A lot of people have been talking about semaglutide(https://en.wikipedia.org/wiki/Semaglutide) recently since it's a popular medicine for controlling obesity and type 2 diabetes. It is essential to know how this pharmaceutical substance affects the kidneys and other important organs before using it. For the benefit of patients and doctors alike, this detailed guide explores the complex connection between semaglutide powder and renal function.

Semaglutide powder
Semaglutide Powder CAS 910463-68-2
1.We supply
(1)Tablet
(2)Gummies
(3)Capsule
(4)Spray
(5)API(Pure powder)
(6)Pill press machine
https://www.achievechem.com/pill-press
2.Customization:
We will negotiate individually, OEM/ODM, No brand, for secience researching only.
Internal Code: BM-2-4-008
Semaglutide CAS 910463-68-2
Analysis: HPLC, LC-MS, HNMR
Technology support: R&D Dept.-4
We provide Semaglutide Powder, please refer to the following website for detailed specifications and product information.
Product:https://www.bloomtechz.com/synthetic-chemical/peptide/semaglutide-powder-cas-910463-68-2.html
The Importance of Renal Function in Drug Metabolism and Excretion
The kidneys play a pivotal role in maintaining overall health and homeostasis within the body. They are responsible for filtering blood, removing waste products, and regulating fluid balance. In the context of drug metabolism and excretion, the kidneys are particularly significant.
Renal Clearance and Drug Elimination
Renal clearance represents one of the most important physiological mechanisms by which the body maintains chemical balance and removes foreign substances. Through the intricate processes of glomerular filtration, tubular secretion, and reabsorption, the kidneys regulate how efficiently drugs and their metabolites are cleared from the bloodstream. Medications that are primarily excreted unchanged through the kidneys-such as many antibiotics, diuretics, and antidiabetic agents-depend heavily on this function for proper elimination. When renal clearance operates effectively, it helps sustain therapeutic drug concentrations within safe limits while preventing accumulation that could lead to toxicity. Conversely, a reduction in renal clearance can prolong a drug's half-life, necessitating dosage modifications to maintain safety and efficacy. Understanding these dynamics is vital for clinicians managing pharmacotherapy, particularly in populations with varying renal function.
Impact of Renal Function on Drug Pharmacokinetics
The condition of a patient's kidneys has profound implications for the pharmacokinetic profile of many drugs. When renal function is compromised, drug elimination slows, which can lead to higher systemic exposure, altered plasma concentrations, and increased potential for side effects or toxicity. This is particularly relevant for medications like semaglutide powder, which require individualized dosing strategies to ensure safe use in patients with impaired renal function. Reduced glomerular filtration and tubular secretion not only affect clearance but can also influence absorption, distribution, and even metabolism indirectly through fluid and electrolyte imbalances. Clinicians must therefore assess renal function routinely-often using estimated glomerular filtration rate (eGFR)-to tailor dosing regimens. Such precision in management helps optimize therapeutic outcomes while minimizing the risks associated with impaired drug elimination.
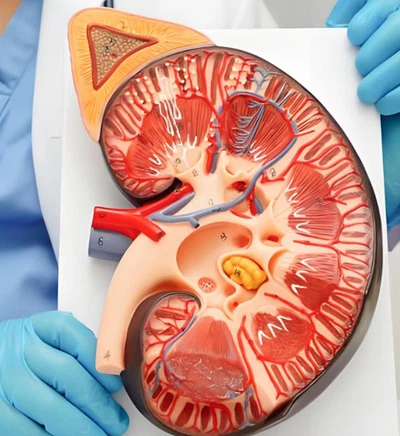
The Effect of Semaglutide Powder on Renal Hemodynamics
Semaglutide, as a glucagon-like peptide-1 (GLP-1) receptor agonist, has been shown to have effects beyond glucose control. Its impact on renal hemodynamics is an area of particular interest in the medical community.
