As a common chemical substance, ibuprofen(link:https://www.bloomtechz.com/synthetic-chemical/api-researching-only/ibuprofen-powder-cas-15687-27-1.html) has a wide range of uses and many synthetic methods. There are six chemical synthesis methods: transposition rearrangement method, alcohol carbonylation method, olefin carbonylation method, halogenated hydrocarbon carbonylation method, olefin catalytic hydrogenation method, and propylene oxide rearrangement method. The details are respectively described below.
1. Transposition rearrangement method
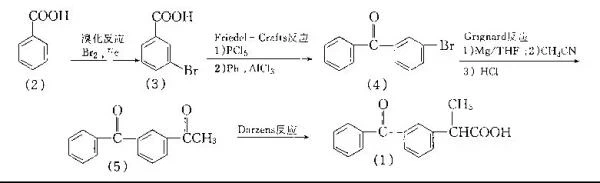
The aryl 1,2-transposition rearrangement method is a synthetic method commonly used by domestic manufacturers. It uses isobutylbenzene as raw material,
Ibuprofen can be prepared by Friedel-Crafts acylation with 2-chloropropionyl chloride, catalytic ketalization with neopentyl glycol, catalytic rearrangement, and hydrolysis.
The detailed steps of transposition rearrangement method:
Step 1: Preparation of α,α-dibromopropionic acid (1,2-dibromo-1,2-diphenylethane)
a. Dissolve benzene in dry carbon tetrachloride and add excess bromine.
Chemical equation: C6H6 + Br2 → C6H5Br + HBr
b. Slowly add the prepared bromobenzene solution into propylene dropwise, and react under the catalysis of ultraviolet light or phenol peroxide.
Chemical equation: C6H5Br + C3H4 → C6H5CHBrCH3
Step 2: Preparation of 2-hydroxy-2-acetophenone (2-hydroxy-2-phenylacetophenone)
a. Dissolve terephthalenone and benzene in n-butanol solvent, then add sodium.
Chemical equation: C6H5COC6H5 + Na → C6H5CO2Na + C6H6
b. Reaction under reflux conditions to obtain 2-hydroxyl-2-acetophenone.
Chemical equation: C6H5CO2Na + H2O → C6H5COCH2OH + NaOH
Step 3: Preparation of Ibuprofen
a. Dissolve the 2-hydroxyl-2-acetophenone obtained in step 2 in n-heptane.
b. Add acid catalyst (such as boric acid) and hydrogen peroxide to carry out Haber rearrangement reaction.
Chemical equation: C6H5COCH2OH + H2O2 → C6H5CH(CO2H)CH3 + H2O
c. Further processing of the resulting product, usually including neutralization, crystallization, filtration and drying steps.
d. Finally get Ibuprofen.
2. Alcohol carbonization method
The alcohol carbonylation method is the BHC method, using isobutylbenzene as a raw material, through Friedel-Crafts acylation with acetyl chloride, catalytic hydrogenation reduction and
Catalyzed carbonylation three-step reaction to prepare ibuprofen.
Detailed steps of alcohol carbonylation method:
Step 1: Preparation of sodium bisulfite (Sodium bisulfite)
Sulfuric acid is reacted with a higher purity alkali metal sulfite such as sodium sulfite.
Chemical equation: H2SO4 + Na2SO3 → NaHSO3 + NaHSO4
Step 2: Preparation of α-Bromoisobutyryl bromide
a. Dissolve Acetyl bromide in dry carbon tetrachloride.
b. Slowly add sodium bisulfate solution dropwise, while cooling the reaction system with ice water.
c. Obtain isoamyl α-bromopropionate by filtration and drying.
Chemical equation: CH3COBr + NaHSO3 → CH2BrCOC(CH3)3 + NaBr + H2SO4
Step 3: Synthesis of Ibuprofen
a. React isoamyl α-bromopropionate, benzyl alcohol and a suitable base, and carry out nuclear carbonylation reaction under an inert atmosphere.
Chemical equation: CH2BrCOC(CH3)3 + C6H5OH + NaOH → C6H5CH(OH)COCH(CH3)2 + NaBr
b. Under the catalysis of acid or base, Ibuprofen can be obtained through condensation and decomposition of alcohol.
c. After corresponding neutralization, crystallization, filtration and drying steps, high-purity Ibuprofen is obtained.
3. Olefin carbonylation method
Aryl substituted alkenes react with CO and water or alcohols to generate aralkyl antelines or antelopes under palladium catalysts and acidic conditions. The catalytic activity of palladium under anaerobic conditions can be enhanced by combining with certain ligands.
Overview steps of olefin carbonylation method:
Step 1: Preparation of α, β-unsaturated ketone (α, β-Unsaturated ketone)
Synthesis of acetone (Acetone) and styrene (Styrene) reaction, usually under the action of acidic catalysts such as sulfuric acid and hydrochloric acid.
Chemical equation: C6H5CH=CH2 + CH3COCH3 → C6H5CH2COCCH3
Step 2: Synthesis of Ibuprofen
a. React α, β-unsaturated ketones with hydroxylamine (Hydroxylamine) under alkaline conditions to form α, β-saturated ketones (α, β-Saturated ketone).
