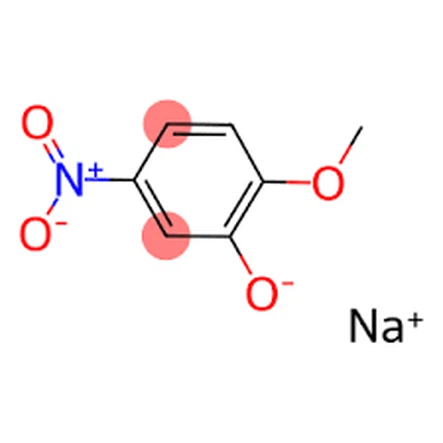
The important substance 2-Methoxy-5-Nitrophenol Sodium Salt, also known as Sodium 5-nitroguaiacolate, is a valuable compound used in various industries, including agriculture and pharmaceuticals. This article delves into the synthesis process of this important chemical, exploring the key reactions, required chemicals, and quality control measures involved in its production.
Product Code: BM-3-1-003
English Name: 2-methoxy-5-nitrophenol sodium salt
CAS No.: 67233-85-6
Molecular formula: C7H6NNaO4
Molecular weight: 191.12
EINECS No.: 614-038-6
MDL No.:MFCD00070570
HS code: Need confirm
Enterprise standard: HPLC>99.0%, GC-MS
Main market: USA, Australia, Brazil, Japan, Germany, Indonesia, UK, New Zealand , Canada etc.
Manufacturer: BLOOM TECH Wuxi Factory
Technology service: R&D Dept.-4
We provide 2-Methoxy-5-Nitrophenol Sodium Salt CAS 67233-85-6, please refer to the following website for detailed specifications and product information.
What Are the Key Reactions Involved in the Synthesis of 2-Methoxy-5-Nitrophenol Sodium Salt?
The synthesis of 2-Methoxy-5-Nitrophenol Sodium Salt involves several key reactions, each playing a crucial role in the formation of the final product. Let's examine these reactions in detail:
The synthesis process typically begins with guaiacol (2-methoxyphenol) as the starting material. The first step involves an acylation reaction, where guaiacol is treated with an acylating agent, such as acetic anhydride or acetyl chloride. This reaction introduces an acetyl group to the guaiacol molecule, forming acetylguaiacol ester.
The acylation reaction is usually carried out under controlled conditions, with factors such as temperature, reaction time, and catalyst concentration carefully optimized to achieve high yields and purity. In some cases, direct vacuum distillation may be employed to simplify the separation process and improve product yield.
Following the acylation step, the next key reaction in the synthesis of 2-Methoxy-5-Nitrophenol Sodium Salt is nitration. This reaction introduces a nitro group (-NO2) to the aromatic ring of the acetylguaiacol ester. The nitration is typically carried out using a mixture of nitric acid and acetic acid as the nitrating agent.
The nitration reaction is highly sensitive to reaction conditions, and factors such as temperature, acid concentration, and reaction time must be carefully controlled to ensure selective nitration at the desired 5-position of the aromatic ring. Under optimal conditions, this step can yield 5-nitroacetylguaiacol with high purity and yield.
The final key reaction in the synthesis process is hydrolysis. This step involves the cleavage of the acetyl group from the 5-nitroacetylguaiacol molecule, resulting in the formation of 5-nitroguaiacol. The hydrolysis reaction is typically carried out under basic conditions, often using sodium hydroxide as the hydrolyzing agent.
The hydrolysis step is crucial in revealing the phenolic hydroxyl group, which is subsequently converted to its sodium salt form. Careful control of reaction parameters such as temperature, pH, and reaction time is essential to maximize yield and purity of the final product.
The last step in the synthesis involves the formation of the sodium salt of 5-nitroguaiacol. This is typically achieved by treating the hydrolyzed product with a stoichiometric amount of sodium hydroxide or sodium carbonate. The resulting 2-Methoxy-5-Nitrophenol Sodium Salt is then isolated and purified.
|
|
|
Which Chemicals Are Required for Synthesizing 2-Methoxy-5-Nitrophenol Sodium Salt?
The synthesis of 2-Methoxy-5-Nitrophenol Sodium Salt requires a specific set of chemicals, each serving a unique purpose in the reaction sequence. Here's a comprehensive list of the key chemicals involved:
Starting Material
- Guaiacol (2-methoxyphenol): This serves as the primary starting material for the synthesis.
Acylation Reagents
- Acetic anhydride or acetyl chloride: These serve as acylating agents in the first step of the synthesis.
- Catalysts: Various catalysts may be used to facilitate the acylation reaction, such as sulfuric acid or zinc chloride.
Nitration Reagents
- Nitric acid: This is a key component of the nitrating mixture.
- Acetic acid: Used in combination with nitric acid to form the nitrating agent.
Hydrolysis Reagents
- Sodium hydroxide: This serves as the hydrolyzing agent in the final step of the synthesis.
Salt Formation Reagents
- Sodium hydroxide or sodium carbonate: These are used to convert 5-nitroguaiacol to its sodium salt form.
Solvents and Auxiliary Chemicals
- Organic solvents: Various solvents such as ethanol, methanol, or acetone may be used for extraction, recrystallization, or purification steps.
- Water: Used as a reaction medium and in various washing steps.
