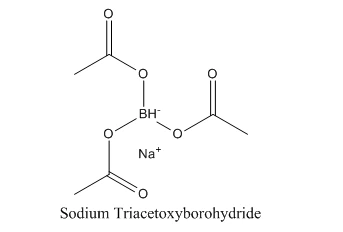
Sodium Triacetoxyborohydride (link:https://www.bloomtechz.com/synthetic-chemical/organic-intermediates/sodium-triacetoxyborohydride-cas-56553-60-7.html) is a strong reducing agent, also known as STAB or STAB-Na, and its chemical formula is NaBH(OAc)3. Stable to a certain extent, it can be stored for several months to several years at room temperature, and will not be easily decomposed. However, the compound is prone to decomposition under extreme conditions such as high temperature or high humidity, so it must be preserved properly to ensure its quality. The compound is stable and easy to store. These physical properties make it an important reducing agent and play an important role in organic synthesis.

The following is a brief introduction about the main chemical properties of the compound:
1. Reducibility:
Sodium Triacetylborohydride is a strong reducing agent that can reduce many organic compounds to lower oxidation states. For compounds containing oxygen functional groups, such as aldehydes, ketones, acids and esters, Sodium Triacetoxyborohydride will usually be selectively reduced to the corresponding alcohols or hydroxyl compounds. For compounds containing sulfur functional groups, such as mercaptans and disulfides, the reducing agent also has strong reducing properties.
2. Reactivity:
In many organic syntheses, Sodium Triacetoxyborohydride is also used as a catalyst for reduction reactions. In these reactions, the compound is typically added to the reaction system to react with other chemicals. For example, it can react with carboxylic acid anhydrides to generate corresponding alcohols, or react with aromatic ketones to generate corresponding aromatic alcohols, etc. In addition, Sodium Triacetoxyborohydride can also be used as a catalyst for condensation reactions, such as condensation reactions between carboxylic acids and amines.
3. Stability:
Although Sodium Triacetoxyborohydride is a strong reducing agent, it is more stable than other commonly used reducing agents such as sodium borohydride. During storage and use, the compound is not easily affected by conditions such as air, moisture and temperature. At the same time, it should also be noted that Sodium Triacetoxyborohydride should avoid contact with oxidizing agents, such as hydrogen peroxide or potassium permanganate, otherwise dangerous reactions will occur.
4. Reversibility:
The reduction reaction of Sodium Triacetoxyborohydride is reversible, so some chemical transformations can be selectively performed by controlling the reaction conditions. For example, by controlling the position of the electronically hindering group, the carbonyl group in aromatic ketones can be selectively reduced without affecting the reaction at other positions.
5. Specificity:
Sodium Triacetylborohydride has high specificity in the reduction reaction. For example, in polyfunctional carboxylic acids or ketones, only one functional group will be reduced to the corresponding alcohol or hydroxyl compound, while the other functional groups are not affected. This makes Sodium Triacetoxyborohydride highly selective in organic synthesis and can reduce the generation of other side reactions.
In conclusion, Sodium Triacetoxyborohydride, as an important reducing agent, has strong reducibility and stability. It has high specificity and selectivity in chemical reactions, so it is widely used in the fields of drug synthesis, organic synthesis and material science. The specific uses are as follows:
1. Reduction reaction:
As a strong reducing agent, Sodium Triacetylborohydride can be widely used in reduction reactions in organic synthesis. It can reduce many organic compounds to lower oxidation states. For compounds containing oxygen functional groups, such as aldehydes, ketones, acids and esters, Sodium Triacetoxyborohydride will usually be selectively reduced to the corresponding alcohols or hydroxyl compounds. For compounds containing sulfur functional groups, such as mercaptans and disulfides, the reducing agent also has strong reducing properties.
2. Condensation reaction:
In addition to playing an important role in the reduction reaction, Sodium Triacetoxyborohydride can also act as a catalyst in the condensation reaction. It can react with carboxylic acid anhydrides to generate corresponding alcohols, or react with aromatic ketones to generate corresponding aromatic alcohols, etc.

3. Synthesis of Heterocyclic Compounds:
Sodium Triacetylborohydride also plays an important role in the synthesis of heterocyclic compounds. It can reduce various nitrogen-containing compounds, such as pyridine, carbazole, pyrimidine, pyran, thiophene and thiazole, etc., to the corresponding dihydro compounds. In addition, Sodium Triacetoxyborohydride can also act as a catalyst in cycloaddition reactions, promoting the formation of normally difficult two-membered and three-membered rings.
4. Catalyst:
Sodium Triacetylborohydride also plays an important role in many catalytic reactions. For example, it is widely used in N-alkylation reaction, cycloaddition reaction, arene hybridization reaction, amidation reaction, olefin polymerization reaction, etc. In addition, it can also catalyze acyl transfer reactions and decarboxylation reactions, etc.
5. Drug synthesis:
Sodium Triacetylborohydride is also widely used in drug synthesis. By selectively reducing or transforming specific functional groups in drug molecules, new compounds can be synthesized, resulting in more effective drug molecules. For example, in the preparation of the anticancer drug Ifosfamide, Sodium Triacetoxyborohydride can be used to remove the carbonyl of aromatic hydrocarbons.
6. Materials Science:
Sodium Triacetylborohydride is also widely used in materials science. It can be used to synthesize nanoparticles, nanowires and nanotubes with certain shape and size. In addition, it can also be used as a reducing agent for electroless plating, the preparation of nanomaterials of metals and their oxides, etc.

7. Semiconductor industry:
Sodium Triacetylborohydride is also widely used in the semiconductor industry. It can be used to prepare high-quality p-type materials. During the preparation of semiconductor materials, Sodium Triacetoxyborohydride is usually added to the synthesis reaction system to control the concentration of impurities such as iron and cobalt in the existing materials and prevent the migration of impurity elements.
In conclusion, Sodium Triacetoxyborohydride is an important organic synthesis reagent with strong reducibility and stability. It has high specificity and selectivity in chemical reactions, so it is widely used in the fields of drug synthesis, organic synthesis and material science.

