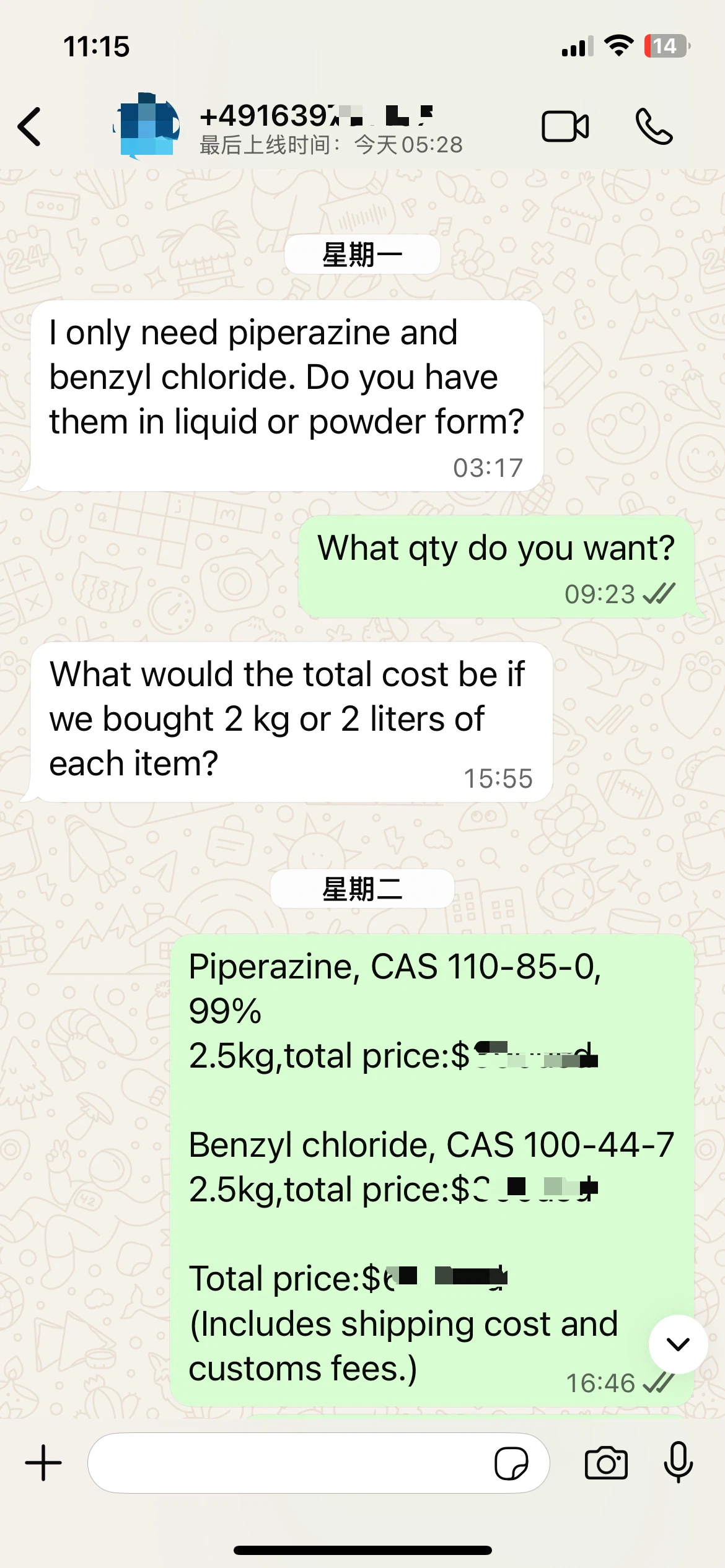
Piperazine and benzyl chloride are two fundamental chemicals widely used in pharmaceuticals, agrochemicals, and industrial processes. Piperazine, a heterocyclic organic compound with a six-membered ring containing two nitrogen atoms, serves as a building block for numerous drugs, including antipsychotics and antihistamines. Benzyl chloride, an aromatic organochlorine compound, is a versatile intermediate in the synthesis of fragrances, dyes, and polymers.
