Sodium carbonate is a white odorless powder or particle under normal temperature. It has water absorption and gradually absorbs 1mol/l of water (about =15%) in the exposed air. Its hydrates include Na2CO3 · H2O, Na2CO3 · 7H2O and Na2CO3 · 10h2o. Sodium carbonate is easily soluble in water and glycerol. At 20 ℃, 20g of sodium carbonate can be dissolved in every 100g of water, and the maximum solubility is at 35.4 ℃. 49.7g of sodium carbonate can be dissolved in 100g of water, which is slightly soluble in absolute ethanol but hardly soluble in propanol.
His chemical properties are as follows:
The aqueous solution of sodium carbonate is alkaline and corrosive to a certain extent. It can reacts with acid and some calcium and barium salts. The solution is alkaline and can turn phenolphthalein red.
(1) Stability - strong stability, but it can also decompose under high temperature to form sodium oxide and carbon dioxide:
![]()
Long term exposure to the air can absorb moisture and carbon dioxide in the air, generate sodium bicarbonate and form hard blocks:
![]()
The crystalline hydrate of sodium carbonate (Na2CO3 · 10h2o) is easily weathered in dry air:
![]()
(2) Thermodynamic function - thermodynamic function at (298.15K, 100k):
Status: solid state
Standard molar formation enthalpy: -1130.8 kJ · mol-1
Standard molar Gibbs free energy of formation: -1048.1 kJ · mol-1
Standard entropy: 138.8 J · mol-1 · K-1
(3) Hydrolysis reaction - as sodium carbonate is hydrolyzed in aqueous solution, the ionized carbonate ions are combined with hydrogen ions in water to form bicarbonate ions, resulting in the reduction of hydrogen ions in the solution and the remaining ionized hydroxide ions, so the pH of the solution is alkaline.
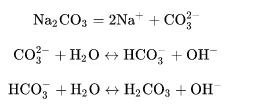
Because carbonate can combine with protons (i.e. hydrogen ions) in water to form bicarbonate and carbonic acid, and can combine with protons in acid to release carbon dioxide. Therefore, sodium carbonate belongs to Bronsted base in acid-base proton theory.
(4) Reaction with acid - Take hydrochloric acid as an example. When hydrochloric acid is sufficient, sodium chloride and carbonic acid are generated, and the unstable carbonic acid is immediately decomposed into carbon dioxide and water. This reaction can be used to prepare carbon dioxide:
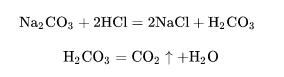
The general chemical equation is:
![]()
When hydrochloric acid is small, the following reactions occur:
![]()
Sodium carbonate can react similarly with other kinds of acids.
(5) Reaction with alkali - sodium carbonate can undergo double decomposition reaction with alkali such as calcium hydroxide and barium hydroxide to generate precipitation and sodium hydroxide. This reaction is commonly used in industry to prepare caustic soda (commonly known as causticization):
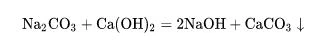
(6) Reaction with salt
Sodium carbonate can undergo double decomposition reaction with calcium salt and barium salt to form precipitation and new sodium salt:
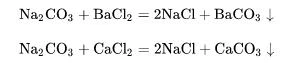
As sodium carbonate is hydrolyzed in water to generate sodium hydroxide and carbonic acid, its reaction with some salts will push the chemical balance to move in a positive direction to generate corresponding alkali and carbon dioxide:
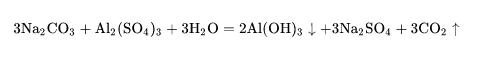
To sum up, it has many chemical properties, which also determines the wide range of its uses. Sodium carbonate is one of the important chemical raw materials. It is widely used in light industry, daily chemical industry, building materials, chemical industry, food industry, metallurgy, textile, petroleum, national defense, medicine and other fields. It is also used as raw material, cleaning agent and detergent for manufacturing other chemicals, as well as photography and analysis. Followed by metallurgy, textile, petroleum, national defense, medicine and other industries. The glass industry is the largest consumer of soda ash, with 0.2T of soda ash consumed per ton of glass. It is mainly used for float glass, kinescope glass shell, optical glass, etc. Among industrial soda ash, light industry, building materials industry and chemical industry account for about 2/3, followed by metallurgy, textile, petroleum, national defense, medicine and other industries.. Used in chemical industry, metallurgy, etc. The use of heavy soda ash can reduce the flying of alkali dust, reduce the consumption of raw materials, improve the working conditions, improve the product quality, reduce the erosion of alkali powder on refractories, and prolong the service life of kilns. As a buffer, neutralizer and dough improver, it can be used in cakes and flour foods, and can be used in an appropriate amount according to production needs.
The development of sodium carbonate is also very much, mainly referring to the development of soda making method from natural alkali: ① as early as 1849, pioneers found sodium bicarbonate in the sweet water river in Wyoming, USA, and used it for washing and pharmacy. In 1905, the first trial production of soda ash was carried out by using the natural soda of Lake Sears, California. In 1938, when the US Intermountain fuel supply company explored for oil and gas in the Green River Basin, Wyoming, it discovered the world's largest natural alkali deposit rich in sodium carbonate. In 1976, the soda ash produced by natural alkali in the United States accounted for 70% of the total output, and in 1982 it accounted for 94% of the total output, with an annual production capacity of 9.5mt. ② Since the 1960s, the Soviet Union has processed alumina with nepheline (natural alkali stone containing sodium, potassium, aluminum and silicon oxides) and simultaneously produced soda ash, potash and cement, realizing industrialization, so that nepheline raw materials can be comprehensively utilized without waste discharge. By 1975, five nepheline processing plants had been established.

