1,3,5-Tribromobenzene is an organic compound with the molecular formula C6H3Br3 and CAS 626-39-1. It is a light yellow brown powder with a certain pungent odor and may cause allergic reactions in some people. Insoluble in water, but soluble in some organic solvents such as hot ethanol and glacial acetic acid. As an important organic compound, it has broad application prospects in fields such as chemistry, medicine, pesticides, and materials science. With the continuous development of science and technology and the emergence of new materials, their application fields will also continue to expand. It should also be noted that its safety and environmental friendliness need to be strictly controlled during use to ensure human health and environmental safety.
(Product link: https://www.bloomtechz.com/chemical-reagent/laboratory-reagent/1-3-5-tribromobenzene-cas-626-39-1.html )

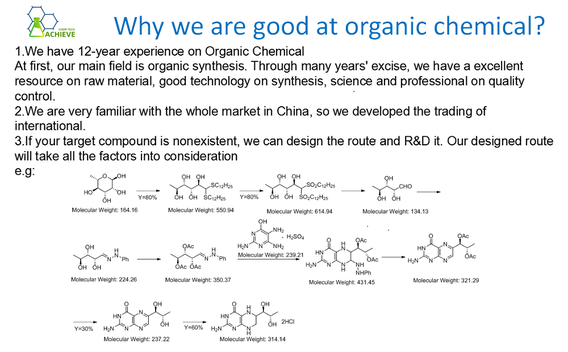
Tribromobenzene is an important organic compound widely used in fields such as chemistry, medicine, and materials science. There are various synthesis methods for Tribromobenzene, and the following is a detailed description of all synthesis methods for Tribromobenzene:
1. Direct bromination method
Direct bromination method is one of the most commonly used synthesis methods for Tribromobenzene. This method uses benzene as the raw material and reacts with bromine under the action of a catalyst to generate Tribromobenzene. The specific steps are as follows:
(1) Preparation of reactants
Add a certain amount of benzene and bromine to the reaction vessel.
Add an appropriate amount of catalyst, such as iron powder or aluminum powder.
(2) Reaction condition control
Heat the reaction kettle to the reaction temperature, generally controlled between 100 ℃ and 130 ℃.
Maintain a constant reaction temperature and continue stirring to thoroughly mix the reactants.
Monitor the reaction process and pay attention to the color and viscosity changes of the reaction solution.
(3) Reaction time control
The reaction time has a significant impact on the synthesis of Tribromobenzene and requires precise control.
Generally speaking, it is advisable to have a reaction time between 3 and 5 hours.
As the reaction time prolongs, the yield of Tribromobenzene gradually increases, but excessive reaction time may lead to the generation of by-products.
(4) Separation and purification
After the reaction is complete, cool the reaction solution to room temperature.
Remove catalysts and other solid impurities through filtration.
Separate the filtrate by distillation or crystallization to obtain high-purity Tribromobenzene.
(5) Chemical equations
In the direct bromination method, benzene undergoes a substitution reaction with bromine to form Tribromobenzene. The specific chemical equation is as follows:
C6H6+3Br2 → C6H3Br3+3HBr
This equation represents the substitution reaction between benzene and bromine in the direct bromination method, generating Tribromobenzene and hydrobromic acid. It should be noted that this equation is only a schematic representation, and the actual reaction process may involve other intermediate products and complex reaction mechanisms.
2, Iron bromide method
The iron bromide method is an improved synthesis method for Tribromobenzene, which can increase yield and purity. This method uses benzene and iron bromide as raw materials and reacts under inert gas protection to generate Tribromobenzene. The specific steps are as follows:
(1) Preparation of reactants
Add a certain amount of benzene and ferric bromide to the reaction vessel.
Add an appropriate amount of ferric bromide as a catalyst, with a general dosage of 10% to 20% by mass of benzene.
(2) Reaction condition control
Heat the reaction kettle to the reaction temperature, generally controlled between 80 ℃ and 100 ℃.
