper does not develop color.
Step S2 specifically includes the following operations: 2-nitroso-1H-indene-1,3 (2H) - dione is substituted with formaldehyde in the presence of hydrochloric acid to obtain indene-1,2,3 trione at 15-20 ℃. The mass fraction of hydrochloric acid is 25-30%, and the mass fraction of formaldehyde is 35-37%.
Step S3 specifically includes the following operations: adding nine-1,2,3 trione into the extraction solution, precipitating the precipitation, and drying the precipitation under reduced pressure at 50-60 ℃ to obtain reduced nine-1 trione; The extraction solution is a solution composed of low sodium sulfite and 35~37wt% industrial formaldehyde in a mass ratio of 1:1.
Step S4 specifically includes the following operations: oxidize the reduced ninhydrin in nitric acid solution at 60~80 ℃ to obtain crude ninhydrin, recrystallize the crude ninhydrin and water at a mass ratio of 1:5, dry the obtained solid with calcium chloride, vacuum dry it at 45~50 ℃ to obtain hydrated ninhydrin.

Characteristic reaction:
Ninhydrin is a reagent used to detect ammonia or primary and secondary amines. When reacting with these free amines, a dark blue or purple substance can be produced, which is called Ruhemann purple. It is often used to detect fingerprints. This is due to the lysine residues contained in the proteins and peptides shed from the fingerprint surface, and the primary amine on them is detected by ninhydrin. At room temperature, it is a white solid substance, soluble in ethanol and acetone. Ninhydrin can be regarded as the hydrate of dihydroinden-1,2,3-trinone.
Reactivity:
The carbon atom of the carbonyl group is partially positive charged. If the adjacent group connected to it has the ability to attract electrons (such as the carbonyl radical), the positive charge of the carbonyl carbon will be further strengthened. Therefore, the central carbon atom of 1,2,3-trichonyl compounds is more electrophilic than simple ketones. Therefore, indane-1,2,3-trione is easy to react with nucleophiles, such as water. However, for most carbonyl compounds, the form of carbonyl is more stable than that of water binding products. The reason why ninhydrin can form a stable hydrate of central carbon atom is due to the destabilization of its adjacent carbonyl group. It should be noted that in order to produce ninhydrin chromophore, amine needs to be condensed with one molecule of ninhydrin hydrate to produce Schiff base. Only ammonia and primary amine can go through this step. There must be a α Protons are used for Schiff base transfer, so if the carbon adjacent to the amine is a tertiary carbon atom, it cannot be detected by product. The reaction of ninhydrin with secondary amine will produce imine salt ([R1R2C=NR3R4]+), and the imine salt also has color, generally orange.
Hot Tags: ninhydrin hydrate cas 485-47-2, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, Iridium III chloride, 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID, CHICAGO SKY BLUE 6B, Cerium sulfate powder, Consumable