3-Nitrobenzaldehyde is an organic compound with the molecular formula C7H5NO3, CAS 99-61-6, and a molecular weight of 151.13. Usually yellow or brown crystalline or solid powder. Its color may vary depending on purity, batch, or storage conditions. It is soluble in water, but soluble in hot water. In organic solvents such as ether and chloroform, it also has good solubility. It is a weakly acidic compound with a pKa value of around 7. This means that it can exist stably under acidic and alkaline conditions, but tends to exhibit better stability under acidic conditions. It is a multifunctional organic intermediate that can react with many other compounds to synthesize other organic compounds. For example, it can react with alcohols to form ester compounds; React with aldehydes to form ketone compounds; React with amines to generate amide compounds, etc. These compounds have extensive application value in fields such as chemical engineering, medicine, and pesticides.
(Product link: https://www.bloomtechz.com/synthetic-chemical/organic-intermediates/3-nitrobenzaldehyde-99-cas-99-61-6.html )
|
|
|
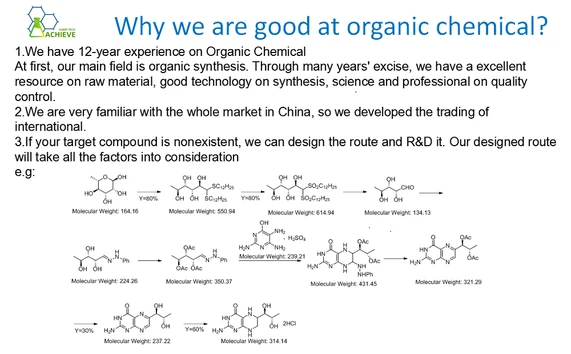
The synthetic resin method is a commonly used method for synthesizing 3-Nitrobenzaldehide.
(1) Condensation reaction: PhOH+HCHO → PhCHOPh+H2O
(2) Closed loop reaction: PhCHOPh+HNO3 → PhCHO+HNO2+H2O
Among them, Ph represents phenyl, HCHO represents formaldehyde, PhCHO represents benzaldehyde, HNO3 represents nitric acid, and HNO2 represents nitrite.
Reaction principle:
The basic principle of the synthetic resin method is to use phenol, formaldehyde, and nitric acid as raw materials, generate resin through condensation reaction, and then convert the resin into 3-Nitrobenzaldehide through closed-loop reaction. In this process, phenol and formaldehyde first undergo a condensation reaction to produce phenolic resin. Then, the resin undergoes a closed-loop reaction under the action of nitric acid, generating 3-Nitrobenzaldehide.
Experimental steps:
(1) Prepare reagents and instruments: phenol, formaldehyde, nitric acid, sodium hydroxide, distilled water, stirrer, thermometer, dropper, flask, etc.
(2) Add an appropriate amount of phenol and formaldehyde to the flask and stir evenly.
(3) Heat the mixture to a certain temperature (usually 80-100 ℃) and maintain it for a period of time (usually 1-2 hours) to allow the condensation reaction to proceed.
(4) After the condensation reaction is completed, add an appropriate amount of nitric acid to the flask, continue heating and stirring.
(5) Maintain the reaction temperature for a period of time (usually 1-2 hours) to allow the resin to undergo a closed-loop reaction under the action of nitric acid.
(6) After the reaction is completed, dilute the reaction solution with distilled water, and then adjust the pH value to neutral with sodium hydroxide.
(7) Separate the generated 3-Nitrobenzaldehide from the reaction solution through filtration.
(8) Washing and drying: Wash the filter cake with an appropriate amount of water, and then dry it with anhydrous sodium sulfate.
(9) Detection: Detect whether the product is 3-Nitrobenzaldehide through infrared spectroscopy, nuclear magnetic resonance, and other methods.
Reducing 3-nitrobenzoic acid to 3-Nitrobenzaldehide is a common organic chemical reaction.
(CH3CO) 2O+2NaOH → CH3COONa+NaOCH3
This reaction equation represents the reaction between acetic anhydride and sodium hydroxide to produce sodium acetate and methanol. This is a typical ester exchange reaction achieved by exchanging the positions of alcohol hydroxyl and carboxyl groups.
Reaction principle:
Firstly, we need to understand the structure and properties of 3-nitrobenzoic acid and 3-Nitrobenzaldehide. 3-nitrobenzoic acid is a carboxylic acid with one carboxyl group and one nitro group. And 3-Nitrobenzaldehide is an aldehyde with one aldehyde group and one nitro group.
To reduce 3-nitrobenzoic acid to 3-Nitrobenzaldehide, we need to use a reducing agent to reduce its carboxyl group to an aldehyde group. Common reducing agents include metal hydrides, alcohol hydrides, boranes, etc. Among them, alcohol hydrides (such as NaBH4) are a commonly used reducing agent that can reduce carboxyl groups to aldehyde groups.
The mechanism of the reduction reaction is that the alcohol hydride first adds to the carbonyl group of the carboxyl group to form an alcohol salt. Then, this alkoxide is further reduced to aldehydes. Meanwhile, nitro groups remain unchanged during the reduction process.
Experimental steps:
(1) Prepare reagents and instruments: 3-nitrobenzoic acid, NaBH4, ethanol, ice bath, stirrer, dropper, test tube, etc.
(2) Dissolve 3-nitrobenzoic acid in ethanol and add an appropriate amount of NaBH4.
(3) Stir the solution in an ice bath to allow the reduction reaction to take place for a period of time.
(4) Monitoring reaction progress: As the reaction progresses, the color of the solution will change gradually from yellow to colorless.
(5) After the reaction is completed, add an appropriate amount of water with a dropper to stop the reaction.
(6) Add an appropriate amount of dilute hydrochloric acid to the reaction solution through a dropper to achieve a neutral pH value.
(7) Separate the generated 3-Nitrobenzaldehide from the reaction solution through filtration.
(8) Washing and drying: Wash the filter cake with an appropriate amount of water, and then dry it with anhydrous sodium sulfate.
(9) Detection: Detect whether the product is 3-Nitrobenzaldehide through infrared spectroscopy, nuclear magnetic resonance, and other methods.
In addition to the various synthesis methods mentioned above, there are also several common methods for synthesizing 3-Nitrobenzaldehide:
Oxidation method: Use oxidants such as nitric acid, potassium permanganate, etc. to oxidize benzaldehyde to 3-Nitrobenzaldehide.
1. Phase transfer catalysis method: Under the action of a phase transfer catalyst, 3-Nitrobenzaldehide is synthesized from phenol, formaldehyde, and nitric acid as raw materials.
2. Electrochemical method: In an electrolytic cell, appropriate electrodes and electrolytes are used to oxidize benzaldehyde to 3-Nitrobenzaldehide through the electrolysis process.
3. Biocatalytic method: Using enzymes or microbial catalysts to convert benzaldehyde into 3-Nitrobenzaldehyde.
It should be noted that different synthesis methods are suitable for different conditions and requirements, and the selection of appropriate synthesis methods should be based on specific circumstances. Meanwhile, due to the complexity and danger of chemical reactions, it is essential to pay attention to safety and follow experimental norms and operating procedures when conducting chemical synthesis.



