Sodium carbonate, also known as soda ash, is classified as salt, not alkali. It is also called soda or soda ash in international trade. It is an important inorganic chemical raw material, mainly used in the production of flat glass, glass products and ceramic glaze. It is also widely used in domestic washing, acid neutralization and food processing.
The development history of sodium carbonate should start from the first soda industry. The soda ash industry began at the end of the 18th century. With the needs of industry and the change of raw materials for Soda Making, the production technology of soda ash (Na2CO3) has developed rapidly, and the production equipment tends to be large-scale, mechanized and automated. In 1983, the world output of soda ash was about 30mt. In the history of soda ash industry, French n. lublanc, Belgian E. Solvay and Chinese houdebang have made outstanding contributions.
Before the artificial synthesis of soda ash, it was found in ancient times that the ash burned by some seaweeds after drying contained alkali. After soaking and filtering with hot water, brown alkali solution could be obtained for washing. A large amount of natural alkali comes from minerals, mainly buried underground or alkaline water lake. The natural alkali ore in the sedimentary layer has the highest grade and is widely distributed. At the end of the 18th century, the method of artificial synthesis of soda ash was first invented in France. Lublanc used mirabilite, limestone and coal to reduce and carbonate at high temperature to obtain a crude product black ash mainly containing Na2CO3. After leaching, evaporation, refining, recrystallization and drying, a heavy soda ash with a purity of about 97% was obtained. In 1861, ernestolvy of Belgium invented soda ash alone and obtained a patent. Since the protection of technical secrets has not been widely applied, it made a breakthrough from the United States in the 1920s. In particular, houdebang, a famous Chinese chemical expert, published the book "soda ash manufacturing" in 1932, which will be kept secret for 70 years. Solvay method was published in the world. Houdebang also established hous alkali making process from 1939 to 1942, and established a pilot plant in Sichuan. In 1952, a joint alkali making workshop was set up in Dalian Chemical Plant. The Na method introduced by Asahi nitrate subsidiary in Japan is essentially a compromise method of bicarbonate alkali and ammonia alkali. The ratio of soda ash to ammonium chloride can be adjusted at will.
In 1783, the French Academy of Sciences offered a reward of 1200 francs for the method of manufacturing soda ash. In 1789, Lubran, an attendant doctor of the feudal lord Duke of Orleans, France, successfully created a method for making soda. In 1791, he obtained a patent and established an soda plant with a daily output of 250~300kg. In addition to table salt, the raw materials used in the Lubran soda process include concentrated sulfuric acid, charcoal and limestone. The production process is as follows:
① Step 1: use concentrated sulfuric acid to convert table salt into sodium sulfate:

② Step 2: heat sodium sulfate, charcoal and limestone together in the furnace. Sodium sulfate reacts with charcoal in the furnace to form sodium sulfide and carbon monoxide:
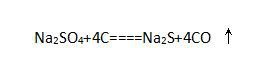
③ Step 3: sodium sulfide reacts with limestone to produce sodium carbonate and calcium sulfide:

The Lubran alkali production method has created a historical precedent and made outstanding contributions to mankind, but it also has many shortcomings. For example, the main production process is carried out in the solid phase, it is difficult to continuously produce with concentrated sulfuric acid as raw material, the equipment is severely corroded, the product quality is impure, the calcium sulfide is not easily soluble in water, the precipitated slag is discarded, the raw materials are not fully utilized, the cost is high, and HCI, CO and other gases are generated, resulting in environmental pollution. Troubled by these shortcomings, in 1861, the Belgian Solvay made sodium carbonate and sodium carbonate from salt, limestone and ammonia Calcium chloride, this is ammonia alkali method. The reaction steps are as follows:

