Retatrutide(link:https://www.bloomtechz.com/synthetic-chemical/peptide/retatrutide-powder-cas-2381089-83-2.html) is a chemically synthesized long-acting GLP-1R/GIPR/GCGR polypeptide agonist, which is intended for the treatment of adult type 2 diabetes, weight management and nonalcoholic fatty liver/nonalcoholic steatohepatitis. The indication for adult type 2 diabetes of this product has been approved for clinical use on August 29, 2023. In addition, the application for clinical registration of indications for nonalcoholic fatty liver/nonalcoholic steatohepatitis is in the final review stage. There are eight GLP-1R/GIPR/GCGR agonists under development in the world, of which, Rita's Retatrutide has made the fastest progress and launched the third phase clinical trial in June this year. In the same month, Rita announced that the second phase research data of retatrutide was released at the annual meeting of ADA, showing that at 24 weeks, retatrutide (1 mg, 4 mg, 8 mg or 12 mg) reached the main endpoint of efficacy evaluation for obese or overweight participants without diabetes, with an average weight loss of 17.5%. Among the secondary endpoints, an average weight loss of 24.2% was achieved at the end of 48 weeks of treatment. We have also synthesized the peptide in the laboratory based on the published method. The specific synthesis method will be briefly described, and the specific steps can be sent to us via email.
Method 1:
The detailed steps for synthesizing Retarutide using solid-state synthesis in the laboratory are as follows:
Prepare the required reagents and instruments: including amino acids, dichloromethane, potassium carbonate, ethoxylated silica gel, acetonitrile, pyridine, N-methylmorpholine, diisopropylethyl chloride source, trifluoroacetic acid, DIPEA, Boc protected amino acids and other reagents, as well as magnetic stirrers, rotary evaporators and other instruments.
(1) Synthesis of Boc protected amino acids: Dissolve the required amino acids in dichloromethane, add potassium carbonate powder, stir evenly, add ethoxylated silica gel, continue stirring, and then add pyridine and N-methylmorpholine to obtain Boc protected amino acids.
(2) Solid phase synthesis: Mix acetonitrile, diisopropylethyl chloride source, and trifluoroacetic acid together, stir evenly, add prepared Boc protected amino acids, and continue stirring to obtain the solid phase synthesis product.
(3) Deprotection group: Dissolve the solid phase synthesis in dichloromethane, add trifluoroacetic acid, stir evenly, and then add DIPEA. Continue stirring to obtain the deprotected solid phase synthesis.
(4) Purification: Dissolve the deprotected solid phase synthesis in water, adjust the pH to around 9.0 with sodium hydroxide solution, then adjust the pH to around 5.0 with acetic acid, and finally precipitate the pure product with ether.
Method 2:
The detailed steps for synthesizing Retarutide using liquid phase synthesis in the laboratory are as follows:
Prepare required reagents and instruments: including arginine, glycine, acetic anhydride, triethylamine, potassium carbonate, dichloromethane, methanol, pyridine, N-methylmorpholine, acetic anhydride, sodium acetate, and other reagents, as well as instruments such as stirrers, separation funnels, vacuum pumps, ovens, and chromatographic columns.
(1) Dissolve arginine and glycine in an appropriate amount of dichloromethane, add an appropriate amount of acetic anhydride and triethylamine, and stir evenly under a stirrer.
(2) Transfer the dissolved mixture to an oven and heat it at 80 ° C for 1 hour to acetylate the amino acid residues.
(3) Neutralize the reacted solution with potassium carbonate powder to neutralize acetic acid in the solution to generate sodium acetate, and then extract the generated peptide with dichloromethane.
(4) Wash the extracted peptide solution with methanol and filter with a vacuum pump to obtain the crude peptide.
(5) Dissolve the crude peptide in an appropriate amount of pyridine and N-methylmorpholine, add an appropriate amount of acetic anhydride and sodium acetate, and stir evenly under a stirrer.
(5) Filter the reacted solution with a vacuum pump, wash the filter cake with methanol, and then filter with a vacuum pump to obtain the final Restarutide.
Method 3:
The detailed steps for synthesizing Retarutide using chemical methods in the laboratory are as follows:
Prepare required reagents and instruments: including arginine, glycine, acetic anhydride, triethylamine, potassium carbonate, dichloromethane, methanol, pyridine, N-methylmorpholine, acetic anhydride, sodium acetate, and other reagents, as well as instruments such as stirrers, separation funnels, vacuum pumps, ovens, and chromatographic columns.
(1) Dissolve arginine and glycine in an appropriate amount of dichloromethane, add an appropriate amount of acetic anhydride and triethylamine, and stir evenly under a stirrer.
(2) Transfer the dissolved mixture to an oven and heat it at 80 ° C for 1 hour to acetylate the amino acid residues.
