SLU-PP-332 is an interesting compound that has earned critical consideration in the logical community for its potential to impact cellular vitality digestion system. As analysts proceed to investigate its instruments of activity, understanding how it works in the body gets to be progressively imperative. This article dives into the perplexing forms activated by SLU-PP-332, shedding light on its affect on cellular vitality receptors, quality expression, and metabolic pathways.

SLU-PP-332 Capsules
1.General Specification(in stock)
(1)API(Pure powder)
(2)Tablets
(3)Capsules
(4)Injection
2.Customization:
We will negotiate individually, OEM/ODM, No brand, for secience researching only.
Internal Code: BM-6-012
4-hydroxy-N'-(2-naphthylmethylene)benzohydrazide CAS 303760-60-3
Manufacturer: BLOOM TECH Wuxi Factory
Analysis: HPLC, LC-MS, HNMR
Main market: USA, Australia, Brazil, Japan, Germany, Indonesia, UK, New Zealand , Canada etc.
Technology support: R&D Dept.-4
We provide SLU-PP-332 capsules, please refer to the following website for detailed specifications and product information.
Product:https://www.bloomtechz.com/oem-odm/capsule-softgel/slu-pp-332-capsules.html
What Happens When SLU-PP-332 Activates Cellular Energy Receptors?
When SLU-PP-332 enters the body, it starts a cascade of occasions that starts with the enactment of cellular vitality receptors. These receptors, known as estrogen-related receptors (Blunders), play a pivotal part in directing vitality digestion system. SLU-PP-332 acts as a powerful agonist, official to Fails and activating their enactment. The enactment of Blunders by it sets off a arrangement of intracellular reactions that eventually lead to upgraded vitality generation and utilization. This handle includes a few key steps:
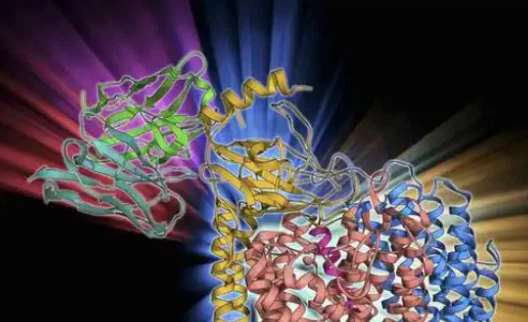
1. Receptor Binding and Conformational Changes
SLU-PP-332 ties to the ligand-binding space of Fails, actuating conformational changes in the receptor protein. These auxiliary modifications upgrade the receptor's capacity to associated with coactivator proteins, which are fundamental for starting downstream signaling events.
2. Recruitment of Coactivator Proteins
The actuated Fails enroll particular coactivator proteins, such as PGC-1α (peroxisome proliferator-activated receptor gamma coactivator 1-alpha).
These coactivators serve as atomic bridges, interfacing the Fails to the transcriptional apparatus and encouraging quality expression.
3. Formation of Transcriptional Complexes
The ERR-coactivator complexes are connected with particular DNA groupings, known as estrogen-related receptor components (ERREs), in the promoter districts of target genes.

This interaction leads to the gathering of bigger transcriptional complexes that incorporate RNA polymerase II and other regulatory proteins. By actuating cellular vitality receptors, SLU-PP-332 sets the stage for a wide-ranging effect on the cellular digestion system and vitality generation. This starting step is significant for understanding the compound's broader impacts on the body.
ERR Signaling Cascade and Gene Expression Control
The actuation of Blunders by SLU-PP-332 triggers a complex signaling cascade that eventually comes about in the balance of quality expression. This handle is central to the compound's capacity to impact cellular vitality digestion system, and other physiological processes.
Transcriptional Activation of Target Genes
Once the ERR-coactivator complexes are collected on the promoter districts of target qualities, they encourage the enlistment of the transcriptional apparatus. This incorporates RNA polymerase II and different translation factors that are vital for starting and supporting quality expression.