Maintain a constant reaction temperature and continue stirring to thoroughly mix the reactants.
Monitor the reaction process and pay attention to the color and viscosity changes of the reaction solution.
(3) Reaction time control
The reaction time has a significant impact on the synthesis of Tribromobenzene and requires precise control.
Generally speaking, it is advisable to have a reaction time between 2 and 4 hours.
As the reaction time prolongs, the yield of Tribromobenzene gradually increases, but excessive reaction time may lead to the generation of by-products.
(4) Separation and purification
After the reaction is complete, cool the reaction solution to room temperature.
Remove catalysts and other solid impurities through filtration.
Separate the filtrate by distillation or crystallization to obtain high-purity Tribromobenzene.
(5) Chemical equations
In the iron bromide method, benzene and bromine undergo a substitution reaction catalyzed by iron bromide to form Tribromobenzene. The specific chemical equation is as follows:
C6H6+Br2FeBr3 → C6H3Br3+FeBr2+HBr
This equation represents a substitution reaction between benzene and bromine catalyzed by iron bromide in the iron bromide method, generating Tribromobenzene, iron bromide, and hydrobromic acid. It should be noted that this equation is only a schematic representation, and the actual reaction process may involve other intermediate products and complex reaction mechanisms.
3. Photochemical synthesis method
Add benzene and bromine to the photoreactor, add an appropriate amount of photosensitizer (such as rose red), and introduce inert gas to remove air.
React under light conditions and maintain for a certain period of time to allow the reaction to proceed fully. Photochemical synthesis is a novel synthesis method that utilizes light energy to drive chemical reactions, with the advantages of mild reaction conditions and high selectivity. The following are the steps and corresponding chemical equations for synthesizing Tribromobenzene using photochemical synthesis method:
(1) Preparation of reactants
Add a certain amount of benzene and bromine to the photoreactor.
Add an appropriate amount of photosensitizer, such as rose red, etc.
Add an appropriate amount of catalyst, such as iron powder or aluminum powder.
(2) Control of photo reaction conditions
Heat the photoreactor to the reaction temperature, generally controlled between 80 ℃ and 100 ℃.
Continue stirring to thoroughly mix the reactants.
Turn on the light source and use ultraviolet or visible light to irradiate the reaction solution, driving the chemical reaction to proceed.
Monitor the reaction process and pay attention to the color and viscosity changes of the reaction solution.
(3) Reaction time control
The reaction time has a significant impact on the synthesis of Tribromobenzene and requires precise control.
Generally speaking, it is advisable to have a reaction time between 3 and 5 hours.
As the reaction time prolongs, the yield of Tribromobenzene gradually increases, but excessive reaction time may lead to the generation of by-products.
(4) Separation and purification
After the reaction is complete, cool the reaction solution to room temperature.
Remove catalysts and other solid impurities through filtration.
Separate the filtrate by distillation or crystallization to obtain high-purity Tribromobenzene.
(5) Chemical equations
In photochemical synthesis, benzene and bromine undergo a substitution reaction driven by light energy to form Tribromobenzene. The specific chemical equation is as follows:
C6H6+3Br2 (light) → C6H3Br3+3HBr
This equation indicates that in photochemical synthesis, benzene and bromine undergo a substitution reaction driven by light energy, generating Tribromobenzene and hydrobromic acid.
4. Other methods
In addition to the above three methods, there are also some other methods for synthesizing Tribromobenzene, such as the synthesis method using phenol as the raw material and the synthesis method using benzoic acid as the raw material. The specific steps and reaction conditions of these methods vary depending on the raw materials and processes. There are various synthesis methods for Tribromobenzene, and suitable methods can be selected for synthesis based on different raw materials and processes. In practical operation, it is necessary to pay attention to controlling the reaction conditions and implementing safety measures to ensure the smooth progress of synthesis.