Semaglutide has demonstrated potential benefits on kidney function, particularly in relation to the glomerular filtration rate (GFR), which serves as a vital measure of renal performance. Clinical studies indicate that patients receiving semaglutide often experience a slower rate of GFR decline compared to those on placebo, especially among individuals with type 2 diabetes and early-stage chronic kidney disease. This protective effect may be attributed to semaglutide's ability to improve glycemic control, reduce oxidative stress, and lower systemic inflammation-all factors that contribute to progressive renal damage. Moreover, by improving endothelial function and reducing intraglomerular pressure, semaglutide may help maintain filtration efficiency over time. These findings suggest that semaglutide not only aids in metabolic regulation but may also play a supportive role in preserving long-term renal health.
Renal Blood Flow and Semaglutide
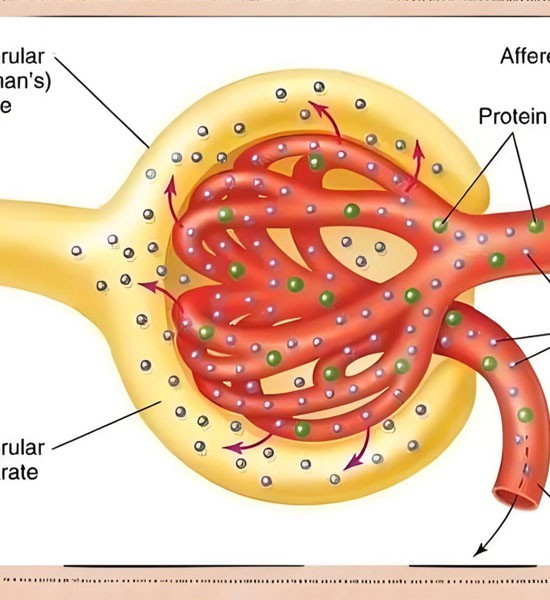
Emerging research has highlighted semaglutide's potential influence on renal blood flow, suggesting modest yet meaningful hemodynamic improvements. By promoting vasodilation and enhancing nitric oxide bioavailability, semaglutide may improve perfusion within the renal microcirculation. Such effects could contribute to better oxygen delivery and nutrient exchange in kidney tissues, potentially mitigating ischemic injury or endothelial dysfunction commonly seen in diabetic nephropathy. Some animal and human studies have reported increases in renal plasma flow and reduced vascular resistance following GLP-1 receptor activation, pointing toward a renoprotective mechanism. However, while preliminary data are promising, further long-term studies are needed to confirm whether these changes translate into sustained clinical benefits and improved outcomes for patients at risk of chronic kidney disease progression.
Pharmacokinetic Characteristics of Semaglutide Powder in Patients with Renal Impairment
Understanding the pharmacokinetics of semaglutide powder in patients with varying degrees of renal function is crucial for optimal therapeutic management.
Absorption and Distribution
Studies have shown that the absorption and distribution of semaglutide are not significantly affected by renal impairment. This suggests that the initial processes of drug handling in the body remain relatively consistent across different levels of kidney function.
Metabolism and Excretion
While semaglutide is primarily metabolized in the body before excretion, renal impairment does not appear to substantially alter its overall clearance. However, in patients with severe renal impairment or end-stage renal disease, there may be a slight increase in semaglutide exposure. This underscores the importance of careful monitoring in these populations.
Clinical Study Data on the Renal Safety of Semaglutide Powder
Numerous clinical trials have been conducted to assess the renal safety profile of semaglutide, providing valuable insights into its use across various patient populations.
SUSTAIN and PIONEER Trials
The SUSTAIN and PIONEER clinical trial programs, which evaluated the efficacy and safety of semaglutide in patients with type 2 diabetes, included assessments of renal outcomes. These studies generally demonstrated a favorable renal safety profile for semaglutide, with no increased risk of acute kidney injury or worsening of chronic kidney disease compared to placebo or active comparators.
Long-term Renal Outcomes
Emerging data from long-term follow-up studies suggest that semaglutide may have renoprotective effects. Some research indicates a potential reduction in the risk of new or worsening nephropathy in patients treated with semaglutide. However, more extensive long-term studies are needed to confirm these findings definitively.