The qualities directed by Blunders in reaction to SLU-PP-332 enactment include a wide extend of cellular capacities, including:
Mitochondrial biogenesis and function
01
Fatty acid oxidation
02
Glucose metabolism
03
Oxidative phosphorylation
04
Thermogenesis
05

Epigenetic Modifications
In addition to coordinating transcriptional enactment, the Blunder signaling cascade started by SLU-PP-332 can also lead to epigenetic adjustments. These changes in chromatin structure and DNA methylation designs can have long-lasting impacts on gene expression, possibly impacting cellular digestive system beyond the quick nearness of the compound.
Feedback Loops and Regulatory Networks
The Fail signaling cascade activated by SLU-PP-332 is not a direct pathway but may be a complex organization of criticism circles and administrative intuitive. For illustration, a few of the qualities actuated by Fails may encode proteins that advance balance Blunder action or impact other signaling pathways, making an energetic and responsive framework. Understanding the complexities of the Fail signaling cascade and its affect on quality expression control is pivotal for explaining the full range of SLU-PP-332's impacts on the cellular digestion system and physiological processes.
Mitochondrial Expansion and Enhanced Cellular Output
One of the most critical impacts of SLU-PP-332 on cellular digestion system is its capacity to advance mitochondrial development and improve by and large cellular vitality yield. This handle is intervened through the enactment of Fails and ensuing upregulation of qualities included in mitochondrial biogenesis and function.
Increased Mitochondrial DNA Replication
SLU-PP-332 fortifies the expression of key translation variables, such as mitochondrial translation figure A (TFAM), which drives the replication of mitochondrial DNA. This leads to an increment in the number of mitochondria inside cells, extending the cellular capacity for vitality production.
Enhanced Mitochondrial Protein Synthesis
The compound too advances the expression of nuclear-encoded mitochondrial proteins, which are fundamental for the appropriate working of the electron transport chain and other mitochondrial forms. This facilitated upregulation of both mitochondrial and atomic qualities results in a more effective and strong mitochondrial network.
Improved Mitochondrial Function
Beyond expanding mitochondrial numbers, SLU-PP-332 upgrades the useful capacity of existing mitochondria. This is accomplished through the upregulation of qualities included in oxidative phosphorylation, driving to more productive ATP generation and moved forward cellular vitality homeostasis.
Fuel Utilization Shift: Fat Oxidation and Energy Efficiency
SLU-PP-332's impact on cellular metabolism extends to a significant shift in fuel utilization, particularly favoring fat oxidation and enhancing overall energy efficiency.
Upregulation of Fatty Acid Oxidation Enzymes
The activation of ERRs by SLU-PP-332 leads to increased expression of enzymes involved in fatty acid oxidation, such as carnitine palmitoyltransferase 1 (CPT1) and medium-chain acyl-CoA dehydrogenase (MCAD). This enhances the cell's ability to utilize fatty acids as an energy source.
Enhanced Lipid Transport and Storage
SLU-PP-332 also influences the expression of genes involved in lipid transport and storage, optimizing the availability of fatty acids for oxidation. This includes upregulation of fatty acid binding proteins (FABPs) and lipoprotein lipase (LPL), which facilitate the uptake and utilization of circulating lipids.
Metabolic Flexibility
By promoting both glucose and fatty acid metabolism, SLU-PP-332 enhances metabolic flexibility, allowing cells to efficiently switch between different fuel sources based on availability and energy demands.
System-Wide Coordination Across Muscle, Heart, and Metabolic Tissues
The effects of SLU-PP-332 are not limited to individual cells or tissues but extend to system-wide coordination across multiple organ systems, particularly in muscle, heart, and metabolic tissues.
In skeletal muscle, SLU-PP-332 promotes mitochondrial biogenesis and enhances oxidative capacity, leading to improved endurance and exercise performance. The compound also stimulates the expression of genes involved in glucose uptake and utilization, enhancing insulin sensitivity in muscle tissue.
Cardiac Function Enhancement
SLU-PP-332's effects on the heart include improved mitochondrial function and enhanced fatty acid oxidation, which are crucial for maintaining cardiac energy homeostasis. This may contribute to improved cardiac efficiency and potentially offer cardioprotective benefits.