|
|
|
|
Chemical Structures

Piperazine
Piperazine (C₄H₁₀N₂) is a six-membered heterocyclic compound featuring two nitrogen atoms at opposite positions in the ring. Its structure resembles piperidine (a five-membered nitrogen-containing ring) but with an additional methylene bridge, conferring greater flexibility and reactivity. Industrially, piperazine is often encountered as its hexahydrate form, (CH₂CH₂NH)₂·6H₂O, which melts at 44°C and boils at 125–130°C. The compound is colorless, hygroscopic, and exhibits a characteristic ammonia-like odor.
Piperazine's basicity (pKₐ ≈ 9.8) arises from the lone pairs on its nitrogen atoms, enabling it to act as a nucleophile or ligand in coordination chemistry. Its derivatives, such as 1-benzylpiperazine (BZP) and dibenzylpiperazine (DBZP), are formed by substituting one or both nitrogen atoms with benzyl groups, altering their pharmacological profiles.
Benzyl Chloride
Benzyl chloride (C₆H₅CH₂Cl) is an aromatic compound consisting of a benzene ring attached to a methylene chloride group. It is a colorless liquid with a pungent odor, slightly soluble in water but miscible with organic solvents. Its density (9.2 lb/gal) and boiling point (179°C) reflect its nonpolar nature. Benzyl chloride is highly reactive due to the polarized C-Cl bond, making it a potent electrophile in nucleophilic substitution reactions.
The compound's toxicity is notable: it acts as a lachrymator (causing eye irritation) and is corrosive to skin, mucous membranes, and metals. Its flash point (153°F) underscores its flammability risk, necessitating careful handling.

Industrial and Pharmaceutical Significance
Piperazine: From Anthelmintics to CNS Drugs
Piperazine's medicinal history dates to the early 20th century when Bayer marketed it as an anthelmintic. Today, its derivatives are integral to:
Antipsychotics: Fluphenazine and trifluoperazine, used to treat schizophrenia, incorporate piperazine rings to enhance dopamine receptor binding.
Antihistamines: Cyclizine and meclizine, which alleviate nausea, rely on piperazine for their antiemetic effects.
Stimulants: BZP, though banned in many countries, was once sold as a legal alternative to ecstasy, highlighting the fine line between therapeutic and recreational use.
Piperazine's industrial utility extends to corrosion inhibitors, emulsifiers, and gas sweetening agents, showcasing its versatility.
Benzyl Chloride: A Cornerstone of Aromatic Chemistry
Benzyl chloride's industrial footprint is vast:
Fragrances: Benzyl salicylate and benzyl benzoate, common in perfumes, are derived from benzyl chloride.
Pharmaceuticals: It is a precursor to ephedrine (a decongestant) and benzylpenicillin (a penicillin antibiotic).
Polymers: Benzyl-functionalized resins are used in chromatography and ion exchange.
However, its toxicity has led to stricter regulations, particularly in consumer products where residual benzyl chloride must be minimized.
Industrial Applications

Gas Treatment and Carbon Capture
Piperazine plays a critical role in amine scrubbing, a process used to remove acidic gases like CO₂ and H₂S from industrial emissions:
Power Plants: Mixed with solvents like monoethanolamine (MEA), piperazine forms stable carbamates, capturing CO₂ with higher efficiency than MEA alone. Its lower vapor pressure reduces solvent loss during regeneration, lowering energy costs.
Natural Gas Processing: Piperazine-based scrubbers purify natural gas by removing sulfur compounds, preventing corrosion in pipelines and meeting environmental standards.
Polymer and Material Science
Piperazine's reactivity enables the synthesis of advanced materials:
Polyamides: Reacting piperazine with diacids (e.g., adipic acid) produces nylon-like polymers used in textiles and engineering plastics.
