Larocaine Hydrochloride is a local anesthetic also known as Dimethocaine Hydrochloride with the chemical name 3-(diethylamino)-2,2-dimethyl-propyl-4-aminobenzoate hydrochloride. In terms of chemical structure, Larocaine Hydrochloride is a benzoate ester substance with a molecular formula of C16H26N2O2·HCl and a molecular weight of 324.85.
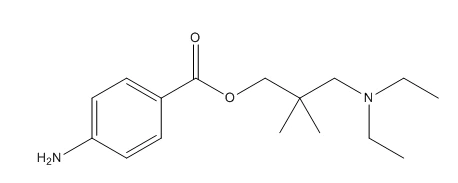
Larocaine Hydrochloride has a deep and long-lasting anesthetic effect, but requires a higher dosage. Its narcotic effect is similar to that of cocaine, but the duration of action is shorter. Larocaine Hydrochloride is a crystalline, usually white or yellowish solid. It has high water solubility and is easy to dissolve in organic solvents such as ethanol.
1. Prepare 3,4-dimethoxy-N-nitrous acid cyclohexylbenzamide by reacting 3,4-dimethoxybenzaldehyde and nitrous acid cyclohexane, and then react with isopropylamine and 3 , Larocaine Hydrochloride was obtained by removing salt after reacting with 4-dichlorobenzoic acid.
The synthetic method of Larocaine Hydrochloride is as follows:
1.1. Dissolve 3,4-dimethoxybenzaldehyde (1 mmol) in absolute ethanol (5 mL) under an ice bath.
1.2. Cyclohexane nitrite (1.2 mmol) was added dropwise to the reaction solution while keeping the temperature at 0-5°C.
1.3. Stir the reaction solution slowly and let it react at room temperature for 24 hours.
1.4. The reaction solution was concentrated to dryness, and then the product was washed with chloroform (10 mL).
1.5. The washings were concentrated and washed with ethanol (5 mL) to obtain 3,4-dimethoxy-N-nitrous acid cyclohexylbenzamide.
Next, we will synthesize Larocaine Hydrochloride by reacting with isopropylamine and 3,4-dichlorobenzoic acid respectively.
Synthetic method 1: by reacting with isopropylamine:
3,4-Dimethoxy-N-nitrosocyclohexylbenzamide (1 mmol) and isopropylamine (3 mmol) were reacted in ethyl acetate (5 mL). The reaction steps are as follows:
1. Put 3,4-dimethoxy-N-nitrosocyclohexylbenzamide (1 mmol) and isopropylamine (3 mmol) into ethyl acetate (5 mL).
2. Slowly add a drop of sulfuric acid, followed by cuprous chloride (0.05 mmol).
3. Stir the reaction at room temperature for 12 hours.
4. The reaction solution was concentrated to dryness, and then the product was washed with ethanol (5 mL).
The product synthesized by this method is 3,4-dimethoxy-N-(1-methylethyl)-benzene-1-carboxamide.
Synthesis method 2: by reacting with 3,4-dichlorobenzoic acid:
3,4-Dimethoxy-N-nitrosocyclohexylbenzamide (1 mmol) and 3,4-dichlorobenzoic acid (1 mmol) were reacted in THF (5 mL). The reaction steps are as follows:
1. Put 3,4-dimethoxy-N-nitrosocyclohexylbenzamide (1 mmol) and 3,4-dichlorobenzoic acid (1 mmol) in THF (5 mL).
2. Add DCC (1 mmol) and stir the reaction at room temperature for 18 hours.
3. Add ultrasonic waves to form a precipitate.
4. Filter the reaction solution and wash the product with ice-cold acetone.
The product synthesized by this method is Larocaine.
Finally, Larocaine and hydrochloric acid are reacted to obtain Larocaine Hydrochloride through the salt removal method. The reaction steps are as follows:
1. Dissolve Larocaine (1mmol) in acetone and add HCl (1mmol).
2. Stir the reaction solution under ice bath for 2 hours until a precipitate forms.
3. Filter the precipitate and wash the product with chloroform.
The product synthesized by this method is Larocaine Hydrochloride.
Larocaine Hydrochloride is prepared by reacting 3,4-dimethoxybenzaldehyde and cyclohexane nitrite to 3,4-dimethoxy-N-cyclohexyl nitrite benzamide, and then reacted with isopropylamine and 3,4-Dichlorobenzoic acid can be prepared by removing salt after reaction. The above steps are the conventional synthesis method of this compound, but it can also be synthesized by other reaction modes.

2. Prepare N-(3,4-dichlorobenzyl)-N-isopropylamine compound by reacting 3,4-dichlorobenzaldehyde and isopropylamine, and then condense with methylcarbene or ethylcarbene respectively After the reaction, Larocaine Hydrochloride is obtained by removing the salt. Specific steps are as follows:
2.1. Preparation of experimental compounds:
The N-(3,4-dichlorobenzyl)-N-isopropylamine compound was prepared by reacting 3,4-dichlorobenzaldehyde with isopropylamine. This step can be achieved by adding 3,4-dichlorobenzaldehyde and isopropylamine to dry tetrahydrofuran, then adding LDA (lithium diisopropylamide) and allowing it to react at room temperature. The reaction time is about 2 hours, and the resulting N-(3,4-dichlorobenzyl)-N-isopropylamine compound needs to be purified and dried in air.
2.2. Methylcarbene condensation:
First, methylcarbene needs to be prepared. The compound can be prepared by reacting chloroform in the presence of methylmagnesium or methyllithium. After the methylcarbene is obtained, it can be added to N-(3,4-dichlorobenzyl)-N-isopropylamine compound for condensation reaction. During the reaction process, it is necessary to stir in ice water, and gradually add glacial acetic acid until the pH value of the solution reaches 6.5. The reaction time is 4 hours. After the reaction, the compound generated needs to be filtered, and the organic phase is separated, and dried by rotary evaporation to obtain a dry solid product.
2.3. Ethyl carbene condensation:
The preparation of ethyl carbene is similar to that of methyl carbene. In the presence of ethyl magnesium or ethyl lithium, chloroform reacts with chloroform to obtain ethyl carbene. Then it was added to N-(3,4-dichlorobenzyl)-N-isopropylamine compound for condensation reaction. During the reaction process, it is necessary to stir in ice water, and gradually add glacial acetic acid until the pH value of the solution reaches 6.5. The reaction time is 2 h, and the compound formed after the reaction needs to be filtered, and the organic phase is separated, and dried by rotary evaporation to obtain a dry solid product.
2.4. Prepare LRC by removing salt:
The resulting product needs to be desalted to prepare LRC. The resulting solid product was dissolved in a solvent, and hydrochloric acid was added, and stirred to mix. The reaction temperature needs to be kept below -20°C, as the concentration of the solution increases gradually, the particles are gradually formed. At this time, it is necessary to remove impurities through processes such as filtration and washing, and obtain the target product LRC.
Prepare N-(3,4-dichlorobenzyl)-N-isopropylamine compound by the reaction of 3,4-dichlorobenzaldehyde and isopropylamine, and then react with methylcarbene or ethylcarbene, by Dismantle the salt to get Larocaine Hydrochloride. Different conditions and reaction conditions can affect the product quality. In the experiment, considering various factors such as reaction conditions, solvent selection, and reactant quality, it is necessary to select a suitable scheme to ensure product quality.
Generally speaking, although the chemical synthesis methods of Larocaine Hydrochloride are complicated, there are many kinds, and different methods can be selected for synthesis according to the needs.