Chemical equation: C6H5CH2COCCH3 + NH2OH → C6H5CH2CONHOH + CH3COCCH3
b. Carry out aldol condensation reaction in the presence of a catalyst to generate Ibuprofen from α, β-saturated ketone and propionic acid.
Chemical equation: C6H5CH2CONHOH + CH3CH2COOH → C13H18O2 + H2O
4. Halogenated hydrocarbon carbonylation method
The halogenated hydrocarbon carbonylation method uses 1-p-isobutylphenyl-1-chloroethane as a raw material, and carbonylates it with CO under a catalyst and alkaline conditions to form a product.
Overview steps of halogenated hydrocarbon carbonylation method:
Step 1: Preparation of Haloalkane
Propionic acid reacts with bromite (such as sodium bromite) under acidic conditions to generate the corresponding halogenated hydrocarbons.
Chemical equation: CH3CH2COOH + NaBr + H2SO4 → CH3CH2Br + NaHSO4 + H2O
Step 2: Synthesis of Ibuprofen
a. The halogenated hydrocarbon is reacted with sodium isobutyrate (Sodium isobutyrate), and a carbonylation reaction occurs to form the precursor molecule of Ibuprofen.
Chemical equation: CH3CH2Br + CH3COONa → CH3CH(COONa)CH3 + NaBr
b. Under alkaline conditions, the precursor molecule of Ibuprofen is hydrolyzed to obtain Ibuprofen.
Chemical equation: CH3CH(COONa)CH3 + H2O → C13H18O2 + CH3COONa
The halogenated hydrocarbon carbonylation method of Ibuprofen involves the preparation of halogenated hydrocarbons from propionic acid and bromite, and then reacts the halogenated hydrocarbons with sodium isovalerate for carbonylation to form the precursor molecule of Ibuprofen. Finally, the precursor molecules of Ibuprofen are hydrolyzed under alkaline conditions to obtain Ibuprofen. This method gradually builds the structure of the target molecule through multi-step reactions, and uses catalysts such as acids and bases to promote the reaction. During the synthesis process, it is necessary to pay attention to controlling the reaction conditions, selecting suitable solvents and catalysts, and performing purification and purification steps to obtain high-purity Ibuprofen. Carbonylation of halogenated hydrocarbons is a commonly used synthetic method, which can be used to prepare important organic compounds such as Ibuprofen.
|
|
|
5. Catalytic Hydrogenation of Olefins
The hydrogenation of 2-(6-methoxy-2-tetrayl)acrylic acid was catalyzed by the nail complex of chiral ligands to prepare Cai Pusheng, and the enantiomeric excess (ee) reached 96%.
Overview steps of catalytic hydrogenation of olefins:
Step 1: Preparation of Olefin Precursor
Synthesis Phenethylamine reacts with formic acid to produce olefin precursors.
Chemical equation: C6H5CH2NH2 + HCOOH → C6H5CH=CH2 + H2O + CO2
Step 2: Synthesis of Ibuprofen
Ibuprofen is formed by catalytic hydrogenation of olefin precursors with hydrogen (H2) in the presence of a catalyst.
Chemical equation: C6H5CH=CH2 + 2H2 → C13H18O2
The olefin catalytic hydrogenation method of Ibuprofen involves the synthesis of olefin precursors from styrene and formic acid, and then catalytic hydrogenation of the olefin precursors with hydrogen in the presence of a catalyst to form Ibuprofen. This method uses catalysts to promote the hydrogenation reaction of olefin molecules, reduce unsaturated bonds to saturated bonds, and gradually build the structure of Ibuprofen. During the synthesis process, it is necessary to pay attention to selecting a suitable catalyst, controlling the reaction conditions and temperature, and performing purification and purification steps to obtain high-purity Ibuprofen.
6. Propylene oxide rearrangement method
A new synthetic method for ibuprofen, wherein p-isobutylacetophenone is prepared and converted from 2-(4-isobutylphenyl) propanal
The 2-step reaction of turning into ibuprofen is the same as that of the classic Darzens condensation method.
The outlined steps of the propylene oxide rearrangement method:
Step 1: Preparation of Propylene Oxide
Propylene is reacted with excess ammonium persulfate to produce propylene oxide.
Chemical equation: C3H6 + (NH4)2S2O8 → C3H6O
Step 2: Propylene oxide ring-opening reaction
Under alkaline conditions, react propylene oxide with tartaric acid (Tartaric acid), and a ring-opening reaction occurs.
Chemical equation: C3H6O + H2C4H4O6 → C9H14O3
Step 3: rearrangement reaction
Under high temperature conditions, the ring-opened product undergoes a rearrangement reaction.
Chemical equation: C9H14O3 → C13H18O2
The propylene oxide rearrangement of Ibuprofen is synthesized through two main steps. First, propylene and ammonium persulfate react to form propylene oxide. Then, under alkaline conditions, propylene oxide reacts with tartaric acid to undergo a ring-opening reaction to obtain a ring-opened product. Finally, under high temperature conditions, a rearrangement reaction is carried out to obtain Ibuprofen. This method gradually builds the structure of Ibuprofen through ring-opening and rearrangement reactions of propylene oxide.