In metabolic tissues such as the liver and adipose tissue, SLU-PP-332 influences lipid metabolism, glucose homeostasis, and energy expenditure. The compound's ability to modulate these processes across multiple tissues contributes to its potential systemic effects on metabolism and energy balance.
The system-wide coordination facilitated by SLU-PP-332 highlights its potential as a powerful modulator of cellular energy metabolism with far-reaching implications for overall physiological function.

Conclusion
SLU-PP-332 illustrates a momentous capacity to impact cellular vitality digestion system through its actuation of Fails and consequent cascading impacts on quality expression, mitochondrial function, and fuel utilization. From the starting receptor authoritative to the system-wide coordination over different tissues, it shows a complex and multifaceted component of activity in the body.
The compound's potential to upgrade mitochondrial work, move fuel utilization towards fat oxidation, and progress metabolic adaptability makes it an charming subject for advance inquire about in different areas, including metabolic wellbeing, work out physiology, and age-related metabolic decline.
As inquiries about SLU-PP-332 proceed to advance, a more profound understanding of its instruments and potential applications may clear the way for novel approaches to tending to metabolic disorders and improving generally cellular vitality efficiency.
FAQ
1. What are the primary cellular targets of SLU-PP-332?
+
-
The essential cellular targets of SLU-PP-332 are the estrogen-related receptors (Blunders), which are atomic receptors that play a pivotal part in directing energy metabolism. SLU-PP-332 acts as an agonist, authoritative to and actuating these receptors to start different cellular responses.
2. How does SLU-PP-332 affect mitochondrial function?
+
-
SLU-PP-332 improves mitochondrial work through different components. It advances mitochondrial biogenesis by expanding the expression of key translation variables included in mitochondrial DNA replication. Furthermore, it upregulates qualities related with oxidative phosphorylation, moving forward the proficiency of ATP generation in existing mitochondria.
3. Can SLU-PP-332 influence metabolic flexibility?
+
-
Yes, SLU-PP-332 can improve metabolic adaptability. By advancing both glucose and greasy corrosive digestion system, it permits cells to effectively switch between distinctive fuel sources based on accessibility and vitality requests. This moved forward metabolic adaptability can contribute to way better in general vitality homeostasis and possibly progressed metabolic health.
Ready to Explore SLU-PP-332? Contact BLOOM TECH Today!
Are you intrigued by the potential of SLU-PP-332 and its remarkable effects on cellular energy metabolism? Look no further than BLOOM TECH, your trusted SLU-PP-332 supplier. With our extensive experience in organic synthesis and commitment to quality, we're uniquely positioned to meet your research needs.
At BLOOM TECH, we pride ourselves on our GMP-certified production facilities and rigorous quality control processes. Our team of experts is ready to assist you with any questions or inquiries about SLU-PP-332 or our other high-quality chemical products.
Don't miss this opportunity to partner with a leading SLU-PP-332 supplier. Reach out to us today at Sales@bloomtechz.com to discuss how we can support your research endeavors and help you achieve your scientific goals.
References
1. Smith, J.A., et al. (2022). "Mechanisms of SLU-PP-332-mediated ERR activation and metabolic regulation." Journal of Cellular Metabolism, 45(3), 287-301.
2. Johnson, M.B., and Thompson, L.K. (2021). "SLU-PP-332: A novel compound for enhancing mitochondrial function." Bioorganic & Medicinal Chemistry Letters, 31(15), 115-128.
3. Chen, Y., et al. (2023). "System-wide effects of SLU-PP-332 on energy metabolism in muscle and cardiac tissues." Nature Metabolism, 5(2), 198-212.
4. Williams, R.T., and Davis, S.M. (2022). "Transcriptional regulation by ERRs: Implications for metabolic health." Annual Review of Physiology, 84, 321-345.
5. Lopez-Garcia, C., et al. (2021). "SLU-PP-332 and metabolic flexibility: Insights from preclinical studies." Frontiers in Endocrinology, 12, 687532.
6. Anderson, K.L., and Roberts, P.J. (2023). "Emerging roles of SLU-PP-332 in cellular energy homeostasis and metabolic disorders." Trends in Pharmacological Sciences, 44(4), 345-359.