CO2 and NH3 generated from the reaction can be reused as raw materials.
The total reaction formula is CaC03+2NaCl ===NaCl2+Na2CO3
Although this soda making method is more simple and environmentally friendly than the Lubran soda making method, realizes continuous production, greatly improves the utilization rate of salt, and has low cost, it has low utilization rate of raw materials and produces a large number of by-products with low use value The shortcomings of CaCl2 still haunt people, and foreign countries have very strict control over this patent. China has been restricted for a long time due to the lack of relevant technology.
Finally, houdebang invented the combined Soda Making Method in 1943, also known as houdebang soda making method, which is used to make soda ash in industry. It broke the technical blockade of foreign countries at that time, and further improved the efficiency of soda making. It has become a widely used soda making method in the world. The specific process is as follows: add CO2 into saturated ammonia salt water (ammonia and sodium chloride are saturated solutions) to produce the following reaction
The reaction chemical equation shall be:

Sodium bicarbonate in the reaction is precipitated due to its low solubility, which can be further calcined and decomposed into sodium carbonate, water and carbon dioxide, of which carbon dioxide can enter the reaction again for reuse. In view of the low utilization rate of table salt, the high cost of Soda Making, the environmental pollution caused by waste liquid and residue and the difficulty in treatment, Mr. houdebang successfully developed the combined Soda Making Method in 1943 after thousands of tests. This new process is to build an ammonia plant and an alkali plant at -- for joint production. Ammonia plant provides ammonia and carbon dioxide required by alkali plant. Ammonium chloride in mother liquor is crystallized by adding table salt as a chemical product or fertilizer. Salt solution can be recycled. The "combination" in the so-called "combined alkali making method" means that the method combines the synthetic ammonia industry and the alkali making industry together, makes use of the by-product CO2 during ammonia production, eliminates the use of limestone decomposition for production, and simplifies the production equipment. In addition, the combined caustic soda process also avoids the production of calcium chloride, which is not very useful by-product in the ammonia caustic soda process. Instead, ammonium chloride, which can be used as fertilizer, is used to recover it, which improves the utilization rate of salt, shortens the production process, reduces the pollution to the environment, reduces the cost of soda ash, and promotes the progress of industry in the world.
The outstanding feature of Hough's Soda making process is to make the process continuous, so as to expand the scale; Secondly, this method does not start with solid ammonium bicarbonate, but uses salt brine to absorb ammonia first and then carbonate for continuous production. Because this method does not need intermediate salt as auxiliary agent, the cost can be reduced. In 1952, China established a 10t / D pilot plant for combined alkali production in Dalian Chemical Plant, which was improved in 1957. The process flow of primary salt addition, secondary ammonia absorption and primary carbonation was determined through experiments, and the equipment selection and operation indexes were affirmed. In 1964, the large-scale combined soda plant was completed and put into operation in Dalian Chemical Industry Company.
Due to the high price of imported salt, Japan must seek new methods to improve the utilization rate of salt. In 1950, the subsidiary of Asahi nitrate established a 30t / D combined soda plant in Mushan chemical plant. In March 1959, it began to establish a new combined soda plant in Chiba chemical plant, with a daily output of 300t soda ash and ammonium chloride, named as A.C. method. In the 1970s, the production of ammonium chloride in Japan was oversupplied. In addition to resuming some ammonia alkali production, Asahi saltpetre established the new Asahi process, also known as the N.A. process. The characteristic of Xinxu process is that the output of ammonium chloride can be adjusted. The excess ammonium chloride in the market can be directly distilled with lime milk to recover ammonia. Therefore, the consumption of steam and lime is less than that of ammonia alkali process. The amount of waste liquid is reduced to nearly 1/3 of that of ammonia alkali process. The concentration of calcium chloride in waste liquid can be increased by 2.5 times, and the utilization rate of raw salt can reach more than 95%. Xinxu process has also made great improvements in carbonization, crystallization and other processes and equipment structure.
So far, the development of sodium carbonate has taken shape in general. Later, people made some minor improvements in processing according to different emphasis on use, so as to provide it for use in different occasions.