Epoxy Resins: Piperazine-cured epoxies exhibit enhanced thermal stability and chemical resistance, making them ideal for aerospace and automotive coatings.
Ion Exchange Resins: Functionalized with sulfonic or carboxylic groups, piperazine-based resins purify water by removing heavy metals like lead and cadmium.
Gas Treatment and Carbon Capture
Piperazine plays a critical role in amine scrubbing, a process used to remove acidic gases like CO₂ and H₂S from industrial emissions:
Power Plants: Mixed with solvents like monoethanolamine (MEA), piperazine forms stable carbamates, capturing CO₂ with higher efficiency than MEA alone. Its lower vapor pressure reduces solvent loss during regeneration, lowering energy costs.
Natural Gas Processing: Piperazine-based scrubbers purify natural gas by removing sulfur compounds, preventing corrosion in pipelines and meeting environmental standards.

Polymer and Material Science
Piperazine's reactivity enables the synthesis of advanced materials:
Polyamides: Reacting piperazine with diacids (e.g., adipic acid) produces nylon-like polymers used in textiles and engineering plastics.
Epoxy Resins: Piperazine-cured epoxies exhibit enhanced thermal stability and chemical resistance, making them ideal for aerospace and automotive coatings.
Ion Exchange Resins: Functionalized with sulfonic or carboxylic groups, piperazine-based resins purify water by removing heavy metals like lead and cadmium.
Niche Applications
Dyes and Pigments
Azo Dyes: Benzyl chloride reacts with diazonium salts to form azo dyes, which impart vibrant colors to textiles, leather, and paper. These dyes are valued for their fastness to light and washing.
Fluorescent Brighteners: Benzyl-substituted compounds enhance the whiteness of fabrics and plastics by absorbing UV light and re-emitting it as visible blue light, counteracting yellowing.
Surfactants and Emulsifiers
Quaternary Ammonium Compounds: Benzyl chloride reacts with tertiary amines to form benzalkonium chlorides, cationic surfactants used as disinfectants, antiseptics, and fabric softeners. Their benzyl group improves solubility in organic solvents and biocidal activity.
Nonionic Surfactants: Ethoxylated benzyl alcohol derivatives (e.g., laureth-9) act as emulsifiers in cosmetics and detergents, stabilizing oil-water mixtures.
Safety and Environmental Considerations
Despite its utility, benzyl chloride poses significant health and environmental risks:
Toxicity: Exposure via inhalation, ingestion, or skin contact can cause severe burns, respiratory distress, and central nervous system depression. Chronic exposure may lead to liver or kidney damage.
Carcinogenicity: The International Agency for Research on Cancer (IARC) classifies benzyl chloride as a Group 2B carcinogen (possibly carcinogenic to humans), based on animal studies linking it to lung tumors.
Environmental Impact: Benzyl chloride is toxic to aquatic life and persists in the environment if not properly contained. Wastewater treatment plants must remove it to prevent ecosystem harm.
Personal Protective Equipment (PPE): Workers handling benzyl chloride must wear gloves, goggles, and respirators to minimize exposure.
Ventilation: Fume hoods and local exhaust ventilation systems are essential to maintain airborne concentrations below permissible exposure limits (PELs).
Storage and Handling: Benzyl chloride should be stored in cool, well-ventilated areas away from incompatible substances like oxidizers or bases. Spills require immediate containment using inert absorbents.
Piperazine and benzyl chloride, though structurally distinct, are indispensable in chemistry and industry. Piperazine's versatility as a nucleophile has cemented its place in drug design, while benzyl chloride's reactivity enables the synthesis of aromatic compounds. However, their safety profiles demand rigorous handling: piperazine's derivatives require careful regulation, while benzyl chloride's toxicity necessitates stringent controls. As chemistry advances, balancing utility with safety will remain paramount, ensuring these compounds continue to benefit society without compromising human health or the environment.