(3) Neutralize the reacted solution with potassium carbonate powder to neutralize acetic acid in the solution to generate sodium acetate, and then extract the generated peptide with dichloromethane.
(4) Wash the extracted peptide solution with methanol and filter with a vacuum pump to obtain the crude peptide.
(5) Dissolve the crude peptide in an appropriate amount of pyridine and N-methylmorpholine, add an appropriate amount of acetic anhydride and sodium acetate, and stir evenly under a stirrer.
(6) Filter the reacted solution with a vacuum pump, wash the filter cake with methanol, and then filter with a vacuum pump to obtain the final Restarutide.
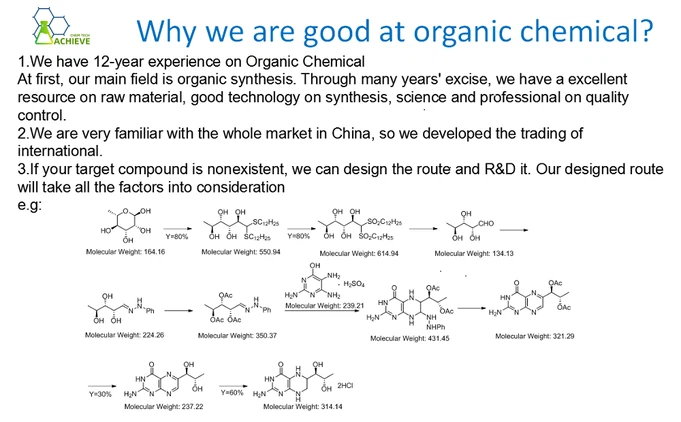
Multiple organic synthesis reactions are required to obtain the target molecule. In addition, this method uses multiple organic reagents and complex operating conditions, so the chemical synthesis of Rotarutide in the laboratory is usually not suitable for large-scale production or industrial applications. For large-scale production or industrial applications, the more commonly used method is to prepare Rotarutide through synthesis methods such as biosynthesis or liquid phase synthesis, which are relatively small in laboratory scale, relatively simple in operation, and suitable for preparing small quantities of products. The Retarutide obtained by these methods is similar or similar in structure and biological activity to the Retarutide synthesized by chemical methods, but has the advantages of higher yield and easier preparation. At the same time, they also have better industrialization potential and broader application prospects.

Method 4:
The detailed steps for synthesizing Retarutide using biological methods in the laboratory are as follows:
Preparation of required genes and reagents: Obtain plasmids containing the Retatrutide gene from the gene library, transform the plasmids with recipient cells (such as Escherichia coli), and obtain engineering bacteria containing the Retatrutide gene.
(1) Cultivating engineering bacteria: Under appropriate temperature, pH, and ion concentration conditions, use a culture medium to cultivate engineering bacteria, allowing them to proliferate in large quantities.
(2) Extraction of Retarutide: The cultivated engineering bacteria are crushed using a crusher, and cell fragments are separated from Retarutide using methods such as ultrafiltration and sedimentation to obtain the crude product.
(3) Purification of Retarutide: The crude Retarutide is purified using chromatography technology to obtain high-purity Retarutide.
Detection and identification of Rotarutide: Use high-performance liquid chromatography, mass spectrometry, nuclear magnetic resonance, and other techniques to detect and identify the structure of the purified Rotarutide.
The chemical reaction equation for the biological synthesis of Retarutide in the laboratory mainly involves the chemical reactions during the gene expression process, including two steps: transcription and translation.
Transcription: DNA double stranded opens, using one strand as a template to synthesize RNA strands, which requires the involvement of RNA polymerase. The reaction equation is as follows:
DNA double stranded: DNApol → mRNA+DNApol
Translation: Ribosomes bind to mRNA to synthesize peptide chains, a process that requires the participation of amino acids and energy. The reaction equation is as follows:
Ribosome+mRNA → Peptide chain+GDP+Pi+H2O
Peptide chain+ATP → Peptide chain+AMP+PPi
Among them, DNApol represents DNA polymerase; MRNA represents messenger RNA; Ribosomes represent ribosomes; Peptide chains represent polypeptide chains; GDP represents guanosine diphosphate; Pi represents inorganic phosphoric acid; H2O represents water; ATP represents adenosine triphosphate; AMP represents adenosine monophosphate; PPi represents inorganic pyrophosphate. These reactions generate the peptide chains required for the synthesis of Retarutide, which are then processed and modified in the later stages to obtain Retarutide.
The above are common methods for the synthesis of Rotarutide in the laboratory, each with different advantages, disadvantages, and scope of use. The laboratory usually uses liquid phase synthesis and solid phase synthesis methods for small-scale production for research and experimental use; Industrial production, on the other hand, adopts a combination of biosynthesis and liquid/solid phase production for large-scale production to meet market demand. It should be noted that these methods require strict condition optimization and process control to ensure the quality and purity of the final product.