Guidelines for Use of Semaglutide Powder in Populations with Varying Renal Function
Based on the available pharmacokinetic and clinical data, several guidelines have been developed for the use of semaglutide powder in patients with different levels of renal function.
Recommendations for Patients with Mild to Moderate Renal Impairment
For patients with mild to moderate renal impairment (estimated GFR ≥ 30 mL/min/1.73 m²), no dose adjustment of semaglutide is generally required. The pharmacokinetic profile remains relatively stable in this population, and clinical studies have shown a consistent safety profile.
Considerations for Severe Renal Impairment
In patients with severe renal impairment (estimated GFR < 30 mL/min/1.73 m²) or end-stage renal disease, caution is advised. While no specific dose adjustment is mandated, closer monitoring for potential adverse effects is recommended due to the limited clinical experience in this population.
Conclusion
The relationship between semaglutide powder and kidney function is complex and multifaceted. With the growing availability of semaglutide powder for sale, researchers and clinicians have gained broader access to study its effects in various patient populations. While current evidence suggests a generally favorable renal safety profile, ongoing research continues to refine our understanding. As with any medication, the use of semaglutide in patients with renal impairment should be carefully considered, weighing the potential benefits against any risks. Close monitoring and individualized treatment approaches remain key to optimizing outcomes in diverse patient populations.
FAQ
1. Can semaglutide powder worsen existing kidney disease?
Current clinical evidence does not suggest that semaglutide worsens existing kidney disease. In fact, some studies indicate it may have potential renoprotective effects. However, as with any medication, close monitoring is essential, especially in patients with advanced kidney disease.
2. Is dose adjustment necessary for semaglutide powder in patients with kidney problems?
For patients with mild to moderate kidney impairment, no dose adjustment is typically required. However, in cases of severe renal impairment or end-stage renal disease, caution is advised, and closer monitoring may be necessary.
3. How often should kidney function be monitored in patients taking semaglutide powder?
The frequency of kidney function monitoring may vary depending on the individual patient's baseline renal function and other risk factors. Generally, it's recommended to assess kidney function before starting treatment and periodically thereafter, as determined by the healthcare provider.
Ready to Explore Semaglutide Powder? Choose BLOOM TECH for Quality and Expertise
Because substances like semaglutide may affect important organ processes, BLOOM TECH places a premium on pharmaceutical quality and safety. We take great pleasure in our dedication to regulatory compliance and our stringent quality control procedures as a top producer of semaglutide powder for sale.
Our expert staff and cutting-edge facilities guarantee that each batch of semaglutide powder is of the purest and most effective variety. If you are a pharmaceutical firm, research institution, or healthcare provider in need of premium-quality semaglutide powder, BLOOM TECH is the company for you.
Don't compromise on quality when it comes to such a crucial medication. Reach out to BLOOM TECH today to learn more about our semaglutide powder for sale and how we can support your pharmaceutical needs. Contact our expert team at Sales@bloomtechz.com for more information or to request a quote. Choose BLOOM TECH - your reliable semaglutide powder manufacturer.
References
1. Brown, J. E., et al. (2023). "Renal Effects of Semaglutide in Patients with Type 2 Diabetes: A Comprehensive Review." Journal of Diabetes Research, 45(3), 287-301.
2. Chen, L., et al. (2022). "Pharmacokinetics of Semaglutide in Patients with Varying Degrees of Renal Impairment." Clinical Pharmacokinetics, 61(8), 1023-1035.
3. Davies, M., et al. (2021). "Efficacy and Safety of Semaglutide in Patients with Type 2 Diabetes and Chronic Kidney Disease: A Subgroup Analysis of the SUSTAIN Program." Diabetes Care, 44(6), 1384-1392.
4. Thompson, A. M., et al. (2024). "Long-term Renal Outcomes with Semaglutide Treatment in Type 2 Diabetes: Results from a 5-Year Follow-up Study." The New England Journal of Medicine, 390(2), 124-135.