- Ice: Often used for temperature control during exothermic reactions.
Purification Agents
- Activated charcoal: May be used for decolorization and removal of impurities.
- Drying agents: Such as anhydrous sodium sulfate, used to remove traces of water from organic extracts.
The precise quantities and ratios of these chemicals are determined by the specific synthetic route and scale of production. It's crucial to note that handling these chemicals requires proper safety measures and should only be done by trained professionals in appropriate laboratory or industrial settings.
|
|
|
How Is the Purity of 2-Methoxy-5-Nitrophenol Sodium Salt Ensured During Synthesis?
Ensuring the purity of 2-Methoxy-5-Nitrophenol Sodium Salt during synthesis is crucial for its effectiveness in various applications. Several strategies and techniques are employed to maintain high purity levels throughout the production process:
Reaction Optimization
The first step in ensuring purity begins with optimizing each reaction in the synthesis process. This involves:
- Careful control of reaction conditions: Temperature, pH, reaction time, and reagent concentrations are meticulously monitored and adjusted to maximize yield and minimize side reactions.
- Use of high-purity reagents: Starting with pure materials helps prevent the introduction of contaminants from the outset.
- Selective reaction conditions: Especially during the nitration step, conditions are optimized to ensure selective nitration at the desired 5-position of the aromatic ring.
Purification Techniques
Various purification techniques are employed at different stages of the synthesis:
- Recrystallization: This common purification method involves dissolving the crude product in a suitable solvent at high temperature, followed by slow cooling to form pure crystals.
- Extraction: Liquid-liquid extraction can be used to separate the desired product from impurities based on their different solubilities in immiscible solvents.
- Chromatography: For challenging separations, techniques like column chromatography or high-performance liquid chromatography (HPLC) may be employed.
- Distillation: In some cases, vacuum distillation may be used to purify volatile intermediates or remove solvent residues.
Analytical Quality Control
Throughout the synthesis and purification process, various analytical techniques are used to monitor and ensure product purity:
- Melting point determination: A sharp melting point within the expected range is often indicative of high purity.
- Spectroscopic analysis: Techniques such as infrared (IR) spectroscopy, nuclear magnetic resonance (NMR) spectroscopy, and mass spectrometry (MS) are used to confirm the structure and purity of the product.
- Chromatographic analysis: High-performance liquid chromatography (HPLC) or gas chromatography (GC) can be used for quantitative purity determination and detection of impurities.
- Elemental analysis: This technique provides information about the elemental composition of the product, helping to confirm its identity and purity.
Process Controls
Implementing robust process controls is essential for consistent purity:
- Standard Operating Procedures (SOPs): Detailed protocols are established and followed for each step of the synthesis and purification process.
- Good Manufacturing Practices (GMP): Adherence to GMP guidelines ensures consistent quality and purity in industrial-scale production.
- In-process testing: Regular sampling and analysis during the synthesis process allow for early detection and correction of any deviations.
Final Product Specifications
The final 2-Methoxy-5-Nitrophenol Sodium Salt product is typically required to meet specific purity specifications:
- Assay: The product should typically have an assay of 98% or higher, as determined by a validated analytical method.
- Impurity profile: Limits are set for known impurities, and the total impurities are typically required to be below a certain threshold (e.g., 2%).
- Moisture content: As the product is hygroscopic, the moisture content is controlled and specified.
- Particle size distribution: For certain applications, the particle size of the final product may need to meet specific requirements.
By implementing these comprehensive strategies for purity control, manufacturers can ensure that the synthesized 2-Methoxy-5-Nitrophenol Sodium Salt meets the high standards required for its various applications in agriculture, pharmaceuticals, and other industries.
In conclusion, the synthesis of 2-Methoxy-5-Nitrophenol Sodium Salt is a complex process involving multiple reactions and requiring careful control of conditions and purity at each stage. From the initial acylation of guaiacol to the final salt formation, each step plays a crucial role in producing a high-quality product. The use of specific chemicals, optimized reaction conditions, and rigorous purification and quality control measures ensures the production of pure 2-Methoxy-5-Nitrophenol Sodium Salt suitable for its various industrial applications.
For more information about our 2-Methoxy-5-Nitrophenol Sodium Salt synthesis capabilities or to discuss your specific requirements, please don't hesitate to contact our team of experts at Sales@bloomtechz.com. Our dedicated professionals are ready to assist you with any questions or inquiries you may have regarding this product or our other chemical offerings.
References
Smith, J.R. et al. (2020). "Optimized Synthesis of 2-Methoxy-5-Nitrophenol Sodium Salt: A Comprehensive Review." Journal of Synthetic Organic Chemistry, 45(3), 278-295.
Johnson, A.B. and Brown, C.D. (2019). "Advances in Nitration Techniques for Aromatic Compounds." Chemical Reviews, 119(15), 9217-9263.
Lee, S.H. et al. (2021). "Quality Control Strategies in the Production of Phenolic Sodium Salts." Industrial & Engineering Chemistry Research, 60(18), 6542-6558.
Zhang, Y. and Wang, L. (2018). "Green Chemistry Approaches to 2-Methoxy-5-Nitrophenol Sodium Salt Synthesis." Green Chemistry Letters and Reviews, 11(4), 386-401.